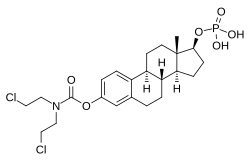
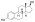Estramustine phosphate
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Emcyt, Estracyt |
| Other names | EMP; Leo 299; NSC-89199; Ro 21-8837/001; Estradiol normustine phosphate; Estradiol 3-normustine 17β-phosphate; Estradiol 3-(bis(2-chloroethyl)carbamate) 17β-(dihydrogen phosphate) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608046 |
| License data |
|
| Pregnancy category |
|
Chemotherapeutic agent; Estrogen; Estrogen ester | |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
JSmol) | |
| |
| |
Estramustine phosphate (EMP), also known as estradiol normustine phosphate and sold under the brand names Emcyt and Estracyt, is a dual
EMP was introduced for medical use in the early 1970s.[3] It is available in the United States, Canada, the United Kingdom, other European countries, and elsewhere in the world.[15][16]
Medical uses
EMP is indicated, in the
Due to its relatively severe side effects and toxicity, EMP has rarely been used in the treatment of prostate cancer.[4] This is especially true in Western countries today.[4] As a result, and also due to the scarce side effects of gonadotropin-releasing hormone modulators (GnRH modulators) like leuprorelin, EMP was almost abandoned.[3] However, encouraging clinical research findings resulted in renewed interest of EMP for the treatment of prostate cancer.[3]
EMP has been used at doses of 140 to 1,400 mg/day orally in the treatment of prostate cancer.[19] However, oral EMP is most commonly used at a dose of 560 to 640 mg/day (280–320 mg twice daily).[1] The recommended dosage of oral EMP in the Food and Drug Administration (FDA) label for Emcyt is 14 mg per kg of body weight (i.e., one 140 mg oral capsule for each 10 kg or 22 lbs of body weight) given in 3 or 4 divided doses per day.[7] The label states that most patients in studies of oral EMP in the United States have received 10 to 16 mg per kg per day.[7] This would be about 900 to 1,440 mg/day for a 90-kg or 200-lb man.[7] Lower doses of oral EMP, such as 280 mg/day, have been found to have comparable effectiveness as higher doses but with improved tolerability and reduced toxicity.[4] Doses of 140 mg/day have been described as a very low dosage.[20] EMP has been used at doses of 240 to 450 mg/day intravenously.[1]
EMP and other estrogens such as polyestradiol phosphate and ethinylestradiol are far less costly than newer therapies such as GnRH modulators, abiraterone acetate, and enzalutamide.[4][21][22] In addition, estrogens may offer significant benefits over other means of androgen deprivation therapy, for instance in terms of bone loss and fractures, hot flashes, cognition, and metabolic status.[4][22]
EMP has been used to prevent the
| Route/form | Estrogen | Dosage | |
|---|---|---|---|
| Oral | Estradiol | 1–2 mg 3x/day | |
| Conjugated estrogens | 1.25–2.5 mg 3x/day | ||
| Ethinylestradiol | 0.15–3 mg/day | ||
| Ethinylestradiol sulfonate | 1–2 mg 1x/week | ||
| Diethylstilbestrol | 1–3 mg/day | ||
| Dienestrol | 5 mg/day | ||
| Hexestrol | 5 mg/day | ||
| Fosfestrol | 100–480 mg 1–3x/day | ||
| Chlorotrianisene | 12–48 mg/day | ||
| Quadrosilan | 900 mg/day | ||
| Estramustine phosphate | 140–1400 mg/day | ||
| Transdermal patch | Estradiol | 2–6x 100 μg/day Scrotal: 1x 100 μg/day | |
SC injection |
Estradiol benzoate | 1.66 mg 3x/week | |
| Estradiol dipropionate | 5 mg 1x/week | ||
| Estradiol valerate | 10–40 mg 1x/1–2 weeks | ||
| Estradiol undecylate | 100 mg 1x/4 weeks | ||
| Polyestradiol phosphate | Alone: 160–320 mg 1x/4 weeks With oral EE: 40–80 mg 1x/4 weeks | ||
| Estrone | 2–4 mg 2–3x/week | ||
IV injection |
Fosfestrol | 300–1200 mg 1–7x/week | |
| Estramustine phosphate | 240–450 mg/day | ||
| Note: Dosages are not necessarily equivalent. Sources: See template. | |||
Available forms
EMP is or has been available in the form of both capsules (140 mg, 280 mg) for oral administration and aqueous solutions (300 mg) for intravenous injection.[19][24][25][7]
Contraindications
EMP is
Side effects
The
Severe adverse effects of EMP are
Adverse
In a small low-dose study using 280 mg/day oral EMP for 150 days, tolerability was significantly improved, with gastrointestinal
System organ class
|
Very common (≥10%) | Common (1–10%) | Frequency unknownc |
|---|---|---|---|
lymphatic system disorders
|
Anemia; Leukopenia | Thrombocytopenia | |
Immune system disorders
|
Hypersensitivity | ||
nutrition disorders
|
Fluid retention
|
||
Psychiatric disorders
|
Confusional state; Depression | ||
Nervous system disorders
|
Lethargy; Headache | ||
Cardiac disorders
|
Congestive heart failure
|
Myocardial infarction | Myocardial ischemia
|
Vascular disorders
|
Embolism | Hypertension | |
Gastrointestinal disorders
|
Nauseab; Vomitingb; Diarrheab | ||
Hepatobiliary disorders
|
Abnormal hepatic function
|
||
Skin and subcutaneous tissue disorders
|
Allergic dermatitis
| ||
connective tissue disorders
|
Muscle weakness | ||
breast disorders
|
Gynecomastia | Erectile dysfunction | |
| General disorders and administration site conditions | IV solution )
| ||
| Footnotes: a = Could not be estimated from available data. b = Especially during the first 2 weeks of therapy. c = Angioedema ( larynx edema) can occur. Often occurs in combination with ACE inhibitors . EMP should be immediately discontinued if angioedema occurs. Sources: See template.
| |||
| Side effect | Estramustine phosphate (oral 11.5–15.9 mg/kg/day) (n = 93) (%) |
Diethylstilbestrol (oral 3.0 mg/day) (n = 93) (%) |
|---|---|---|
respiratory
| ||
| Cardiac arrest | 0 | 2 |
Cerebrovascular accident (stroke) |
2 | 0 |
| Myocardial infarction (heart attack) | 3 | 1 |
| Thrombophlebitis | 3 | 7 |
| Pulmonary embolism | 2 | 5 |
Congestive heart failure |
3 | 2 |
swelling ) |
19 | 17 |
Dyspnea (shortness of breath) |
11 | 3 |
Leg cramps |
8 | 11 |
Upper respiratory discharge |
1 | 1 |
Hoarseness |
1 | 0 |
Gastrointestinal
| ||
| Nausea | 15 | 8 |
| Diarrhea | 12 | 11 |
| Minor gastrointestinal upset |
11 | 6 |
| Anorexia (appetite loss) | 4 | 3 |
| Flatulence | 2 | 0 |
| Vomiting | 1 | 1 |
| Gastrointestinal bleeding | 1 | 0 |
Burning throat |
1 | 0 |
| Thirst | 1 | 0 |
Integumentary
| ||
| Rash | 1 | 4 |
Pruritus (itching) |
2 | 2 |
Dry skin |
2 | 0 |
Pigment changes |
0 | 3 |
Easy bruising |
3 | 0 |
| Flushing | 1 | 0 |
Night sweats |
0 | 1 |
Peeling of fingertip skin |
1 | 0 |
Thinning hair |
1 | 1 |
| Breast changes | ||
Breast tenderness |
66 | 64 |
| Breast enlargement (gynecomastia or male breast development) | ||
| Mild | 60 | 54 |
| Moderate | 10 | 16 |
| Marked | 0 | 5 |
| Miscellaneous | ||
| Lethargy alone | 4 | 3 |
| Depression | 0 | 2 |
| Emotional lability | 2 | 0 |
| Insomnia | 3 | 0 |
| Headache | 1 | 1 |
| Anxiety | 1 | 0 |
| Chest pain | 1 | 1 |
| Hot flashes | 0 | 1 |
| Eye pain | 0 | 1 |
Lacrimation (eye tears) |
1 | 1 |
| Tinnitus | 0 | 1 |
| Laboratory abnormalities | ||
Hematologic (blood )
| ||
| Leukopenia (low white blood cells) | 4 | 2 |
Thrombopenia (low platelets ) |
1 | 2 |
Hepatic (liver)
| ||
| Bilirubin alone | 1 | 5 |
| Bilirubin and LDH | 0 | 1 |
| Bilirubin and AST | 2 | 1 |
| Bilirubin, LDH, and SGOT | 2 | 0 |
| LDH and/or SGOT | 31 | 28 |
| Miscellaneous | ||
| Transient hypercalcemia (high calcium ) |
0 | 1 |
| Sources: [34] | ||
Overdose
There has been no clinical experience with
EMP has been used at high doses of as much as 1,260 mg/day by the
Interactions
EMP has been reported to increase the
Pharmacology
Pharmacodynamics

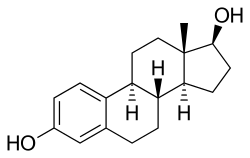
EMP, also known as estradiol normustine phosphate, is a combined
EMP acts by a dual
Extremely high levels of estradiol and estrone occur during EMP therapy.
The mechanism of action of the
The cytostatic metabolites of EMP are accumulated in tissues in a selective manner, for instance in prostate cancer cells.[5][1][4] This may be due to the presence of a specific estramustine-binding protein (EMBP) (Kd = 10–35 nM for estramustine), also known as prostatin or prostatic secretion protein (PSP), which has been detected in prostate cancer, glioma, melanoma, and breast cancer cells.[1][4][44] Because of its tissue selectivity, EMP is said to produce minimal cytostatic effects in healthy tissues, and its tissue selectivity may be responsible for its therapeutic cytostatic efficacy against prostate cancer cells.[5][4][1]
EMP was originally developed as a dual ester prodrug of an estrogen and normustine as a nitrogen mustard alkylating antineoplastic agent which, due to the affinity of the estrogen
Extremely high,
Antigonadotropic effects
EMP at a dosage 280 mg/day has been found to suppress testosterone levels in men into the castrate range (to 30 ng/dL) within 20 days and to the low castrate range (to 10 ng/dL) within 30 days.[3] Similarly, a dosage of 70 mg/day EMP suppressed testosterone levels into the castrate range within 4 weeks.[3]
Pharmacokinetics

| Parameter | IV 300 mg | Oral 420 mg |
|---|---|---|
| Cmax | 506 ± 61 ng/mL | 362 ± 38 ng/mL |
Tmax |
2.6 ± 0.4 hours | 2.2 ± 0.2 hours |
t1/2 |
10.3 ± 0.95 hours | 13.6 ± 3.09 hours |
| AUC0–32 | 4.82 ± 0.62 | 2.88 ± 0.34 |
| Bioavailability | 100.0% | 43.7% ± 4.6% |
| Sources: [31] | ||
| Metabolite | Plasma | Ratio |
|---|---|---|
| Estramustine | 20,000–23,000 pg/mL | 1:9.6–9.8 |
| Estromustine | 191,000–267,000 pg/mL | |
| Estradiol | 4,900–9,000 pg/mL | 1:9.4–11.8 |
| Estrone | 71,000–85,000 pg/mL | |
| Sources: [31] | ||
Upon
The oral
Following a single oral dose of 420 mg EMP in men with prostate cancer, maximal levels of estromustine were 310 to 475 ng/mL (475,000 pg/mL) and occurred after 2 to 3 hours.[1] Estradiol levels with 280 mg/day oral EMP have been found to increase to very high concentrations within one week of therapy.[3] In one study, levels of estradiol were over 20,000 pg/mL after 10 days, were about 30,000 pg/mL after 30 days, and peaked at about 40,000 pg/mL at 50 days.[3] Another study found lower estradiol levels of 4,900 to 9,000 pg/mL during chronic therapy with 560 mg/day oral EMP.[31] An additional study found estradiol levels of about 17,000 pg/mL with 140 mg/day oral EMP and 38,000 pg/mL with 280 mg/day oral EMP.[citation needed] The circulating levels of estradiol and estrone during EMP therapy have been reported to exceed normal levels in men by more than 100- and 1,000-fold, respectively.[4][31] Levels of estramustine and estradiol in the circulation are markedly lower than those of estromustine and estrone, respectively, with a ratio of about 1:10 in both cases.[1][31] Nonetheless, estradiol levels during EMP therapy appear to be similar to those that occur in mid-to-late pregnancy, which range from 5,000 to 40,000 pg/mL.[50] No unchanged EMP is seen in the circulation with oral administration.[1]
The
The

Chemistry
EMP, also known as estradiol 3-normustine 17β-phosphate or as estradiol 3-(bis(2-chloroethyl)carbamate) 17β-(dihydrogen phosphate), is a
Due to its
The molecular weight of EMP sodium is 564.3 g/mol, of EMP meglumine is 715.6 g/mol, of EMP is 520.4 g/mol, of estramustine is 440.4 g/mol, and of estradiol is 272.4 g/mol.[54] As a result of these differences in molecular weights, EMP contains about 52%, EMP sodium about 48%, and EMP meglumine about 38% of the amount of estradiol within their structures as does an equal-mass quantity of estradiol.[54]
| Estrogen | Structure | Ester(s) | Relative mol. weight |
Relative E2 contentb |
log Pc | ||||
|---|---|---|---|---|---|---|---|---|---|
| Position(s) | Moiet(ies) | Type | Lengtha | ||||||
| Estradiol | – | – | – | – | 1.00 | 1.00 | 4.0 | ||
| Estradiol acetate | C3 | Ethanoic acid |
Straight-chain fatty acid | 2 | 1.15 | 0.87 | 4.2 | ||
| Estradiol benzoate | C3 | Benzoic acid | Aromatic fatty acid | – (~4–5) | 1.38 | 0.72 | 4.7 | ||
| Estradiol dipropionate | C3, C17β | Propanoic acid (×2) |
Straight-chain fatty acid | 3 (×2) | 1.41 | 0.71 | 4.9 | ||
| Estradiol valerate | C17β | Pentanoic acid |
Straight-chain fatty acid | 5 | 1.31 | 0.76 | 5.6–6.3 | ||
| Estradiol benzoate butyrate | C3, C17β | Benzoic acid, butyric acid | Mixed fatty acid | – (~6, 2) | 1.64 | 0.61 | 6.3 | ||
| Estradiol cypionate | C17β | Cyclopentylpropanoic acid |
Cyclic fatty acid | – (~6) | 1.46 | 0.69 | 6.9 | ||
Estradiol enanthate |
C17β | Heptanoic acid |
Straight-chain fatty acid | 7 | 1.41 | 0.71 | 6.7–7.3 | ||
Estradiol dienanthate |
C3, C17β | Heptanoic acid (×2) |
Straight-chain fatty acid | 7 (×2) | 1.82 | 0.55 | 8.1–10.4 | ||
| Estradiol undecylate | C17β | Undecanoic acid |
Straight-chain fatty acid | 11 | 1.62 | 0.62 | 9.2–9.8 | ||
| Estradiol stearate | C17β | Octadecanoic acid |
Straight-chain fatty acid | 18 | 1.98 | 0.51 | 12.2–12.4 | ||
| Estradiol distearate | C3, C17β | Octadecanoic acid (×2) |
Straight-chain fatty acid | 18 (×2) | 2.96 | 0.34 | 20.2 | ||
| Estradiol sulfate | C3 | Sulfuric acid | Water-soluble conjugate | – | 1.29 | 0.77 | 0.3–3.8 | ||
| Estradiol glucuronide | C17β | Glucuronic acid | Water-soluble conjugate | – | 1.65 | 0.61 | 2.1–2.7 | ||
| Estramustine phosphated | C3, C17β | Normustine, phosphoric acid | Water-soluble conjugate | – | 1.91 | 0.52 | 2.9–5.0 | ||
| Polyestradiol phosphatee | C3–C17β | Phosphoric acid | Water-soluble conjugate | – | 1.23f | 0.81f | 2.9g | ||
| Footnotes: a = Length of hydrophobicity). Retrieved from PubChem, ChemSpider, and DrugBank. d = Also known as estradiol normustine phosphate. e = Polymer of estradiol phosphate (~13 repeat units ). f = Relative molecular weight or estradiol content per repeat unit. g = log P of repeat unit (i.e., estradiol phosphate). Sources: See individual articles.
| |||||||||
History
EMP was first
Society and culture
Generic names
EMP is provided as the
Brand names
EMP is most commonly marketed under the brand names Estracyt and Emcyt, but has also been sold under a number of other brand names, including Amsupros, Biasetyl, Cellmustin, Estramustin HEXAL, Estramustina Filaxis, Estranovag, Multosin, Multosin Injekt, Proesta, Prostamustin, and Suloprost.[15][16][24]
Availability
EMP is marketed in the United States,[57] Canada, and Mexico under the brand name Emcyt, whereas the medication is marketed under the brand name Estracyt in the United Kingdom and elsewhere throughout Europe as well as in Argentina, Chile, and Hong Kong.[15] It has been discontinued in a number of countries, including Australia, Brazil, Ireland, and Norway.[58]
Research
EMP has been studied in the treatment of other cancers such as glioma and breast cancer.[1] It has been found to slightly improve quality of life in people with glioma during the first 3 months of therapy.[1]
References
- ^ S2CID 1943973.
- ^ S2CID 24616324.
- ^ S2CID 43384150.
- ^ ISBN 978-981-10-7012-9.
- ^ S2CID 36725810.
- ^ a b c "Emcyt (estramustine) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved 8 February 2014.
- ^ a b c d e f g h i j k l "Emcyt estramustine phosphate sodium capsules" (PDF). FDA. Retrieved 14 March 2022.
- ^ PMID 27684806.
- ^ S2CID 33456591.
- ^ S2CID 70507026.
- PMID 9050135.
- ^ PMID 8595142.
- ^ PMID 9274433.
- ^ ISBN 978-3-642-60107-1.
- ^ ISBN 978-3-88763-075-1.
- ^ a b c d e "Estramustine Uses, Side Effects & Warnings".
- ^ a b c d e f g h i j "Estracyt Capsules - Summary of Product Characteristics (SPC)". electronic Medicines Compendium. Pfizer Limited. 12 August 2013. Archived from the original on 22 February 2014. Retrieved 8 February 2014.
- ISBN 978-0-8493-5973-6.
- ^ ISBN 978-0-387-49782-2.
- S2CID 7710924.
- PMID 14644018.
- ^ PMID 10699602.
- PMID 7541359.
- ^ ISBN 978-3-7692-2114-5.
- ISBN 978-0-7817-1750-2.
- S2CID 25425351.
- PMID 7500443.
- ^ S2CID 21465413.
- PMID 17942366.
- PMID 25920994.
- ^ PMID 6375076.
- ^ PMID 86404.
- ^ PMID 6349745.
- ^ "Emcyt estramustine phosphate sodium capsules" (PDF). FDA. Retrieved 14 March 2022.
- ^ ISBN 978-1-4757-2085-3.
- ^ PMID 10076535.
- ^ PMID 2093302.
- ^ PMID 7190620.
- S2CID 31084737.
- ^ PMID 3092893.
- ^ Fredholm, B., Jensen, G., Lindskog, M., & Muntzing, J. (1974, January). Effects of estramustine phosphate (Estracyt) on growth of DMBA-induced mammary tumors in rats. In Acta Pharmacologica et Toxicologica (Vol. 35, pp. 28-28). 35 Norre Sogade, PO Box 2148, DK-1016 Copenhagen, Denmark: Munksgaard Int Publ Ltd.
- ^ PMID 1545778.
- ^ S2CID 25303747.
- PMID 2682681.
- ISBN 978-0-203-09289-7.
- S2CID 22105700.
- ^ S2CID 38350076.
- ^ S2CID 71166897.
- ISBN 978-90-481-9703-3.
- ^ "Estradiol" (PDF). Abbott Laboratories. November 2009.
- ISBN 978-0-12-411562-0.
- ISBN 978-1-4200-0209-6.
- ISBN 978-1-60590-705-5.
- ^ a b "Estramustine phosphate". PubChem. U.S. National Library of Medicine.
- ISBN 978-1-60327-829-4.
- ISBN 978-94-011-4439-1.
- ^ "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 29 January 2017.
- ^ Sweetman, S, ed. (12 February 2013). "Estramustine Sodium Phosphate". Martindale: The Complete Drug Reference. London, UK: Pharmaceutical Press. Retrieved 8 February 2014.
Further reading
- Tew KD (September 1983). "The mechanism of action of estramustine". Seminars in Oncology. 10 (3 Suppl 3): 21–26. PMID 6364362.
- Hoisaeter PA, Bakke A (September 1983). "Estramustine phosphate (Estracyt): experimental and clinical studies in Europe". Seminars in Oncology. 10 (3 Suppl 3): 27–33. PMID 6364363.
- Sandberg AA (September 1983). "Metabolic aspects and actions unique to Estracyt". Seminars in Oncology. 10 (3 Suppl 3): 3–15. PMID 6364364.
- Haukaas SA (1984). "Immunological effects of diethylstilbestrol and estramustine phosphate". Scandinavian Journal of Urology and Nephrology. Supplementum. 83: 1–32. PMID 6387896.
- Hauser AR, Merryman R (May 1984). "Estramustine phosphate sodium". Drug Intelligence & Clinical Pharmacy. 18 (5): 368–374. S2CID 25303747.
- Sandberg AA (June 1984). "Metabolic parameters of Estracyt pertinent to its effects in prostatic cancer". Urology. 23 (6 Suppl): 11–21. PMID 6375075.
- Gunnarsson PO, Forshell GP (June 1984). "Clinical pharmacokinetics of estramustine phosphate". Urology. 23 (6 Suppl): 22–27. PMID 6375076.
- Forsgren B, Björk P (June 1984). "Specific binding of estramustine to prostatic proteins". Urology. 23 (6 Suppl): 34–38. PMID 6375077.
- Kalland T, Haukaas SA (June 1984). "Immunologic effects of estramustine phosphate". Urology. 23 (6 Suppl): 39–45. PMID 6375078.
- Høisaeter PA (June 1984). "Mode of action of Emcyt". Urology. 23 (6 Suppl): 46–48. PMID 6375079.
- Müntzing J, Gunnarsson K (June 1984). "Preclinical pharmacology and toxicology of estramustine phosphate". Urology. 23 (6 Suppl): 6–10. PMID 6375082.
- Slack NH, Murphy GP (June 1984). "Clinical toxicity and long-term results of Emcyt therapy for prostate cancer". Urology. 23 (6 Suppl): 73–77. PMID 6375085.
- Hedlund PO (1985). "Mode of action of estramustine phosphate in hormone dependent and hormone non-dependent prostate cancer". Progress in Clinical and Biological Research. 185A: 197–202. PMID 3898129.
- Hedlund PO (1987). "Estracyt--mode of action and clinical experience". Progress in Clinical and Biological Research. 243B: 215–219. PMID 3309981.
- Murphy GP (1987). "A current review of the clinical experience with Estracyt". Progress in Clinical and Biological Research. 243B: 221–225. PMID 3309982.
- Forsgren B (1988). "Estramustine-binding protein in rat and human prostate". Scandinavian Journal of Urology and Nephrology. Supplementum. 107: 56–58. PMID 3287598.
- Tew KD, Stearns ME (1989). "Intracellular effects of estramustine (Estracyt/Emcyt)". Progress in Clinical and Biological Research. 303: 169–175. PMID 2674983.
- Tew KD, Stearns ME (1989). "Estramustine--a nitrogen mustard/steroid with antimicrotubule activity". Pharmacology & Therapeutics. 43 (3): 299–319. PMID 2682681.
- Könyves I (1989). "Estramustine phosphate (Estracyt) in the treatment of prostatic carcinoma". International Urology and Nephrology. 21 (4): 393–397. S2CID 9727646.
- Benson R, Hartley-Asp B (1990). "Mechanisms of action and clinical uses of estramustine". Cancer Investigation. 8 (3–4): 375–380. PMID 2207764.
- Van Poppel H, Baert L (1991). "The present role of estramustine phosphate in advanced prostate cancer". Progress in Clinical and Biological Research. 370: 323–341. PMID 1924466.
- Janknegt RA (1992). "Estramustine phosphate and other cytotoxic drugs in the treatment of poor prognostic advanced prostate cancer". The Prostate. Supplement. 4: 105–110. S2CID 40562112.
- Tew KD, Glusker JP, Hartley-Asp B, Hudes G, Speicher LA (December 1992). "Preclinical and clinical perspectives on the use of estramustine as an antimitotic drug". Pharmacology & Therapeutics. 56 (3): 323–339. PMID 1301594.
- Kreis W (1995). "Estramustine revisited". Concepts, Mechanisms, and New Targets for Chemotherapy. Cancer Treatment and Research. Vol. 78. Springer. pp. 163–184. PMID 8595142.
- Perry CM, McTavish D (July 1995). "Estramustine phosphate sodium. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy in prostate cancer". Drugs & Aging. 7 (1): 49–74. S2CID 36725810.
- Hudes G (February 1997). "Estramustine-based chemotherapy". Seminars in Urologic Oncology. 15 (1): 13–19. PMID 9050135.
- Bergenheim AT, Henriksson R (February 1998). "Pharmacokinetics and pharmacodynamics of estramustine phosphate". Clinical Pharmacokinetics. 34 (2): 163–172. S2CID 1943973.
- Kitamura T (February 2001). "Necessity of re-evaluation of estramustine phosphate sodium (EMP) as a treatment option for first-line monotherapy in advanced prostate cancer". International Journal of Urology. 8 (2): 33–36. S2CID 43384150.
- Ravery V, Fizazi K, Oudard S, Drouet L, Eymard JC, Culine S, et al. (December 2011). "The use of estramustine phosphate in the modern management of advanced prostate cancer". BJU International. 108 (11): 1782–1786. S2CID 33456591.
- Qin Z, Li X, Zhang J, Tang J, Han P, Xu Z, et al. (September 2016). "Chemotherapy with or without estramustine for treatment of castration-resistant prostate cancer: A systematic review and meta-analysis". Medicine. 95 (39): e4801. PMID 27684806.