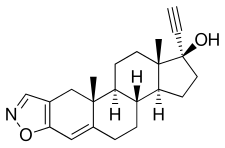
Danazol
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Danatrol, Danocrine, Danol, Danoval, others |
| Other names | WIN-17757; 2,3-Isoxazolethisterone; 2,3-Isoxazol-17α-ethynyltestosterone; 17α-Ethynyl-17β-hydroxyandrost-4-en-[2,3-d]isoxazole |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682599 |
| Pregnancy category |
|
| ATC code | |
| Legal status | |
| Legal status | |
| Metabolism | Liver (CYP3A4)[9][6] |
| Metabolites | • 2-OHM-Ethisterone[6] • Ethisterone[7][8] |
| Elimination half-life | Acute: 3–10 hours[9][2] Chronic: 24–26 hours[9] |
| Excretion | Urine, feces[9][2] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Danazol, sold as Danocrine and other brand names, is a medication used in the treatment of
The use of danazol is limited by
Danazol was discovered in 1963 and was introduced for medical use in 1971.
Medical uses
Danazol is used primarily in the treatment of
Available forms
Danazol comes in the form of 50, 100, and 200 mg oral capsules.[2] It is taken at a dose of 50 to 400 mg two or three times per day, for a total of 100 to 800 mg per day depending on the indication.[2]
Contraindications
Danazol is
Since danazol is metabolized by the liver, it cannot be used by patients with liver disease, and in patients receiving long-term therapy, liver function must be monitored on a periodic basis.[24]
Side effects
Androgenic side effects are of concern, as some women taking danazol may experience unwanted hair growth (
The use of danazol for endometriosis has been linked to an increased risk of
Pharmacology
Pharmacodynamics
Danazol possesses a complex
Modulation of steroid hormone receptors
Danazol is described as a possessing high affinity for the
| Steroid | PR | AR | ER | GR | MR | SHBG | CBG
|
|---|---|---|---|---|---|---|---|
| Danazol | 9 | 8 | ? | <0.2a | ? | 40 | 10 |
| Ethisterone | 35 | <1 | <1 | <1b | <1 | 92–121 | 0.33 |
| 5α-Dihydroethisterone | 12 | 38–100c | 4 | 120b | ? | 100 | ? |
| Notes: Values are percentages (%). Reference ligands (100%) were | |||||||
| Receptor | Affinity |
Action |
|---|---|---|
| Androgen receptor | 90 | Agonist |
| Progesterone receptor | 6,000 | Agonist–antagonist
|
| Glucocorticoid receptor | 5,000 | Agonist |
| Estrogen receptor | 80,000 | Agonist |
| Sources: [4][5] | ||
Inhibition of steroidogenesis enzymes
Danazol has been found to act as an
In accordance with its steroidogenesis inhibition, clinical studies have demonstrated that danazol directly and markedly inhibits adrenal, ovarian, and testicular steroidogenesis in vivo.[5] The enzymatic production of estradiol, progesterone, and testosterone have all specifically been found to be inhibited.[5]
| Enzyme | Affinity (Ki) | Inhibition type | Estimated inhibition at 2 μM |
|---|---|---|---|
| Cholesterol side-chain cleavage enzyme | 20 μM | Competitive | ? |
| 3β-Hydroxysteroid dehydrogenase/Δ5-4 isomerase | 5.8 μM | Competitive | 4.3% |
17α-Hydroxylase |
2.4 μM | Competitive | 2.9% |
17,20-Lyase |
1.9 μM | Competitive | 3.9% |
| 17β-Hydroxysteroid dehydrogenase | 4.4 μM | Competitive | 15% |
| 21-Hydroxylase | 0.8 μM | Competitive | 37% |
11β-Hydroxylase |
1 μM | Competitive | 21% |
| Aromatase | >100 μM | – | 0% |
| Sources: [5] | |||
For reference, circulating concentrations of danazol are in the range of 2 μM at a dosage of 600 mg/day in women.[5]
Occupation and downregulation of carrier proteins
| Group | Free | Albumin | SHBG |
|---|---|---|---|
| Normal (no danazol) | 1% | 39% | 60% |
| Danazol treatment | 3% | 79% | 18% |
| Sources: [5] | |||
Danazol is known to bind to two steroid hormone carrier proteins:
As can be seen, the percentage of free testosterone is tripled in women being treated with danazol.[5][35] The ability of danazol to increase free testosterone levels suggests that a portion of its weak androgenic effects are mediated indirectly by facilitating the activity of testosterone and dihydrotestosterone through the displacement of them from SHBG.[5][35] In addition to binding to and occupying SHBG however, danazol also decreases the hepatic production of SHBG and therefore SHBG levels, and so downregulation of SHBG may be involved as well.[4][5] Danazol likely decreases hepatic production of SHBG by reducing estrogenic and increasing androgenic activity in the liver (as androgens and estrogens decrease and increase, respectively, hepatic SHBG synthesis).[36] In accordance with the notion that suppression of SHBG is involved in the androgenic effects of danazol, the drug has synergistic rather than additive androgenic effects in combination with testosterone in bioassays (which is most likely secondary to the increased free testosterone levels).[16]
It is noteworthy that 2-hydroxymethylethisterone, a major metabolite of danazol, circulates at concentrations 5–10 times greater than those of danazol and is twice as potent as danazol in displacing testosterone from SHBG.[37] As such, most of the occupation of SHBG by danazol may actually be due to this metabolite.[37]
Antigonadotropic activity
Via its weak progestogenic and androgenic activity, through activation of the PR and AR in the
Mechanism of action in endometriosis
Because danazol reduces estrogen production and levels,[38] it has functional antiestrogenic properties.[41] The combination of its antiestrogenic, androgenic, and progestogenic or antiprogestogenic actions cause atrophy of the endometrium, which alleviates the symptoms of endometriosis.[4][5][16][38][42]
Effects in men
In men, danazol has been found to inhibit gonadotropin secretion and markedly decrease testosterone levels, likely due to its actions as a steroidogenesis inhibitor and antigonadotropin.[43] However, even at the highest dosage assessed (800 mg/day), spermatogenesis remained unaffected.[43]
Pharmacokinetics
The
Danazol is
Chemistry
Danazol, also known as 2,3-isoxazol-17α-ethynyltestosterone or as 17α-ethynyl-17β-hydroxyandrost-4-en-[2,3-d]isoxazole, is a
History
Danazol was synthesized in 1963 by a team of scientists at Sterling Winthrop in Rensselaer, New York by a team that included Helmutt Neumann, Gordon Potts, W.T. Ryan, and Frederik W. Stonner.[17][18] It was approved by the U.S. Food and Drug Administration in 1971 as the first drug in the country to specifically treat endometriosis.[14][19]
Society and culture
Generic names
Danazol is the
Brand names
Danazol is or has been marketed under many brand names throughout the world including Anargil, Azol, Benzol, Bonzol, Cyclolady, Cyclomen, Danal, Danalol, Danamet, Danamin, Danasin, Danatrol, Danazant, Danazol, Danocrine, Danodiol, Danogen, Danokrin, Danol, Danonice, Danoval, Danzol, Dogalact (
Availability
Danazol is available in the United States, Europe, and widely elsewhere throughout the world.[9][11][47]
Research
Danazol has been studied in the treatment of
Low-dose danazol has been investigated in the treatment of
A 2016 phase I/II prospective study orally administered 800 mg per day to 27 patients with telomere diseases. The primary efficacy endpoint was a 20% reduction in the annual rate of telomere attrition measured. Toxic effects formed the primary safety endpoint. The study was halted early, after telomere attrition was reduced in all 12 patients who could be evaluated. 12 of 27 patients achieved the primary efficacy end point, 11 of whom increased telomere length at 24 months. Hematologic responses (secondary efficacy endpoint) occurred in 10 of 12 patients who could be evaluated at 24 months. Elevated liver-enzyme levels and muscle cramps (known adverse effects) of grade 2 or less occurred in 41% and 33% of the patients, respectively.[52]
References
- FDA. Retrieved 22 Oct 2023.
- ^ a b c d e f g h i j k l m n o p q r s "Danocrine Brand of Danazol Capsules, USP" (PDF). Sanofi-Aventis U.S. LLC. U.S. Food and Drug Administration.
- ^ ISBN 978-0-08-052583-9.
- ^ ISBN 978-3-540-78355-8.
- ^ ISBN 978-94-011-3864-2.
- ^ ISBN 978-1-60913-345-0.
- ^ ISBN 978-3-527-64565-7.
- ^ ISBN 978-1-4757-3889-6.
- ^ a b c d e f g h i j k Brayfield A, ed. (30 October 2013). "Danazol". Martindale: The Complete Drug Reference. Pharmaceutical Press. Retrieved 1 April 2014.
- ^ ISBN 978-1-4757-2085-3.
- ^ ISBN 978-3-88763-075-1.
- ^ ISBN 978-94-011-4439-1.
- PMID 17943735.
- ^ ISBN 978-1-4963-1521-2.
- ^ ISBN 978-0-8493-5973-6.
- ^ ISBN 978-0-08-049451-7.
- ^ PMID 4318768.
- ^ ISBN 9780030603211.
- ^ PMID 5538758.
- ISBN 978-0-443-06911-6.
- PMID 18930222.
- PMID 20541792.
- ^ ISBN 9780071716727. Archived from the originalon March 28, 2013.
- ^ "Danazol: MedlinePlus Drug Information". medlineplus.gov. Retrieved 2018-11-07.
- PMID 14613992.
- S2CID 42150333.
- S2CID 21336380.
- PMID 359134.
- PMID 7382482.
- PMID 7195405.
- S2CID 2702353.
- S2CID 24785562.
- ^ S2CID 25273633.
- S2CID 45817485.
- ^ ISBN 978-0-7817-5734-8.
- ISBN 978-1-4822-2286-9.
- ^ ISBN 978-3-642-82486-9.
- ^ ISBN 978-0-7817-3160-7.
- ^ PMID 6104649.
- ^ ISBN 978-0-07-176401-8.
- ISBN 978-0-387-98569-5.
- PMID 2113661.
- ^ PMID 4422449. Archived from the originalon 2018-09-04. Retrieved 2018-03-04.
Danazol (2-3-isoxazol-17alpha-ethinyl-testosterone)
- ISBN 978-1-61779-222-9.
- ^ "PRODUCT INFORMATION" (PDF). Therapeutic Goods Administration. June 21, 2005.
- ISBN 978-81-85790-03-9.
- ^ a b c d "Danazol : Uses, Dosage, Side Effects".
- ISBN 978-0-8493-5973-6.
- ISBN 978-3-662-22346-8.
- ^ "A Safety and Efficacy Study of Oral Danazol (a Previously Approved Drug)in the Treatment of Diabetic Macular Edema". Clinicaltrials.gov. Ampio Pharmaceuticals. Inc. Retrieved 27 June 2015.
- ^ "Ampio Pharmaceuticals Announces Additional Statistically Significant Study Results for Optina™ in the Treatment of Diabetic Macular Edema (DME)". Ampio Pharmaceuticals. Inc. Archived from the original on 29 June 2015. Retrieved 27 June 2015.
- PMID 27192671.
Further reading
- Dmowski WP (March 1979). "Endocrine properties and clinical application of danazol". Fertility and Sterility. 31 (3): 237–251. PMID 374128.
- Fraser IS (April 1979). "Danazol--a steroid with a unique combination of actions". Scottish Medical Journal. 24 (2): 147–150. S2CID 11557840.
- Potts GO, Schane HP, Edelson J (May 1980). "Pharmacology and pharmacokinetics of danazol". Drugs. 19 (5): 321–330. S2CID 39178416.
- Jenkin G (May 1980). "Review: The mechanism of action of danazol, a novel steroid derivative". The Australian & New Zealand Journal of Obstetrics & Gynaecology. 20 (2): 113–118. S2CID 29935606.
- Barbieri RL, Ryan KJ (October 1981). "Danazol: endocrine pharmacology and therapeutic applications". American Journal of Obstetrics and Gynecology. 141 (4): 453–463. PMID 7025640.
- Dmowski WP (January 1990). "Danazol. A synthetic steroid with diverse biologic effects". The Journal of Reproductive Medicine. 35 (1 Suppl): 69–74, discussion 74–5. PMID 2404115.
- Barbieri RL (1990). "Danazol: molecular, endocrine, and clinical pharmacology". Progress in Clinical and Biological Research. 323: 241–252. PMID 2406750.
- Hughes E, Brown J, Tiffin G, Vandekerckhove P (January 2007). "Danazol for unexplained subfertility". The Cochrane Database of Systematic Reviews (1): CD000069. PMID 17253444.
- Beaumont H, Augood C, Duckitt K, Lethaby A (July 2007). "Danazol for heavy menstrual bleeding". The Cochrane Database of Systematic Reviews. 2007 (3): CD001017. PMID 17636649.
- Selak V, Farquhar C, Prentice A, Singla A (October 2007). "Danazol for pelvic pain associated with endometriosis". The Cochrane Database of Systematic Reviews (4): CD000068. PMID 17943735.
- Craig TJ (2008). "Appraisal of danazol prophylaxis for hereditary angioedema". Allergy and Asthma Proceedings. 29 (3): 225–231. PMID 18387221.
- Ke LQ, Yang K, Li J, Li CM (July 2009). "Danazol for uterine fibroids". The Cochrane Database of Systematic Reviews. 2009 (3): CD007692. PMID 19588442.
- Letchumanan P, Thumboo J (February 2011). "Danazol in the treatment of systemic lupus erythematosus: a qualitative systematic review". Seminars in Arthritis and Rheumatism. 40 (4): 298–306. PMID 20541792.
