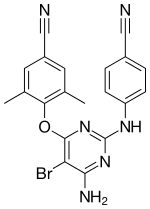
Etravirine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Intelence |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608016 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 99.9% |
| Metabolism | Hepatic (CYP3A4, CYP2C9 & CYP2C19-mediated) |
| Elimination half-life | 41±20 hours |
| Excretion | Faeces (93.7%), urine (1.2%) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Etravirine (ETR,
Etravirine is licensed in the United States, Canada, Israel, Russia, Australia, New Zealand and the European Union,[5] and is under regulatory review in Switzerland.[6]
Indications and dosage
Etravirine, in combination with other anti-retrovirals, is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in antiretroviral treatment-experienced adult patients, who have evidence of viral replication and HIV-1 strains resistant to a non-nucleoside reverse transcriptase inhibitor (NNRTI) and other antiretroviral agents.
The recommended dose of etravirine is 200 mg (2 x 100 mg tablets, or 1 x 200 mg tablet as of 03/18/2011) taken twice daily following a meal. The type of food does not affect the exposure to etravirine.[7]
Contraindication
Each 100 mg etravirine tablet contains 160 mg of
Mechanism of action
Etravirine is a second-generation
Warnings and risks
In 2009, the prescribing information for etravirine was modified to include "postmarketing reports of cases of Stevens–Johnson syndrome, toxic epidermal necrolysis and erythema multiforme, as well as hypersensitivity reactions characterized by rash, constitutional findings, and sometimes organ dysfunction, including liver failure. Intelence therapy should be immediately discontinued when signs and symptoms of severe skin or hypersensitivity reactions develop."[11]
Repositioning
Etravine has been studied for use in a drug repositioning application. In a paper[12] published in the medical journal Movement Disorders, etravirine was shown to cause an increase in frataxin production. Frataxin deficiency is a key component to Friedreich's ataxia, a genetically inherited disease that causes the progressive loss of coordination and muscle strength leading to motor incapacitation and the full-time use of a wheelchair.
Chemistry
Etravine forms as colourless orthorhombic crystals in space group Pna21.[13] The structures of these and of a number of solvate and salt forms have been reported.[13][14]
References
- Department of Health and Human Services. Archived from the originalon 2012-08-31.
- PMID 17933730.
- ^ "FDA Approves HIV Drug Etravirine". Associated Press. January 18, 2008.[dead link]
- ^ "First New NNRTI in Nearly a Decade to Benefit Canadians with HIV/AIDS" (PDF) (Press release). Janssen-Ortho Inc. 2008-04-01. Archived from the original (PDF) on 2010-11-02. Retrieved 2008-07-09.
- ^ "Intelence receives marketing authorisation in the European Union for HIV combination therapy". Tibotec. Archived from the original on 2011-09-28. Retrieved 2008-08-29.
- ^ "Etravirine (TMC125, Intelence) granted accelerated approval in US". aidsmap. Archived from the original on 2010-01-02. Retrieved 2008-01-24.
- ^ "Intelence prescribing information" (PDF). FDA. Retrieved January 19, 2012.
- ^ "Etravine: Summary of product characteristics" (PDF). EMEA. p. 5. Archived from the original (PDF) on August 20, 2016. Retrieved July 13, 2011.
- ^ Evans D (2008-01-15). "Etravirine—Countdown to Launch". AIDSmeds.com. Archived from the original on 2008-01-19. Retrieved 2008-02-02.
- PMID 15115397.
- ^ "FDA Medwatch Safety Information". Food and Drug Administration. Retrieved 2009-08-27.
- S2CID 58567610.
- ^ ISSN 1528-7483.
- S2CID 243761396.
External links
- "Etravirine". Drug Information Portal. U.S. National Library of Medicine.
