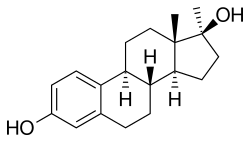
Methylestradiol
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Ginecosid, Ginecoside, Mediol, Renodiol |
| Other names | NSC-52245; 17α-Methylestradiol; 17α-ME; 17α-Methylestra-1,3,5(10)-triene-3,17β-diol |
| Routes of administration | By mouth[1] |
| Drug class | Estrogen |
| Identifiers | |
| |
JSmol) | |
| |
| |
Methylestradiol, sold under the brand names Ginecosid, Ginecoside, Mediol, and Renodiol, is an
Methylestradiol is or has been marketed in Brazil, Venezuela, and Indonesia.[3] In addition to its use as a medication, methylestradiol has been studied for use as a radiopharmaceutical for the estrogen receptor.[7]
Medical uses
Methylestradiol is used in combination with the progestin and androgen/anabolic steroid
Available forms
Methylestradiol is marketed in combination with normethandrone in the form of oral tablets containing 0.3 mg methylestradiol and 5 mg normethandrone.[8][9]
Side effects
Pharmacology
Pharmacodynamics
Methylestradiol is an
Methylestradiol is an
| Compound | PR | AR | ER | GR | MR | SHBG | CBG
|
|---|---|---|---|---|---|---|---|
| Estradiol | 2.6 | 7.9 | 100 | 0.6 | 0.13 | 8.7 | <0.1 |
| Ethinylestradiol | 15–25 | 1–3 | 112 | 1–3 | <1 | ? | ? |
| Methylestradiol | 3–10, 15–25 | 1–3 | 67 | 1–3 | <1 | ? | ? |
| Methyltestosterone | 3 | 45, 100–125 | ? | 1–5 | ? | 5 | ? |
| Normethandrone | 100 | 146 | <0.1 | 1.5 | 0.6 | ? | ? |
| Sources: Values are percentages (%). Reference | |||||||
Pharmacokinetics
Due to the presence of its C17α
Chemistry
Methylestradiol, or 17α-methylestradiol (17α-ME), also known as 17α-methylestra-1,3,5(10)-triene-3,17β-diol, is a
History
Methylestradiol was first marketed, alone as Follikosid and in combination with methyltestosterone as Klimanosid, in 1955.[17][18][19][20]
Society and culture
Generic names
Methylestradiol has not been assigned an
Brand names
Methylestradiol is or has been marketed under the brand names Ginecosid, Ginecoside, Mediol, and Renodiol, all in combination with normethandrone.[3][2]
Availability
Methylestradiol is or has been marketed in Brazil, Venezuela, and Indonesia.[3]
References
- ^ PMID 13673847.
- ^ ISBN 978-1-4757-2085-3.
- ^ a b c d e f g h i j k "Methylestradiol". Drugs.com. Retrieved 2 January 2016.
- ^ ISBN 978-92-832-1291-1.
- ^ ISBN 978-3-642-67570-6.
- ^ PMID 359134.
- PMID 6406650.
- ^ Unlisted Drugs. Pharmaceutical Section, Special Libraries Association. 1982.
Batynid. C. Each dragee contains: normethandrone, 5 mg.; and methylestradiol, 0.3 mg. E. (Formerly) Gynaekosid. M. Boehringer Biochemia, Florence. A. Estrogenic; Rx of secondary amenorrhea. R. Notiz Med Farm 32;295, Nov-Dec 81.
- S2CID 38008851.
- ^ ISBN 978-3-540-79088-4.
- ISBN 978-0-9828280-1-4.
- PMID 1964199.
- ISBN 9780120603084.
- PMID 3695484.
- PMID 7421203.
- ^ Falconi G, Rossi GL, Ercoli A (September 1970). James VH (ed.). Quinestrol and other cyclopentyl ethers of estrogenic steroids: different rates of storage in body fat. Third International Congress on Hormonal Steroids, Hamburg. International Congress Series No. 210. Amsterdam, Excerpta Medica. pp. 218–219. Archived from the original on 28 March 2018.
- S2CID 1678069.
- ISBN 978-3-7091-5694-0.
- ISBN 978-3-662-36329-4.
- ^ Helwig B (1956). Moderne Arzneimittel: eine Spezialitätenkunde nach Indikationsgebieten für Ärzte und Apotheker. Wissenschaftliche Verlagsgesellschaft. p. 240.
