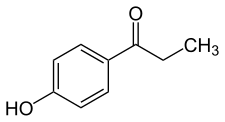
Paroxypropione
Appearance
 | |
| Clinical data | |
|---|---|
| Trade names | Frenantol, Frenormon, Hypophenon, Paroxon, Possipione, Profenone, others |
| Other names | Paraoxypropiophenone; H-365; NSC-2834; 4'-Hydroxypropiophenone; Ethyl p-hydroxyphenyl ketone; p-Propionylphenol; Paroxypropiophenone; Parahydroxypropiophenone; PHP |
| Drug class | Nonsteroidal estrogen; Antigonadotropin |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
Paroxypropione, also known as paraoxypropiophenone, is a
synthetic nonsteroidal estrogen which has been used medically as an antigonadotropin in Spain and Italy but appears to no longer be marketed.[1][2][3][4] It was first synthesized in 1902.[1] The antigonadotropic properties of the drug were discovered in 1951[3] and it entered clinical use shortly thereafter.[5]
Pharmacology
Pharmacodynamics
Paroxypropione is closely related structurally to
affinity for the estrogen receptor[4] and must be given at high dosages to achieve significant estrogenic and antigonadotropic effects, for instance, 0.8 to 1.6 g/day.[10][11] It possesses 0.1% of the estrogenic activity and less than 0.5% of the antigonadotropic potency of estrone.[12]
Chemistry
Synthesis
The highest reported yield, approximately 96%, is from the between
esterification to give phenyl propionate, which then undergoes a Fries rearrangement
.
Derivatives
Paroxypropione is a precursor in the chemical synthesis of diethylstilbestrol and dienestrol.[14][15]
Society and culture
Names
Brand names Frenantol, Frenormon, Hypophenon, Paroxon, Possipione, Profenone, numerous others; former developmental code name NSC-2834), also known as paroxypropiophenone (P.O.P.) or 4'-hydroxypropiophenone.
Research
Paroxypropione was studied and used in the treatment of breast cancer.[16][17][18]
References
- ^ ISBN 978-1-4757-2085-3.
- ISBN 978-3-88763-075-1.
- ^ PMID 14861299.
- ^ S2CID 19751228.
- ISSN 0022-3263.
- ISBN 978-3-642-67265-1.
- PMID 426279.
- ISBN 978-0-12-382185-0.
- S2CID 12342018.
- ^ De Vega R (1955). "Protein breakdown before and after operations. Influence of growth hormone and of inhibitors of the pituitary adrenal system". Cirug., Ginecol. Urol. 9: 289–326.
- PMID 5006470.[permanent dead link]
- PMID 12980070.
- hdl:2115/44794.
- ISBN 978-0-8155-1856-3.
- ISBN 978-0-471-85545-3.
- PMID 13241536.
- S2CID 22364847.
- PMID 5188348.
Further reading
- Gustavo RP (July 1958). "[Anti-gonadotropic action of possipione]" [Anti-gonadotropic action of possipione]. Quaderni di Clinica Ostetrica e Ginecologica (in Italian). 13 (7): 307–15. PMID 13579130.
