Estriol (medication)
| Drug class | Estrogen |
|---|---|
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| Melting point | 82 to 86 °C (180 to 187 °F) (experimental) |
| Solubility in water | 0.119 mg/mL (20 °C) |
| |
| |
| (verify) | |
Estriol (E3), sold under the brand name Ovestin among others, is an
Estriol is well-
Estriol was first discovered in 1930,
Medical uses
Estriol is used in
| Route/form | Estrogen | Low | Standard | High | |||
|---|---|---|---|---|---|---|---|
| Oral | Estradiol | 0.5–1 mg/day | 1–2 mg/day | 2–4 mg/day | |||
| Estradiol valerate | 0.5–1 mg/day | 1–2 mg/day | 2–4 mg/day | ||||
| Estradiol acetate | 0.45–0.9 mg/day | 0.9–1.8 mg/day | 1.8–3.6 mg/day | ||||
| Conjugated estrogens | 0.3–0.45 mg/day | 0.625 mg/day | 0.9–1.25 mg/day | ||||
| Esterified estrogens | 0.3–0.45 mg/day | 0.625 mg/day | 0.9–1.25 mg/day | ||||
| Estropipate | 0.75 mg/day | 1.5 mg/day | 3 mg/day | ||||
| Estriol | 1–2 mg/day | 2–4 mg/day | 4–8 mg/day | ||||
| Ethinylestradiola | 2.5–10 μg/day | 5–20 μg/day | – | ||||
| Nasal spray | Estradiol | 150 μg/day | 300 μg/day | 600 μg/day | |||
| Transdermal patch | Estradiol | 25 μg/dayb | 50 μg/dayb | 100 μg/dayb | |||
Transdermal gel |
Estradiol | 0.5 mg/day | 1–1.5 mg/day | 2–3 mg/day | |||
Vaginal |
Estradiol | 25 μg/day | – | – | |||
| Estriol | 30 μg/day | 0.5 mg 2x/week | 0.5 mg/day | ||||
SC injection |
Estradiol valerate | – | – | 4 mg 1x/4 weeks | |||
| Estradiol cypionate | 1 mg 1x/3–4 weeks | 3 mg 1x/3–4 weeks | 5 mg 1x/3–4 weeks | ||||
| Estradiol benzoate | 0.5 mg 1x/week | 1 mg 1x/week | 1.5 mg 1x/week | ||||
| SC implant | Estradiol | 25 mg 1x/6 months | 50 mg 1x/6 months | 100 mg 1x/6 months | |||
| Footnotes: a = No longer used or recommended, due to health concerns. b = As a single patch applied once or twice per week (worn for 3–4 days or 7 days), depending on the formulation. Note: Dosages are not necessarily equivalent. Sources: See template. | |||||||
Available forms (except USA)
Estriol is available in
Estriol was originally marketed in the 1930s in the form of oral capsules containing 0.06, 0.12, or 0.24 mg estriol under the brand names Theelol (Parke-Davis) and Estriol (Lilly, Abbott).[30][31][32][33][34] Subsequently, many decades later, oral tablets containing 0.35, 1, or 2 mg estriol were introduced under brand names such as Gynäsan, Hormomed, Ovestin, and Ovo-Vinces.[35]
Contraindications
General contraindications of estrogens include breast cancer, endometrial cancer, and others.[19] In animals, estriol is contraindicated during pregnancy and in ferrets.[17]
Side effects
Estriol is well-
Overdose
Estrogens and other
Interactions
Interactions with estriol might be expected to be similar to those of estradiol.[40] No interactions with estriol have been reported in animals.[17] However, it should not be used in combination with other drugs that suppress bone marrow production in dogs.[17]
Pharmacology
Pharmacodynamics
Estriol is an estrogen, or an
Estriol is a much less
The weak and atypical estrogenicity of estriol is thought to be related to its short
Although estriol is an estrogen, it has also been reported to have mixed
| Ligand | Other names | Relative binding affinities (RBA, %)a |
Absolute binding affinities (Ki, nM)a |
Action | ||
|---|---|---|---|---|---|---|
ERα |
ERβ |
ERα |
ERβ
| |||
| Estradiol | E2; 17β-Estradiol | 100 | 100 | 0.115 (0.04–0.24) | 0.15 (0.10–2.08) | Estrogen |
| Estrone | E1; 17-Ketoestradiol | 16.39 (0.7–60) | 6.5 (1.36–52) | 0.445 (0.3–1.01) | 1.75 (0.35–9.24) | Estrogen |
| Estriol | E3; 16α-OH-17β-E2 | 12.65 (4.03–56) | 26 (14.0–44.6) | 0.45 (0.35–1.4) | 0.7 (0.63–0.7) | Estrogen |
| Estetrol | E4; 15α,16α-Di-OH-17β-E2 | 4.0 | 3.0 | 4.9 | 19 | Estrogen |
| Alfatradiol | 17α-Estradiol | 20.5 (7–80.1) | 8.195 (2–42) | 0.2–0.52 | 0.43–1.2 | Metabolite |
16-Epiestriol |
16β-Hydroxy-17β-estradiol | 7.795 (4.94–63) | 50 | ? | ? | Metabolite |
17-Epiestriol |
16α-Hydroxy-17α-estradiol | 55.45 (29–103) | 79–80 | ? | ? | Metabolite |
16,17-Epiestriol |
16β-Hydroxy-17α-estradiol | 1.0 | 13 | ? | ? | Metabolite |
| 2-Hydroxyestradiol | 2-OH-E2 | 22 (7–81) | 11–35 | 2.5 | 1.3 | Metabolite |
| 2-Methoxyestradiol | 2-MeO-E2 | 0.0027–2.0 | 1.0 | ? | ? | Metabolite |
| 4-Hydroxyestradiol | 4-OH-E2 | 13 (8–70) | 7–56 | 1.0 | 1.9 | Metabolite |
| 4-Methoxyestradiol | 4-MeO-E2 | 2.0 | 1.0 | ? | ? | Metabolite |
| 2-Hydroxyestrone | 2-OH-E1 | 2.0–4.0 | 0.2–0.4 | ? | ? | Metabolite |
| 2-Methoxyestrone | 2-MeO-E1 | <0.001–<1 | <1 | ? | ? | Metabolite |
| 4-Hydroxyestrone | 4-OH-E1 | 1.0–2.0 | 1.0 | ? | ? | Metabolite |
| 4-Methoxyestrone | 4-MeO-E1 | <1 | <1 | ? | ? | Metabolite |
| 16α-Hydroxyestrone | 16α-OH-E1; 17-Ketoestriol | 2.0–6.5 | 35 | ? | ? | Metabolite |
| 2-Hydroxyestriol | 2-OH-E3 | 2.0 | 1.0 | ? | ? | Metabolite |
| 4-Methoxyestriol | 4-MeO-E3 | 1.0 | 1.0 | ? | ? | Metabolite |
| Estradiol sulfate | E2S; Estradiol 3-sulfate | <1 | <1 | ? | ? | Metabolite |
| Estradiol disulfate | Estradiol 3,17β-disulfate | 0.0004 | ? | ? | ? | Metabolite |
| Estradiol 3-glucuronide | E2-3G | 0.0079 | ? | ? | ? | Metabolite |
Estradiol 17β-glucuronide |
E2-17G | 0.0015 | ? | ? | ? | Metabolite |
| Estradiol 3-gluc. 17β-sulfate | E2-3G-17S | 0.0001 | ? | ? | ? | Metabolite |
| Estrone sulfate | E1S; Estrone 3-sulfate | <1 | <1 | >10 | >10 | Metabolite |
| Estradiol benzoate | EB; Estradiol 3-benzoate | 10 | ? | ? | ? | Estrogen |
| Estradiol 17β-benzoate | E2-17B | 11.3 | 32.6 | ? | ? | Estrogen |
| Estrone methyl ether | Estrone 3-methyl ether | 0.145 | ? | ? | ? | Estrogen |
| ent-Estradiol | 1-Estradiol | 1.31–12.34 | 9.44–80.07 | ? | ? | Estrogen |
| Equilin | 7-Dehydroestrone | 13 (4.0–28.9) | 13.0–49 | 0.79 | 0.36 | Estrogen |
| Equilenin | 6,8-Didehydroestrone | 2.0–15 | 7.0–20 | 0.64 | 0.62 | Estrogen |
| 17β-Dihydroequilin | 7-Dehydro-17β-estradiol | 7.9–113 | 7.9–108 | 0.09 | 0.17 | Estrogen |
| 17α-Dihydroequilin | 7-Dehydro-17α-estradiol | 18.6 (18–41) | 14–32 | 0.24 | 0.57 | Estrogen |
| 17β-Dihydroequilenin | 6,8-Didehydro-17β-estradiol | 35–68 | 90–100 | 0.15 | 0.20 | Estrogen |
| 17α-Dihydroequilenin | 6,8-Didehydro-17α-estradiol | 20 | 49 | 0.50 | 0.37 | Estrogen |
| Δ8-Estradiol | 8,9-Dehydro-17β-estradiol | 68 | 72 | 0.15 | 0.25 | Estrogen |
| Δ8-Estrone | 8,9-Dehydroestrone | 19 | 32 | 0.52 | 0.57 | Estrogen |
| Ethinylestradiol | EE; 17α-Ethynyl-17β-E2 | 120.9 (68.8–480) | 44.4 (2.0–144) | 0.02–0.05 | 0.29–0.81 | Estrogen |
| Mestranol | EE 3-methyl ether | ? | 2.5 | ? | ? | Estrogen |
| Moxestrol | RU-2858; 11β-Methoxy-EE | 35–43 | 5–20 | 0.5 | 2.6 | Estrogen |
| Methylestradiol | 17α-Methyl-17β-estradiol | 70 | 44 | ? | ? | Estrogen |
| Diethylstilbestrol | DES; Stilbestrol | 129.5 (89.1–468) | 219.63 (61.2–295) | 0.04 | 0.05 | Estrogen |
| Hexestrol | Dihydrodiethylstilbestrol | 153.6 (31–302) | 60–234 | 0.06 | 0.06 | Estrogen |
| Dienestrol | Dehydrostilbestrol | 37 (20.4–223) | 56–404 | 0.05 | 0.03 | Estrogen |
| Benzestrol (B2) | – | 114 | ? | ? | ? | Estrogen |
| Chlorotrianisene | TACE | 1.74 | ? | 15.30 | ? | Estrogen |
| Triphenylethylene | TPE | 0.074 | ? | ? | ? | Estrogen |
| Triphenylbromoethylene | TPBE | 2.69 | ? | ? | ? | Estrogen |
| Tamoxifen | ICI-46,474 | 3 (0.1–47) | 3.33 (0.28–6) | 3.4–9.69 | 2.5 | SERM |
| Afimoxifene | 4-Hydroxytamoxifen; 4-OHT | 100.1 (1.7–257) | 10 (0.98–339) | 2.3 (0.1–3.61) | 0.04–4.8 | SERM |
| Toremifene | 4-Chlorotamoxifen; 4-CT | ? | ? | 7.14–20.3 | 15.4 | SERM |
| Clomifene | MRL-41 | 25 (19.2–37.2) | 12 | 0.9 | 1.2 | SERM |
| Cyclofenil | F-6066; Sexovid | 151–152 | 243 | ? | ? | SERM |
| Nafoxidine | U-11,000A | 30.9–44 | 16 | 0.3 | 0.8 | SERM |
| Raloxifene | – | 41.2 (7.8–69) | 5.34 (0.54–16) | 0.188–0.52 | 20.2 | SERM |
| Arzoxifene | LY-353,381 | ? | ? | 0.179 | ? | SERM |
| Lasofoxifene | CP-336,156 | 10.2–166 | 19.0 | 0.229 | ? | SERM |
| Ormeloxifene | Centchroman | ? | ? | 0.313 | ? | SERM |
| Levormeloxifene | 6720-CDRI; NNC-460,020 | 1.55 | 1.88 | ? | ? | SERM |
| Ospemifene | Deaminohydroxytoremifene | 0.82–2.63 | 0.59–1.22 | ? | ? | SERM |
| Bazedoxifene | – | ? | ? | 0.053 | ? | SERM |
| Etacstil | GW-5638 | 4.30 | 11.5 | ? | ? | SERM |
ICI-164,384 |
– | 63.5 (3.70–97.7) | 166 | 0.2 | 0.08 | Antiestrogen |
| Fulvestrant | ICI-182,780 | 43.5 (9.4–325) | 21.65 (2.05–40.5) | 0.42 | 1.3 | Antiestrogen |
| Propylpyrazoletriol | PPT | 49 (10.0–89.1) | 0.12 | 0.40 | 92.8 | ERα agonist |
| 16α-LE2 | 16α-Lactone-17β-estradiol | 14.6–57 | 0.089 | 0.27 | 131 | ERα agonist |
| 16α-Iodo-E2 | 16α-Iodo-17β-estradiol | 30.2 | 2.30 | ? | ? | ERα agonist |
| Methylpiperidinopyrazole | MPP | 11 | 0.05 | ? | ? | ERα antagonist |
| Diarylpropionitrile | DPN | 0.12–0.25 | 6.6–18 | 32.4 | 1.7 | ERβ agonist |
| 8β-VE2 | 8β-Vinyl-17β-estradiol | 0.35 | 22.0–83 | 12.9 | 0.50 | ERβ agonist |
| Prinaberel | ERB-041; WAY-202,041 | 0.27 | 67–72 | ? | ? | ERβ agonist |
| ERB-196 | WAY-202,196 | ? | 180 | ? | ? | ERβ agonist |
| Erteberel | SERBA-1; LY-500,307 | ? | ? | 2.68 | 0.19 | ERβ agonist |
| SERBA-2 | – | ? | ? | 14.5 | 1.54 | ERβ agonist |
| Coumestrol | – | 9.225 (0.0117–94) | 64.125 (0.41–185) | 0.14–80.0 | 0.07–27.0 | Xenoestrogen |
| Genistein | – | 0.445 (0.0012–16) | 33.42 (0.86–87) | 2.6–126 | 0.3–12.8 | Xenoestrogen |
| Equol | – | 0.2–0.287 | 0.85 (0.10–2.85) | ? | ? | Xenoestrogen |
| Daidzein | – | 0.07 (0.0018–9.3) | 0.7865 (0.04–17.1) | 2.0 | 85.3 | Xenoestrogen |
| Biochanin A | – | 0.04 (0.022–0.15) | 0.6225 (0.010–1.2) | 174 | 8.9 | Xenoestrogen |
| Kaempferol | – | 0.07 (0.029–0.10) | 2.2 (0.002–3.00) | ? | ? | Xenoestrogen |
| Naringenin | – | 0.0054 (<0.001–0.01) | 0.15 (0.11–0.33) | ? | ? | Xenoestrogen |
| 8-Prenylnaringenin | 8-PN | 4.4 | ? | ? | ? | Xenoestrogen |
| Quercetin | – | <0.001–0.01 | 0.002–0.040 | ? | ? | Xenoestrogen |
| Ipriflavone | – | <0.01 | <0.01 | ? | ? | Xenoestrogen |
| Miroestrol | – | 0.39 | ? | ? | ? | Xenoestrogen |
Deoxymiroestrol |
– | 2.0 | ? | ? | ? | Xenoestrogen |
β-Sitosterol |
– | <0.001–0.0875 | <0.001–0.016 | ? | ? | Xenoestrogen |
| Resveratrol | – | <0.001–0.0032 | ? | ? | ? | Xenoestrogen |
| α-Zearalenol | – | 48 (13–52.5) | ? | ? | ? | Xenoestrogen |
| β-Zearalenol | – | 0.6 (0.032–13) | ? | ? | ? | Xenoestrogen |
| Zeranol | α-Zearalanol | 48–111 | ? | ? | ? | Xenoestrogen |
| Taleranol | β-Zearalanol | 16 (13–17.8) | 14 | 0.8 | 0.9 | Xenoestrogen |
| Zearalenone | ZEN | 7.68 (2.04–28) | 9.45 (2.43–31.5) | ? | ? | Xenoestrogen |
| Zearalanone | ZAN | 0.51 | ? | ? | ? | Xenoestrogen |
| Bisphenol A | BPA | 0.0315 (0.008–1.0) | 0.135 (0.002–4.23) | 195 | 35 | Xenoestrogen |
| Endosulfan | EDS | <0.001–<0.01 | <0.01 | ? | ? | Xenoestrogen |
Kepone |
Chlordecone | 0.0069–0.2 | ? | ? | ? | Xenoestrogen |
o,p'-DDT |
– | 0.0073–0.4 | ? | ? | ? | Xenoestrogen |
p,p'-DDT |
– | 0.03 | ? | ? | ? | Xenoestrogen |
| Methoxychlor | p,p'-Dimethoxy-DDT | 0.01 (<0.001–0.02) | 0.01–0.13 | ? | ? | Xenoestrogen |
| HPTE | Hydroxychlor; p,p'-OH-DDT | 1.2–1.7 | ? | ? | ? | Xenoestrogen |
| Testosterone | T; 4-Androstenolone | <0.0001–<0.01 | <0.002–0.040 | >5000 | >5000 | Androgen |
| Dihydrotestosterone | DHT; 5α-Androstanolone | 0.01 (<0.001–0.05) | 0.0059–0.17 | 221–>5000 | 73–1688 | Androgen |
| Nandrolone | 19-Nortestosterone; 19-NT | 0.01 | 0.23 | 765 | 53 | Androgen |
| Dehydroepiandrosterone | DHEA; Prasterone | 0.038 (<0.001–0.04) | 0.019–0.07 | 245–1053 | 163–515 | Androgen |
5-Androstenediol |
A5; Androstenediol | 6 | 17 | 3.6 | 0.9 | Androgen |
| 4-Androstenediol | – | 0.5 | 0.6 | 23 | 19 | Androgen |
4-Androstenedione |
A4; Androstenedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| 3α-Androstanediol | 3α-Adiol | 0.07 | 0.3 | 260 | 48 | Androgen |
| 3β-Androstanediol | 3β-Adiol | 3 | 7 | 6 | 2 | Androgen |
| Androstanedione | 5α-Androstanedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| Etiocholanedione | 5β-Androstanedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| Methyltestosterone | 17α-Methyltestosterone | <0.0001 | ? | ? | ? | Androgen |
Ethinyl-3α-androstanediol |
17α-Ethynyl-3α-adiol | 4.0 | <0.07 | ? | ? | Estrogen |
Ethinyl-3β-androstanediol |
17α-Ethynyl-3β-adiol | 50 | 5.6 | ? | ? | Estrogen |
| Progesterone | P4; 4-Pregnenedione | <0.001–0.6 | <0.001–0.010 | ? | ? | Progestogen |
| Norethisterone | NET; 17α-Ethynyl-19-NT | 0.085 (0.0015–<0.1) | 0.1 (0.01–0.3) | 152 | 1084 | Progestogen |
Norethynodrel |
5(10)-Norethisterone | 0.5 (0.3–0.7) | <0.1–0.22 | 14 | 53 | Progestogen |
| Tibolone | 7α-Methylnorethynodrel | 0.5 (0.45–2.0) | 0.2–0.076 | ? | ? | Progestogen |
| Δ4-Tibolone | 7α-Methylnorethisterone | 0.069–<0.1 | 0.027–<0.1 | ? | ? | Progestogen |
| 3α-Hydroxytibolone | – | 2.5 (1.06–5.0) | 0.6–0.8 | ? | ? | Progestogen |
| 3β-Hydroxytibolone | – | 1.6 (0.75–1.9) | 0.070–0.1 | ? | ? | Progestogen |
| Footnotes: a = (1) ERβ proteins (except the ERβ values from Kuiper et al. (1997), which are rat ERβ). Sources: See template page.
| ||||||
| Estrogen | Relative binding affinities (%)
| ||||||
|---|---|---|---|---|---|---|---|
| ER | AR | PR | GR | MR | SHBG | CBG
| |
| Estradiol | 100 | 7.9 | 2.6 | 0.6 | 0.13 | 8.7–12 | <0.1 |
| Estradiol benzoate | ? | ? | ? | ? | ? | <0.1–0.16 | <0.1 |
| Estradiol valerate | 2 | ? | ? | ? | ? | ? | ? |
| Estrone | 11–35 | <1 | <1 | <1 | <1 | 2.7 | <0.1 |
| Estrone sulfate | 2 | 2 | ? | ? | ? | ? | ? |
| Estriol | 10–15 | <1 | <1 | <1 | <1 | <0.1 | <0.1 |
| Equilin | 40 | ? | ? | ? | ? | ? | 0 |
| Alfatradiol | 15 | <1 | <1 | <1 | <1 | ? | ? |
| Epiestriol | 20 | <1 | <1 | <1 | <1 | ? | ? |
| Ethinylestradiol | 100–112 | 1–3 | 15–25 | 1–3 | <1 | 0.18 | <0.1 |
| Mestranol | 1 | ? | ? | ? | ? | <0.1 | <0.1 |
| Methylestradiol | 67 | 1–3 | 3–25 | 1–3 | <1 | ? | ? |
| Moxestrol | 12 | <0.1 | 0.8 | 3.2 | <0.1 | <0.2 | <0.1 |
| Diethylstilbestrol | ? | ? | ? | ? | ? | <0.1 | <0.1 |
| Notes: Reference CBG . Sources: See template.
| |||||||
| Estrogen | RBA (%) |
Uterine weight (%) | Uterotrophy |
LH levels (%) | RBA (%)
|
|---|---|---|---|---|---|
| Control | – | 100 | – | 100 | – |
| Estradiol (E2) | 100 | 506 ± 20 | +++ | 12–19 | 100 |
| Estrone (E1) | 11 ± 8 | 490 ± 22 | +++ | ? | 20 |
| Estriol (E3) | 10 ± 4 | 468 ± 30 | +++ | 8–18 | 3 |
| Estetrol (E4) | 0.5 ± 0.2 | ? | Inactive | ? | 1 |
| 17α-Estradiol | 4.2 ± 0.8 | ? | ? | ? | ? |
| 2-Hydroxyestradiol | 24 ± 7 | 285 ± 8 | +b | 31–61 | 28 |
| 2-Methoxyestradiol | 0.05 ± 0.04 | 101 | Inactive | ? | 130 |
| 4-Hydroxyestradiol | 45 ± 12 | ? | ? | ? | ? |
| 4-Methoxyestradiol | 1.3 ± 0.2 | 260 | ++ | ? | 9 |
| 4-Fluoroestradiola | 180 ± 43 | ? | +++ | ? | ? |
| 2-Hydroxyestrone | 1.9 ± 0.8 | 130 ± 9 | Inactive | 110–142 | 8 |
| 2-Methoxyestrone | 0.01 ± 0.00 | 103 ± 7 | Inactive | 95–100 | 120 |
| 4-Hydroxyestrone | 11 ± 4 | 351 | ++ | 21–50 | 35 |
| 4-Methoxyestrone | 0.13 ± 0.04 | 338 | ++ | 65–92 | 12 |
| 16α-Hydroxyestrone | 2.8 ± 1.0 | 552 ± 42 | +++ | 7–24 | <0.5 |
| 2-Hydroxyestriol | 0.9 ± 0.3 | 302 | +b | ? | ? |
| 2-Methoxyestriol | 0.01 ± 0.00 | ? | Inactive | ? | 4 |
| Notes: Values are mean ± SD or range. ER RBA = endogenous ). b = Atypical uterotrophic effect which plateaus within 48 hours (estradiol's uterotrophy continues linearly up to 72 hours). Sources: See template.
| |||||
| Estrogen | HF |
VE | UCa | FSH | LH | HDL-C | SHBG | CBG |
AGT |
Liver |
|---|---|---|---|---|---|---|---|---|---|---|
| Estradiol | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Estrone | ? | ? | ? | 0.3 | 0.3 | ? | ? | ? | ? | ? |
| Estriol | 0.3 | 0.3 | 0.1 | 0.3 | 0.3 | 0.2 | ? | ? | ? | 0.67 |
| Estrone sulfate | ? | 0.9 | 0.9 | 0.8–0.9 | 0.9 | 0.5 | 0.9 | 0.5–0.7 | 1.4–1.5 | 0.56–1.7 |
| Conjugated estrogens | 1.2 | 1.5 | 2.0 | 1.1–1.3 | 1.0 | 1.5 | 3.0–3.2 | 1.3–1.5 | 5.0 | 1.3–4.5 |
Equilin sulfate |
? | ? | 1.0 | ? | ? | 6.0 | 7.5 | 6.0 | 7.5 | ? |
| Ethinylestradiol | 120 | 150 | 400 | 60–150 | 100 | 400 | 500–600 | 500–600 | 350 | 2.9–5.0 |
| Diethylstilbestrol | ? | ? | ? | 2.9–3.4 | ? | ? | 26–28 | 25–37 | 20 | 5.7–7.5 |
Sources and footnotes
Notes: Values are ratios, with estradiol as standard (i.e., 1.0). Abbreviations: HF = Clinical relief of liver proteins. Liver = Ratio of liver estrogenic effects to general/systemic estrogenic effects (hot flashes/gonadotropins ). Sources: See template. | ||||||||||
| Compound | Dosage for specific uses (mg usually)[a] | ||||||
|---|---|---|---|---|---|---|---|
| ETD[b] | EPD[b] | MSD[b] | MSD[c] | OID[c] | TSD[c] | ||
| Estradiol (non-micronized) | 30 | ≥120–300 | 120 | 6 | - | - | |
| Estradiol (micronized) | 6–12 | 60–80 | 14–42 | 1–2 | >5 | >8 | |
| Estradiol valerate | 6–12 | 60–80 | 14–42 | 1–2 | - | >8 | |
| Estradiol benzoate | - | 60–140 | - | - | - | - | |
| Estriol | ≥20 | 120–150[d] | 28–126 | 1–6 | >5 | - | |
| Estriol succinate | - | 140–150[d] | 28–126 | 2–6 | - | - | |
| Estrone sulfate | 12 | 60 | 42 | 2 | - | - | |
| Conjugated estrogens | 5–12 | 60–80 | 8.4–25 | 0.625–1.25 | >3.75 | 7.5 | |
| Ethinylestradiol | 200 μg | 1–2 | 280 μg | 20–40 μg | 100 μg | 100 μg | |
| Mestranol | 300 μg | 1.5–3.0 | 300–600 μg | 25–30 μg | >80 μg | - | |
| Quinestrol | 300 μg | 2–4 | 500 μg | 25–50 μg | - | - | |
| Methylestradiol | - | 2 | - | - | - | - | |
| Diethylstilbestrol | 2.5 | 20–30 | 11 | 0.5–2.0 | >5 | 3 | |
| DES dipropionate | - | 15–30 | - | - | - | - | |
| Dienestrol | 5 | 30–40 | 42 | 0.5–4.0 | - | - | |
| Dienestrol diacetate | 3–5 | 30–60 | - | - | - | - | |
| Hexestrol | - | 70–110 | - | - | - | - | |
| Chlorotrianisene | - | >100 | - | - | >48 | - | |
| Methallenestril | - | 400 | - | - | - | - | |
| Estrogen | Form | Dose (mg) | Duration by dose (mg) | ||
|---|---|---|---|---|---|
| EPD | CICD | ||||
| Estradiol | Aq. soln. | ? | – | <1 d | |
| Oil soln. | 40–60 | – | 1–2 ≈ 1–2 d | ||
| Aq. susp. | ? | 3.5 | 0.5–2 ≈ 2–7 d; 3.5 ≈ >5 d | ||
| Microsph. | ? | – | 1 ≈ 30 d | ||
| Estradiol benzoate | Oil soln. | 25–35 | – | 1.66 ≈ 2–3 d; 5 ≈ 3–6 d | |
| Aq. susp. | 20 | – | 10 ≈ 16–21 d | ||
| Emulsion | ? | – | 10 ≈ 14–21 d | ||
| Estradiol dipropionate | Oil soln. | 25–30 | – | 5 ≈ 5–8 d | |
| Estradiol valerate | Oil soln. | 20–30 | 5 | 5 ≈ 7–8 d; 10 ≈ 10–14 d; 40 ≈ 14–21 d; 100 ≈ 21–28 d | |
| Estradiol benz. butyrate | Oil soln. | ? | 10 | 10 ≈ 21 d | |
| Estradiol cypionate | Oil soln. | 20–30 | – | 5 ≈ 11–14 d | |
| Aq. susp. | ? | 5 | 5 ≈ 14–24 d | ||
Estradiol enanthate |
Oil soln. | ? | 5–10 | 10 ≈ 20–30 d | |
Estradiol dienanthate |
Oil soln. | ? | – | 7.5 ≈ >40 d | |
| Estradiol undecylate | Oil soln. | ? | – | 10–20 ≈ 40–60 d; 25–50 ≈ 60–120 d | |
| Polyestradiol phosphate | Aq. soln. | 40–60 | – | 40 ≈ 30 d; 80 ≈ 60 d; 160 ≈ 120 d | |
| Estrone | Oil soln. | ? | – | 1–2 ≈ 2–3 d | |
| Aq. susp. | ? | – | 0.1–2 ≈ 2–7 d | ||
| Estriol | Oil soln. | ? | – | 1–2 ≈ 1–4 d | |
| Polyestriol phosphate | Aq. soln. | ? | – | 50 ≈ 30 d; 80 ≈ 60 d | |
Notes and sources
Notes: All ovulation-inhibiting dose of estradiol undecylate is 20–30 mg/month. Sources: See template. | |||||
| Class | Examples | RE complex retention | Pharmacodynamic profile | Uterine effects |
|---|---|---|---|---|
| Short-acting (a.k.a. "weak" or "impeded") |
Estriol • 16-Epiestriol • 17α-Estradiol • ent-Estradiol • 16-Ketoestradiol • Dimethylstilbestrol • meso-Butestrol
|
Short (1–4 hours) | Single or once-daily injections: partial agonist or antagonist | Early responsesa |
| Implant or multiple injections per day: full agonist
|
Early and late responsesb | |||
| Long-acting | A. Estradiol • Estrone • Ethinylestradiol • Diethylstilbestrol • Hexestrol | Intermediate (6–24 hours) | Single or once-daily injections: full agonist | Early and late responses |
| B. Clomifene • Nafoxidine • Nitromifene • Tamoxifen | Long (>24–48 hours) | Single injection: agonist Repeated injections: antagonist |
Early and late responses | |
| Footnotes: a = Early responses occur after 0–6 hours and include hyperemia, amino acid and nucleotide uptake, activation of RNA polymerases I and II, and stimulation of induced protein, among others. b = Late responses occur after 6–48 hours and include cellular hypertrophy and hyperplasia and sustained RNA polymerase I and II activity, among others. Sources: [80][81][82][83][84][85][86]
| ||||
Pharmacokinetics

Absorption
Estriol has significant
The bioavailability of estriol is markedly increased with
The absorption of estrogens by the skin is described as low for estriol, moderate for estradiol, and high for estrone.
Rectal administration of estriol has been assessed in one study.[87] Administration of a rectal suppository containing 100 mg estriol resulted in estriol levels in pregnant women at term increasing by about 53%.[87] Estriol levels at term are normally between 5,000 and 20,000 pg/mL, which suggests that estriol levels may have increased following the suppository by about 5,000 to 10,000 pg/mL (precise levels were not provided).[88][89][90]
Distribution
Similarly to estradiol, but unlike
Metabolism
Estriol is
The
A single 1 to 2 mg dose of estriol in oil solution by intramuscular injection has a duration of about 3 or 4 days.
Excretion
Estriol is
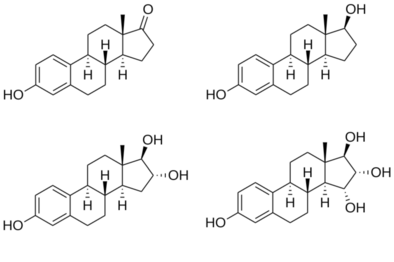
Chemistry
Structures of major endogenous estrogens
hydroxyl (–OH) groups : estrone (E1) has one, estradiol (E2) has two, estriol (E3) has three, and estetrol (E4) has four. |
Estriol, also known as 16α-hydroxyestradiol or as estra-1,3,5(10)-triene-3,16α,17β-triol, is a
Analogues
A variety of
History
Estriol was discovered in 1930.
Society and culture
Generic names
Estriol is the
Brand names
Estriol is or has been marketed under a variety of brand names throughout the world, including Aacifemine, Colpogyn, Elinol, Estriel, Estriol, Estriosalbe, Estrokad, Evalon, Gydrelle, Gynäsan, Gynest, Gynoflor (in combination with
Estriol for multiple sclerosis had the tentative brand name Trimesta but did not complete development and was never marketed.[117]
Availability
Estriol is marketed widely throughout the world, including in
Research
Estriol may have
Veterinary use
Estriol is used in
References
- ^ a b c Estriol. Dictionary.com.
- ^ ISBN 978-3-88763-075-1.
- ^ a b c d e f g h i j k l "Estriol". Archived from the original on 2018-07-05. Retrieved 2018-05-20.
- ^ S2CID 24616324.
- ^ ISBN 978-3-642-60107-1.
- ^ S2CID 27098486.
- ^ PMID 4203562.
- ^ ISBN 978-3-527-64565-7.
- ^ ISBN 978-0-683-08931-8.
Estriol is considered a short-acting estrogen with a half-life of 5 hours.
- ^ S2CID 23568599.
- ^ ISBN 978-0-397-50768-9.
Because its half-life is about 20 minutes, unconjugated estriol rapidly reflects changes in estriol production.
- ^ S2CID 407950.
- ^ ISBN 978-81-8061-427-9.
- ^ ISBN 978-0-323-31239-4.
- ^ ISBN 978-1-4377-2357-1.
- ^ ISBN 978-1-4377-1152-3.
- ^ ISBN 978-0-323-24485-5.
- ^ S2CID 41137736.
- ^ ISBN 978-0-7817-1750-2.
- ^ PMID 6356176.
- ^ PMID 6202959.
- ^ ISBN 978-1-4613-2157-6.
- ^ ISBN 978-3-642-65806-8.
- ^ PMID 13567043.
- ^ PMID 18744783.
- ^ PMID 7493689.
- ^ "Estriol/Lactobacillus - Acerus Pharmaceuticals/Medinova -". AdisInsight. Springer Nature Switzerland AG.
- ^ PMID 17627398.
- ^ ISBN 978-1-4757-2085-3.
- ^ Mazer C, Israel SL, Charny CW (1946). Diagnosis and treatment of menstrual disorders and sterility. Hoeber. p. 525.
8. PREPARATIONS OF ESTRIOL. Estriol is the least active of all commercially available natural estrogenic substances. A milligram of estriol yields approximately 350 Allen-Doisy rat units. Estriol (Abbott). Capsules containing 0.06, 0.12, and 0.24 mg. Estriol (Lilly). Puvules containing 0.06, 0.12, and 0.24 mg. Theelol (Parke-Davis). Capsules containing 0.06, 0.12, and 0.24 mg.
- ISBN 978-0-598-66833-2.
Estriol. Estriol (theelol) is trihydroxyestrin. It is a crystalline estrogenic steroid obtained from the urine of pregnant women. It is less actively estrogenic than estrone. Several pharmaceutical houses supply capsules containing 0.06 or 0.12 mg. These may be obtained as Theelol (Parke-Davis), Estriol (Abbott), and Estriol (Lilly).
- ^ Sollmann TH (1948). A manual of pharmacology and its applications to therapeutics and toxicology. W. B. Saunders.
Estriol (Theelol), N.N.R.; characters and solubility as for estrone; considerably less potent. Marketed as capsules of 0.06, 0.12 and 0.24 mg. Dose, 0.06 to 0.12 mg. once to four times daily.
- ^ Council on Drugs (American Medical Association) (1950). New and Nonofficial Drugs. Lippincott. p. 322.
Abbott Laboratories Capsules Estriol: 0.12 mg. and 0.24 mg. Eli Lilly and Company Pulvules Estriol: 0.06 mg., 0.12 mg. and 0.24 mg. Parke, Davis & Company Kapseals Theelol: 0.24 mg.
- ^ New York State Journal of Medicine. Medical Society of the State of New York. 1939. p. 1760.
- OCLC 35483492.
- ^ PMID 17549577.
- PMID 6250009.
- PMID 1186230.
- ISBN 978-1-4165-1085-7.
- ISBN 978-0-323-02015-2.
- ^ ISBN 978-3-527-33527-5.
- ^ PMID 16554039.
- PMID 26023144.
- ^ S2CID 24525995.
- ^ PMID 25496649.
- ISBN 978-0-12-060308-4.
- PMID 359134.
- PMID 3695484.
- PMID 7421203.
- PMID 7195404.
- ^ ISBN 978-3-642-96158-8.
The polymer of estradiol or estriol and phosphoric acid has an excellent depot action when given intramuscularly (polyestriol phosphate or polyestradiol phosphate) (Table 16). Phosphoric acid combines with the estrogen molecule at C3 and C17 to form a macromolecule. The compound is stored in the liver and spleen where the estrogen is steadily released by splitting off of the phosphate portion due to the action of alkaline phosphatase. [...] Conjugated estrogens and polyestriol and estradiol phosphate can also be given intravenously in an aqueous solution. Intravenous administration of ovarian hormones offers no advantages, however, and therefore has no practical significance. [...] The following duarations of action have been obtained with a single administration (WlED, 1954; LAURITZEN, 1968): [...] 50 mg polyestradiol phosphate ~ 1 month; 50 mg polyestriol phosphate ~ 1 month; 80 mg polyestriol phosphate ~ 2 months.
- ^ PMID 2170822.
- ISSN 0031-9333.
- ISBN 978-0-397-59010-0.
- ISBN 978-3-642-49506-9.
- ^ PMID 390365.
- ^ PMID 830547.
- ISBN 978-3-662-07635-4.
- ISSN 0172-777X.
- PMID 6429481.
- PMID 2215269.
- PMID 559617.
- ISBN 978-3-642-75101-1.
- ISBN 978-3-11-024568-4.
- ISBN 978-3-662-00942-0.
- ISBN 978-94-009-8195-9.
- ISBN 978-3-11-150424-7.
- PMID 779393.
- ISSN 0001-6349.
- ISSN 0172-777X.
- PMID 29603164.
- PMID 29756046.
- PMID 15432047.
- PMID 14902290.
- ISSN 0001-6349.
There is no doubt that the conversion of the endometrium with injections of both synthetic and native estrogenic hormone preparations succeeds, but the opinion whether native, orally administered preparations can produce a proliferation mucosa changes with different authors. PEDERSEN-BJERGAARD (1939) was able to show that 90% of the folliculin taken up in the blood of the vena portae is inactivated in the liver. Neither KAUFMANN (1933, 1935), RAUSCHER (1939, 1942) nor HERRNBERGER (1941) succeeded in bringing a castration endometrium into proliferation using large doses of orally administered preparations of estrone or estradiol. Other results are reported by NEUSTAEDTER (1939), LAUTERWEIN (1940) and FERIN (1941); they succeeded in converting an atrophic castration endometrium into an unambiguous proliferation mucosa with 120–300 oestradiol or with 380 oestrone.
- ISBN 978-3-642-57636-2.
- ^ Martinez-Manautou J, Rudel HW (1966). "Antiovulatory Activity of Several Synthetic and Natural Estrogens". In Robert Benjamin Greenblatt (ed.). Ovulation: Stimulation, Suppression, and Detection. Lippincott. pp. 243–253.
- ISBN 978-3-642-49506-9.
- PMID 13370006.
- PMID 830547.
- PMID 400777.
- S2CID 9628572.
- ISBN 978-3-662-07635-4.
- PMID 6356176.
- PMID 6202959.
- PMID 4347201.
- ^ PMID 8288717.
- ISBN 978-0-08-053413-8.
- ISBN 978-1-4398-1469-7.
- ISBN 978-1-4377-2135-5.
- ^ PMID 6847874.
- ISBN 978-1-4612-5525-3.
- ^ ISBN 978-0-12-800627-6.
- PMID 4366454.
- PMID 3227988.
- S2CID 35881837.
- PMID 13491750.
- ^ PMID 13759555.
- ISBN 978-0-12-803717-1.
- ^ PMID 5107471.
- ISBN 978-94-011-6165-7.
In the Federal Republic of Germany between 10 and 20% of all climacteric women are on estrogen treatment. We have the following oral estrogens for a treatment. (t) Conjugated estrogens, (2) estradiol valerate, (3) ethinyl-estradiol and its cyclopentyl-enol ether, (4) stilbestrol, (5) ethinyl-estradiol-methyltestosterone, (6) estriol and estriol succinate, most of them as coated tablets. Several long acting injectable preparations are available: several esters of combined estradiol-testosterone, one of estradiol-dehydroepiandrosterone enanthate and a prolonged polyestriol phosphate are also available. Lastly, depot injections of estradiol- and stilbestrol-esters are on the market.
- ^ S2CID 24003341.
- ^ S2CID 32081001.
- ^ "Estetrol - Mithra Pharmaceuticals/Pantarhei Bioscience". AdisInsight. Springer Nature Switzerland AG.
- ^ "Drospirenone/Estetrol - Mithra Pharmaceuticals". AdisInsight. Springer Nature Switzerland AG.
- ^ ISSN 0025-7125.
- PMID 29648134.
- ^ PMID 18746057.
- ^ ISSN 0002-9955.
- PMID 29646930.
- ^ ISBN 978-1-60917-326-5.
- ^ ISBN 978-1-4832-1945-5.
- PMID 13359169.
- ^ ISBN 978-94-011-4439-1.
- ISBN 978-3-527-30247-5.
8075-01 (6628-01) 37452-43-0 R Polymeric ester with phosphoric acid S Klimadurin, Polyestriol phosphate, Polyostriolphosphat, Triodurin U Depot-estrogen
- ISBN 978-0-85369-300-0.
Polyoestriol Phosphate. [...] ingredient of Klimadurin. [...] Triodurin [...].
- ^ a b c "Estriol succinate - Synthetic Biologics". AdisInsight. Springer Nature Switzerland AG.
- ISBN 978-3-642-68976-5.
- PMID 21531972.
- ISBN 0-7216-9315-6.
Further reading
- Merrill RC (1958). "Estriol: a review". Physiol. Rev. 38 (3): 463–80. PMID 13567043.
- Clark JH, Hardin JW, McCormack SA (1979). "Mechanism of action of estrogen agonists and antagonists". J. Anim. Sci. 49 (Suppl 2): 46–65. PMID 400777.
- Clark JH, Markaverich BM (1983). "The agonistic and antagonistic effects of short acting estrogens: a review". Pharmacol. Ther. 21 (3): 429–53. PMID 6356176.
- Clark JH, Markaverich BM (April 1984). "The agonistic and antagonistic actions of estriol". J. Steroid Biochem. 20 (4B): 1005–13. PMID 6202959.
- Heimer GM (1987). "Estriol in the postmenopause". Acta Obstet Gynecol Scand Suppl. 139: 2–23. S2CID 38712328.
- Esposito G (June 1991). "Estriol: a weak estrogen or a different hormone?". Gynecol. Endocrinol. 5 (2): 131–53. PMID 1927578.
- Vooijs GP, Geurts TB (September 1995). "Review of the endometrial safety during intravaginal treatment with estriol". Eur. J. Obstet. Gynecol. Reprod. Biol. 62 (1): 101–6. PMID 7493689.
- Head KA (1998). "Estriol: safety and efficacy" (PDF). Altern Med Rev. 3 (2): 101–13. PMID 9577246.
- Taylor M (December 2001). "Unconventional estrogens: estriol, biest, and triest". Clin Obstet Gynecol. 44 (4): 864–79. S2CID 27098486.
- Ciszko B, Zdrojewicz Z (July 2006). "Znaczenie endogennego i egzogennego estriolu w praktyce klinicznej" [Compliance endogenous and exogenous estriol in clinical practice]. Ginekol. Pol. (in Polish). 77 (7): 559–65. PMID 17076208.
- Perepanova TS, Khazan PL (2007). "[The role of estriol in therapy of urogenital disorders in postmenopausal women]". Urologiia (in Russian) (3): 102–104, 107. PMID 17722629.
- Holtorf K (January 2009). "The bioidentical hormone debate: are bioidentical hormones (estradiol, estriol, and progesterone) safer or more efficacious than commonly used synthetic versions in hormone replacement therapy?". Postgrad Med. 121 (1): 73–85. S2CID 2060730.
- Unlü C, Donders G (2011). "Use of lactobacilli and estriol combination in the treatment of disturbed vaginal ecosystem: a review". J Turk Ger Gynecol Assoc. 12 (4): 239–46. PMID 24592002.
- Ali ES, Mangold C, Peiris AN (September 2017). "Estriol: emerging clinical benefits". Menopause. 24 (9): 1081–1085. S2CID 41137736.
- Rueda C, Osorio AM, Avellaneda AC, Pinzón CE, Restrepo OI (August 2017). "The efficacy and safety of estriol to treat vulvovaginal atrophy in postmenopausal women: a systematic literature review". Climacteric. 20 (4): 321–330. S2CID 407950.
- Mueck AO, Ruan X, Prasauskas V, Grob P, Ortmann O (April 2018). "Treatment of vaginal atrophy with estriol and lactobacilli combination: a clinical review". Climacteric. 21 (2): 140–147. PMID 29381086.
- La Rosa VL, Ciebiera M, Lin LT, Fan S, Butticè S, Sathyapalan T, et al. (June 2019). "Treatment of genitourinary syndrome of menopause: the potential effects of intravaginal ultralow-concentration oestriol and intravaginal dehydroepiandrosterone on quality of life and sexual function". Prz Menopauzalny. 18 (2): 116–122. PMID 31488961.