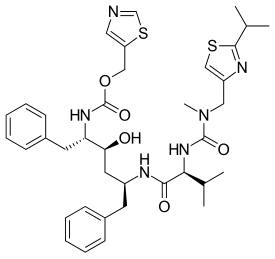
Ritonavir
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /rɪˈtɒnəˌvɪər/ rih-TO-nə-veer |
| Trade names | Norvir |
| Other names | RTV |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a696029 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 98–99% |
| Metabolism | Liver |
| Elimination half-life | 3–5 hours |
| Excretion | Mostly fecal |
| Identifiers | |
| |
JSmol) | |
| |
| |
Ritonavir, sold under the brand name Norvir, is an
Common side effects of ritonavir include nausea, vomiting, loss of appetite, diarrhea, and numbness of the hands and feet.
Ritonavir was patented in 1989 and came into medical use in 1996.
Medical uses
This section needs expansion with: a lead sentence that better describes the very important (pharmacokinetic-pharmakodynamic) PK-PD observations for this first-in-class inhibitor design, and its initial application. You can help by adding to it. (February 2020) |
HIV
Ritonavir is
COVID-19
Two SARS-CoV-2 3CLpro inhibitors are prepackaged with ritonavir to enhance their blood concentration.[20]
In December 2021, the
In January 2023, simnotrelvir/ritonavir was conditionally approved by China's National Medical Products Administration (NMPA) for COVID-19.[20]
Other uses
The use of ritonavir as a CYP3A inhibitor is also seen in the Hepatitis C medication ombitasvir/paritaprevir/ritonavir.[7]
Side effects
When administered at the initially tested higher doses effective for anti-HIV therapy, the side effects of ritonavir are those shown below.[4]
- Asthenia, malaise
- Diarrhea
- Nausea and vomiting
- Abdominal pain
- Dizziness
- Insomnia
- Kidney failure
- Sweating
- Taste abnormality
- Metabolic effects, including
- Hypercholesterolemia
- Hypertriglyceridemia
- Elevated transaminases
- Elevated creatine kinase
Adverse drug reactions
Ritonavir exhibits hepatic activity.[27] It induces CYP1A2 and inhibits CYP3A4 and CYP2D6. Concomitant therapy of ritonavir with a variety of medications may result in serious and sometimes fatal drug interactions.[28]
Due to it being a strong inhibitor (that causes at least a five-fold increase in the plasma
CYP3A4 inducers can counteract the inhibiting effects of ritonavir and lead to drastically reduced levels of "boosted" drugs, increasing the risk of developing drug resistance. Other CYP3A4 inhibitors may have an additive effect with ritonavir, causing increased drug levels.[7]
Pharmacology
This section needs expansion with: with an accurate description of both its HIV protease and CYP3A4 binding characteristics and structural details. You can help by adding to it. (February 2020) |

Pharmacodynamics
Ritonavir was originally developed as an
Ritonavir is rarely used for its own antiviral activity but remains widely used as a booster of other
Pharmacokinetics
The capsules of the medication do not have the same bioavailability as the tablets.[6]
Ritonavir was demonstrated to have an in vitro potency of EC50=0.02µM on HIV-1 protease and highly sustained concentration in plasma after oral administration in several species.[36]
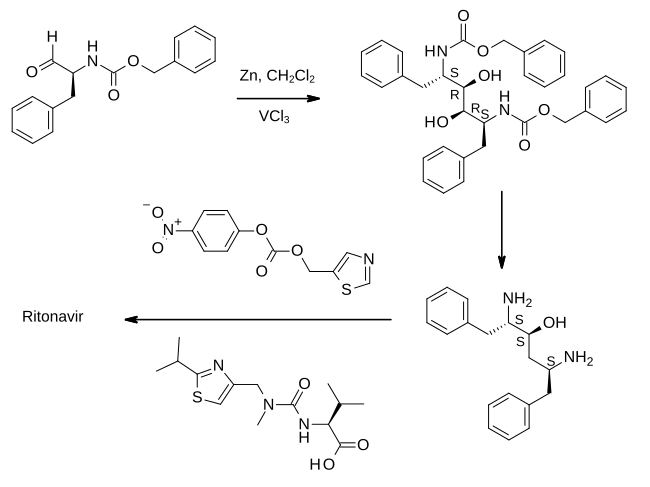
Chemistry
Ritonavir was initially derived from a moderately potent and orally bioavailable small molecule, A-80987. The P3 and P2′ heterocyclic groups of A-80987 were redesigned to create an analogue, now known as ritonavir, with improved pharmacokinetic properties to the original.[36]
Full details of the synthesis of ritonavir were first published by scientists from Abbott Laboratories.
In the first step shown, an
History

Ritonavir is sold as Norvir by AbbVie, Inc.[4][5] The US Food and Drug Administration (FDA) approved ritonavir on March 1, 1996,[39][40] As a result of the introduction of "highly active antiretroviral thearap[ies]" the annual U.S. HIV-associated death rate fell from over 50,000 to about 18,000 over a period of two years.[38]
In 2014, the FDA approved a combination of ombitasvir/paritaprevir/ritonavir for the treatment of hepatitis C virus (HCV) genotype 4.[8]
After the start of the COVID pandemic in 2020, many antivirals, including protease inhibitors in general and ritonavir in particular, were repurposed in an effort to treat the new infection. Lopinavir/ritonavir was found not to work in severe COVID-19.[41] Virtual screening followed by molecular dynamics analysis predicted ritonavir blocks the binding of the SARS-CoV-2 spike (S) protein to the human angiotensin-converting enzyme 2 (hACE2) receptor, which is critical for the virus entry into human cells.[42]
Finally in 2021, a combination of ritonavir with nirmatrelvir, a newly developed orally active 3C-like protease inhibitor, was developed for the treatment of COVID-19.[43][44][45][46] Ritonavir serves to slow down metabolism of nirmatrelvir by cytochrome enzymes to maintain higher circulating concentrations of the main drug.[47] In November that year, Pfizer announced positive phase 2/3 results, including 89% reduction in hospitalizations when given within three days after symptom onset.[48][49]
Polymorphism and temporary market withdrawal
Ritonavir was originally dispensed as a capsule that did not require refrigeration. This contained a crystal form of ritonavir that is now called form I.
During development—ritonavir was introduced in 1996—only the crystal form now called form I was found; however, in 1998, a lower free energy,[52] more stable polymorph, form II, was discovered. This more stable crystal form was less soluble, which resulted in significantly lower bioavailability. The compromised oral bioavailability of the drug led to temporary removal of the oral capsule formulation from the market.[51] As a consequence of the fact that even a trace amount of form II can result in the conversion of the more bioavailable form I into form II, the presence of form II threatened the ruin of existing supplies of the oral capsule formulation of ritonavir; and indeed, form II was found in production lines, effectively halting ritonavir production.[50] Abbott (now AbbVie) withdrew the capsules from the market, and prescribing physicians were encouraged to switch to a Norvir suspension.[citation needed] It has been estimated that Abbott lost more than US$250 million as a result, and the incident is often cited as a high-profile example of disappearing polymorphs.[53]
The company's research and development teams ultimately solved the problem by replacing the capsule formulation with a refrigerated gelcap.[when?][citation needed] In 2000, Abbott (now AbbVie) received FDA-approval for a tablet formulation of lopinavir/ritonavir (Kaletra) which contained a preparation of ritonavir that did not require refrigeration.[54] Ritonavir tablets produced in a solid dispersion by melt-extrusion was found to remain in form I, and was re-introduced commercially in 2010.[55]
Society and culture
Economics
In 2003, Abbott (AbbVie, Inc.) raised the price of a Norvir course from US$1.71 per day to US$8.57 per day, leading to claims of price gouging by patients' groups and some members of Congress. Consumer group Essential Inventions petitioned the NIH to override the Norvir patent, but the NIH announced on August 4, 2004, that it lacked the legal right to allow generic production of Norvir.[56]
References
- FDA. Retrieved October 22, 2023.
- ^ "Notice: Nirmatrelvir (COVID-19) added to Prescription Drug List (PDL)". Health Canada. January 17, 2022. Archived from the original on May 29, 2022. Retrieved June 25, 2022.
- ^ "Norvir Product information". Health Canada. April 25, 2012. Retrieved June 25, 2022.
- ^ a b c d e "Norvir- ritonavir tablet, film coated Norvir- ritonavir solution Norvir- ritonavir powder". DailyMed. Archived from the original on November 18, 2021. Retrieved November 17, 2021.
- ^ a b c d "Norvir EPAR". European Medicines Agency (EMA). September 17, 2018. Archived from the original on October 2, 2018. Retrieved August 20, 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ a b c d e f g h i j k "Ritonavir". The American Society of Health-System Pharmacists. Archived from the original on October 17, 2015. Retrieved October 23, 2015.
- ^ PMID 31335032. Retrieved January 6, 2024.
- ^ a b "FDA approves Viekira Pak to treat hepatitis C" (Press release). U.S. Food and Drug Administration (FDA). December 19, 2014. Archived from the original on October 31, 2015.
- PMID 36423149.
- ^ "Ritonavir Pregnancy and Breastfeeding Warnings". drugs.com. Archived from the original on September 7, 2015. Retrieved October 23, 2015.
- ^ ISBN 9780857111562.
- ISBN 9780080919225. Archivedfrom the original on June 17, 2020. Retrieved September 10, 2017.
- ISBN 9783527607495. Archivedfrom the original on June 20, 2021. Retrieved August 27, 2020.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "First Generic Drug Approvals". U.S. Food and Drug Administration (FDA). Archived from the original on January 26, 2021. Retrieved February 13, 2021.
- ^ "Ritonavir - Patient | NIH". clinicalinfo.hiv.gov. Retrieved January 11, 2023.
- ^ "Ritonavir". go.drugbank.com. Retrieved January 11, 2023.
- ^ "Protease | NIH". clinicalinfo.hiv.gov. Retrieved January 11, 2023.
- ^ PMID 37705591.
- ^ "Paxlovid- nirmatrelvir and ritonavir kit". DailyMed. Archived from the original on December 31, 2021. Retrieved December 30, 2021.
- ^ a b c "FDA Authorizes First Oral Antiviral for Treatment of COVID-19". U.S. Food and Drug Administration (FDA) (Press release). December 22, 2021. Archived from the original on December 27, 2021. Retrieved December 22, 2021.
- ^ a b "Pfizer Receives U.S. FDA Emergency Use Authorization for Novel COVID-19 Oral Antiviral Treatment" (Press release). Pfizer. December 22, 2021. Archived from the original on December 22, 2021. Retrieved December 22, 2021 – via Business Wire.
- ^ "Frequently Asked Questions on the Emergency Use Authorization for Paxlovid for Treatment of COVID-19" (PDF). U.S. Food and Drug Administration (FDA). December 22, 2021. Archived from the original on January 5, 2022. Retrieved January 3, 2022.
- ^ "Oral COVID-19 antiviral, Paxlovid, approved by UK regulator" (Press release). Medicines and Healthcare products Regulatory Agency. December 31, 2021. Archived from the original on January 11, 2022. Retrieved January 3, 2022.
- ^ Reed J (December 31, 2021). "Paxlovid: UK medicines regulator approves second Covid antiviral pill". BBC News Online. Archived from the original on January 1, 2022. Retrieved January 3, 2022.
- S2CID 39632668.
- ^ "Ritonavir: Drug Information Provided by Lexi-Comp: Merck Manual Professional". Merck Manuals Professional Edition. April 30, 2008. Archived from the original on April 30, 2008.
- PMID 32556272.
- PMID 31038898.
- ^ a b WO 1994014436, Kempf DJ, Norbeck DW, Sham HL, Zhao C, Sowin TJ, Reno DS, Haight AR, Cooper AJ, "Retroviral protease inhibiting compounds", published July 7, 1994, assigned to Abbott Laboratories.
- PMID 2213822.
- PMID 14657084.
- PMID 9084785.
- ^ "Drug Development and Drug Interactions: Table of Substrates, Inhibitors and Inducers". U.S. Food and Drug Administration (FDA). December 3, 2019. Archived from the original on March 12, 2020. Retrieved February 17, 2020.
- ^ PMID 9484509.
- S2CID 75449475.
- ^ PMID 21637182. Archived from the original(PDF) on September 24, 2015.
- ^ "Ritonavir FDA approval package" (PDF). March 1, 1996. Archived (PDF) from the original on March 1, 2021. Retrieved August 20, 2020.
- ^ "FDA Approval of HIV Medicines". HIVINFO. Retrieved September 29, 2022.
- PMID 32187464.
- S2CID 222217607.
- PMID 34029993.
- S2CID 236450769.
- PMID 34502033.
- ^ "Pfizer begins dosing in Phase II/III trial of antiviral drug for Covid-19". Clinical Trials Arena. September 2, 2021. Archived from the original on November 5, 2021. Retrieved November 5, 2021.
- ^ Woodley M (October 19, 2021). "What is Australia's potential new COVID treatment?". The Royal Australian College of General Practitioners (RACGP). Archived from the original on November 5, 2021. Retrieved November 6, 2021.
- ^ "Pfizer's Novel COVID-19 Oral Antiviral Treatment Candidate Reduced Risk Of Hospitalization Or Death By 89% In Interim Analysis Of Phase 2/3 EPIC-HR Study". November 5, 2021. Archived from the original on November 16, 2021. Retrieved November 17, 2021.
- ^ Weintraub K (November 5, 2021). "Pfizer antiviral drug could nearly end deaths from COVID-19, company study suggests". USA Today. Archived from the original on November 5, 2021. Retrieved November 5, 2021.
- ^ S2CID 20923508.
- ^ PMID 12604798.
- .
- PMID 26031248.
- ^ "Kaletra FAQ". AbbVie's Kaletra product information. AbbVie. 2011. Archived from the original on July 7, 2014. Retrieved July 5, 2014.
- PMID 31537981.
- ^ Connolly C (August 5, 2004). "NIH Declines to Enter AIDS Drug Price Battle". The Washington Post. Archived from the original on August 20, 2008. Retrieved January 16, 2006.
Further reading
- Chemburkar SR, Bauer J, Deming K, Spiwek H, Patel K, Morris J, et al. (2000). "Dealing with the Impact of Ritonavir Polymorphs on the Late Stages of Bulk Drug Process Development". Organic Process Research & Development. 4 (5): 413–417. .