Etynodiol diacetate
 | |
| Clinical data | |
|---|---|
| Trade names | Ovulen, Demulen, others |
| Other names | Ethynodiol diacetate; Norethindrol diacetate; 3β-Hydroxynorethisterone 3β,17β-diacetate;[1] 17α-Ethynylestr-4-ene-3β,17β-diyl diacetate; CB-8080; SC-11800 |
| License data |
|
| Pregnancy category |
|
Progestin; Progestogen ester | |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
JSmol) | |
| |
| |
Etynodiol diacetate, or ethynodiol diacetate, sold under the brand name Ovulen among others, is a
Etynodiol diacetate is a progestin, or a
Etynodiol, a related compound, was discovered in 1954, and etynodiol diacetate was introduced for medical use in 1965.[15][16] The combination ethynodiol with mestranol (Ovulen) was approved for medical use in the United States in 1966.[17] The combination ethinylestradiol with ethynodiol (Demulen) was approved for medical use in the United States in 1970.[18]
In 2021, the combination with ethinylestradiol was the 276th most commonly prescribed medication in the United States, with more than 800,000 prescriptions.[19][20]
Medical uses
Etynodiol diacetate is used in combination with an
Side effects
Pharmacology

Etynodiol diacetate is virtually inactive in terms of
The pharmacokinetics of etynodiol diacetate have been reviewed.[22]
| Compound | Typea | PR | AR | ER | GR | MR | SHBG | CBG
|
|---|---|---|---|---|---|---|---|---|
| Norethisterone | – | 67–75 | 15 | 0 | 0–1 | 0–3 | 16 | 0 |
| 5α-Dihydronorethisterone | Metabolite | 25 | 27 | 0 | 0 | ? | ? | ? |
| 3α,5α-Tetrahydronorethisterone | Metabolite | 1 | 0 | 0–1 | 0 | ? | ? | ? |
| 3α,5β-Tetrahydronorethisterone | Metabolite | ? | 0 | 0 | ? | ? | ? | ? |
| 3β,5α-Tetrahydronorethisterone | Metabolite | 1 | 0 | 0–8 | 0 | ? | ? | ? |
| Ethinylestradiol | Metabolite | 15–25 | 1–3 | 112 | 1–3 | 0 | 0.18 | 0 |
| Norethisterone acetate | Prodrug | 20 | 5 | 1 | 0 | 0 | ? | ? |
| Norethisterone enanthate | Prodrug | ? | ? | ? | ? | ? | ? | ? |
| Noretynodrel | Prodrug | 6 | 0 | 2 | 0 | 0 | 0 | 0 |
| Etynodiol | Prodrug | 1 | 0 | 11–18 | 0 | ? | ? | ? |
| Etynodiol diacetate | Prodrug | 1 | 0 | 0 | 0 | 0 | ? | ? |
| Lynestrenol | Prodrug | 1 | 1 | 3 | 0 | 0 | ? | ? |
| Notes: Values are percentages (%). Reference , or neither of norethisterone. Sources: See template. | ||||||||
Chemistry
Etynodiol diacetate, also known as 3β-hydroxy-17α-ethynyl-19-nortestosterone 3β,17β-diaceate, 3β-hydroxynorethisterone 3β,17β-diacetate, or 17α-ethynylestr-4-ene-3β,17β-diol 3β,17β-diacetate, is a
Synthesis
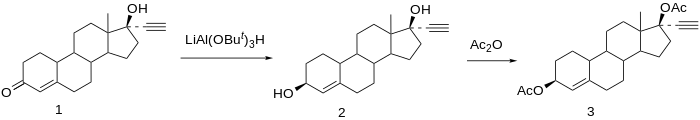
Chemical syntheses of etynodiol diacetate have been published.[22]
Reduction of norethisterone (1) affords the 3,17-diol. The 3β-hydroxy compound is the desired product; since reactions at C3 do not show nearly the stereoselectivity as those at C17 by virtue of the relative lack of stereo-directing proximate substituents, the formation of the desired isomer is engendered by use of a bulky reducing agent, lithium tri-tert-butoxyaluminum hydride. Acetylation of the 3β,17β-diol affords etynodiol diacetate (3).[23]
History
Etynodiol was first synthesized in 1954, via reduction of norethisterone, and etynodiol diacetate was introduced for medical use in 1965.[15][16]
Society and culture
Generic names
Etynodiol diacetate is the
Brand names
Etynodiol diacetate is or has been marketed under brand names including Conova, Continuin, Demulen,[18][25] Femulen, Kelnor,[3][25] Lo-Malmorede,[26] Luteonorm, Luto-Metrodiol, Malmorede,[27] Metrodiol, Ovulen,[17][25] Soluna, Zovia,[2] and others.[5][6][7]
Availability
Etynodiol diacetate is marketed in only a few countries, including the United States, Canada, Argentina, and Oman.[7]
References
- ^ PMID 14670641.
- ^ a b "Zovia 1/35- ethynodiol diacetate and ethinyl estradiol tablets kit". Archived from the original on 28 September 2022. Retrieved 20 January 2024.
- ^ a b "Kelnor 1/35- ethynodiol diacetate and ethinyl estradiol kit". Archived from the original on 29 March 2023. Retrieved 20 January 2024.
- ISBN 978-1-4612-2730-4.
- ^ ISBN 978-1-4757-2085-3.
- ^ ISBN 978-3-88763-075-1. Retrieved 30 May 2012.
- ^ a b c d e "Etynodiol". Drugs.com. Archived from the original on 5 February 2018. Retrieved 4 February 2018.
- ^ ISBN 978-1-4832-7738-7.
- ^ PMID 2256526.
- ^ ISBN 978-92-832-1291-1.
- ^ ISBN 978-1-4511-1805-6. Archivedfrom the original on 11 January 2023. Retrieved 11 October 2016.
- ^ ISBN 978-0-7817-1750-2. Retrieved 30 May 2012.
- ^ ISBN 978-0-7817-7513-7. Retrieved 30 May 2012.
- ^ S2CID 27018468.
- ^ PMID 4947236.
- ^ ISBN 978-0-8155-1856-3. Archivedfrom the original on 20 January 2024. Retrieved 4 February 2018.
- ^ a b "Ovulen: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 8 December 2022. Retrieved 20 January 2024.
- ^ a b "Demulen: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 10 May 2021. Retrieved 20 January 2024.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Ethinyl Estradiol; Ethynodiol - Drug Usage Statistics". ClinCalc. Archived from the original on 18 January 2024. Retrieved 14 January 2024.
- ISBN 978-3-642-73790-9.
- ^ ISBN 978-3-642-99941-3.
- ^ PMID 6064262.
- .
- ^ a b c "Estrogen and Progestin (Oral Contraceptives)". Archived from the original on 18 January 2024. Retrieved 20 January 2024.
- ^ "Lo-Malmorede". Archived from the original on 24 March 2021. Retrieved 20 January 2024.
- ^ "Malmorede". Archived from the original on 1 October 2023. Retrieved 20 January 2024.
