Persistent carbene

A persistent carbene (also known as stable carbene) is an
Modern theoretical analysis suggests that the term "persistent carbene" is in fact a
Persistent carbenes in general, and Arduengo carbenes in particular, are popular ligands in organometallic chemistry.
History
Early evidence
In 1957,
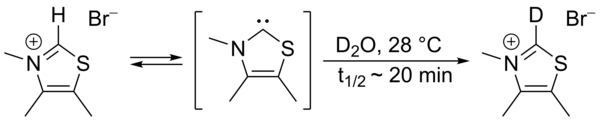
This exchange was proposed to proceed via intermediacy of a thiazol-2-ylidene. In 2012 the isolation of the so-called Breslow intermediate was reported.[5][6]
In 1960,
Isolation of persistent carbenes
In 1970, Wanzlick's group generated imidazol-2-ylidene carbenes by the deprotonation of an
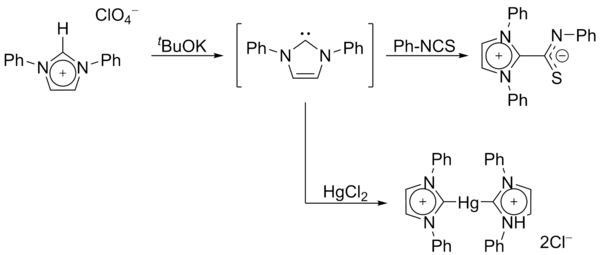
In 1988,

These compounds were called "push-pull carbenes" in reference to the contrasting electron affinities of the phosphorus and silicon atoms. They exhibit both carbenic and alkynic reactivity. An X-ray structure of this molecule has not been obtained and at the time of publication some doubt remained as to their exact carbenic nature.
In 1991, Arduengo and coworkers crystallized a diaminocarbene by deprotonation of an imidazolium cation:[18]

This carbene, the forerunner of a large family of carbenes with the imidazol-2-ylidene core, is indefinitely stable at room temperature in the absence of oxygen and moisture. It melts at 240–241 °C without decomposition. The 13C NMR spectrum shows a signal at 211 ppm for the carbenic atom.
The first air-stable ylidic carbene, a chlorinated member of the imidazol-2-ylidene family, was obtained in 1997.[21]
In 2000, Bertrand obtained additional carbenes of the phosphanyl type, including (phosphanyl)(trifluoromethyl)carbene, stable in solution at -30 °C[22] and a moderately stable (amino)(aryl)carbene with only one heteroatom adjacent to the carbenic atom.[23][24]
Stabilization through adjacent orbitals
In the modern understanding, the superficially unoccupied p-orbital on a (meta)stable carbene is not, in fact, fully empty. Instead, the carbene Lewis structures are in

Early workers attributed the stability of Arduengo carbenes to the bulky N-

In 1995, Arduengo's group obtained a carbene derivative of
Unlike the cyclic derivatives, acyclic carbenes are flexible and bonds to the carbenic atom admit rotation. But bond rotation in the compound appeared

Classes of stable carbenes
The following are examples of the classes of stable carbenes isolated to date:
Imidazol-2-ylidenes
The first stable carbenes to be isolated were based on an
A considerable range of imidazol-2-ylidenes have been synthesised, including those in which the 1,3-positions have been functionalised with


(View the 3D structure with external viewer.)
In particular, substitution of two chlorine atoms for the two hydrogens at ring positions 4 and 5 yielded the first air-stable carbene.[21] Its extra stability probably results from the electron-withdrawing effect of the chlorine substituents, which reduce the electron density on the carbon atom bearing the lone pair, via induction through the sigma-backbone.
Molecules containing two and even three imidazol-2-ylidene groups have also been synthesised.[37][38]
Imidazole-based carbenes are thermodynamically stable and generally have diagnostic 13C NMR chemical shift values between 210 and 230 ppm for the carbenic carbon. Typically, X-ray structures of these molecules show N–C–N bond angles of 101–102°.[citation needed]
Triazol-5-ylidenes
Depending on the arrangement of the three nitrogen atoms in triazol-5-ylidene, there are two possible isomers, namely 1,2,3-triazol-5-ylidenes and 1,2,4-triazol-5-ylidenes.

The

Triazole-based carbenes are thermodynamically stable and have diagnostic 13C NMR chemical shift values between 210 and 220 ppm for the carbenic carbon. The X-ray structure of the triphenyl substituted carbene above shows an N–C–N bond angle of around 101°. The 5-methoxytriazole precursor to this carbene was made by the treatment of a triazolium salt with sodium methoxide, which attacks as a nucleophile.[39] This may indicate that these carbenes are less aromatic than imidazol-2-ylidenes, as the imidazolium precursors do not react with nucleophiles due to the resultant loss of aromaticity.[citation needed]
Other diaminocarbenes
The two families above can be seen as special cases of a broader class of compounds which have a carbenic atom bridging two nitrogen atoms. A range of such diaminocarbenes have been prepared principally by Roger Alder's research group. In some of these compounds, the N–C–N unit is a member of a five- or six-membered non-aromatic ring,[27][29][40] including a bicyclic example. In other examples, the adjacent nitrogens are connected only through the carbenic atom, and may or may not be part of separate rings.[28][31][32]

Unlike the aromatic imidazol-2-ylidenes or triazol-5-ylidenes, these carbenes appear not to be thermodynamically stable, as shown by the dimerisation of some unhindered cyclic and acyclic examples.[29][31] Studies[30] suggest that these carbenes dimerise via acid catalysed dimerisation (as in the Wanzlick equilibrium).
Diaminocarbenes have diagnostic 13C NMR chemical shift values between 230 and 270 ppm for the carbenic atom. The X-ray structure of dihydroimidazole-2-ylidene shows a N–C–N bond angle of about 106°, whilst the angle of the acyclic carbene is 121°, both greater than those seen for imidazol-2-ylidenes.
Heteroamino carbenes
There exist several variants of the stable carbenes above where one of the nitrogen atoms adjacent to the carbene center (the α nitrogens) has been replaced by an alternative heteroatom, such as oxygen, sulfur, or phosphorus.[16][17][33][34]

In particular, the formal substitution of sulfur for one of the nitrogens in imidazole would yield the aromatic heterocyclic compound thiazole. A thiazole based carbene (analogous to the carbene postulated by Breslow)[41] has been prepared and characterised by X-ray crystallography.[33] Other non-aromatic aminocarbenes with O, S and P atoms adjacent (i.e. alpha) to the carbene centre have been prepared, for example, thio- and oxyiminium based carbenes have been characterised by X-ray crystallography.[34]
Since
Carbenes that formally derive from imidazole-2-ylidenes by substitution of sulfur, oxygen, or other chalcogens for both α-nitrogens are expected to be unstable, as they have the potential to dissociate into an alkyne (R1C≡CR2) and a carbon dichalcogenide (X1=C=X2).[42][43]
Non-amino carbenes
The reaction of
Bertrand's carbenes
In Bertrand's persistent carbenes, the unsaturated carbon is bonded to a phosphorus and a silicon.[44] However, these compounds seem to exhibit some alkynic properties, and when published the exact carbenic nature of these red oils was in debate.[17]
Other nucleophilic carbenes
One stable N-heterocyclic carbene[45] has a structure analogous to borazine with one boron atom replaced by a methylene group. This results in a planar six-electron compound.

Cyclopropenylidenes
Another family of carbenes is based on a cyclopropenylidene core, a three-carbon ring with a double bond between the two atoms adjacent to the carbenic one. This family is exemplified by bis(diisopropylamino)cyclopropenylidene.[35]
Triplet state carbenes
Persistent carbenes tend to exist in the singlet, dimerizing when forced into triplet states. Nevertheless, Hideo Tomioka and associates used electron delocalization to produce a comparatively stable triplet carbene (bis(9-anthryl)carbene) in 2001. It has an unusually long half-life of 19 minutes.[46][47]
Although the figure below shows the two parts of the molecule in one flat plane, molecular geometry puts the two aromatic parts in

In 2006 a triplet carbene was reported by the same group with a
Again the figure below is not an adequate representation of the actual molecular structure: both

Exposure to oxygen (a triplet diradical) converts this carbene to the corresponding
Mesoionic carbenes
Mesoionic carbenes (MICs) are similar to N-heterocyclic carbenes (NHCs) except that canonical resonance structures with the carbene depicted cannot be drawn without adding additional charges. Mesoionic carbenes are also referred to as abnormal N-heterocyclic carbenes (aNHC) or remote N-heterocyclic carbenes (rNHC). A variety of free carbenes can be isolated and are stable at room temperature. Other free carbenes are not stable and are susceptible to intermolecular decomposition pathways.[citation needed]
Chemical properties
Basicity and nucleophilicity
The imidazol-2-ylidenes are strong bases, having

However, further work showed that diaminocarbenes will deprotonate the DMSO solvent, with the resulting anion reacting with the resulting amidinium salt.
Reaction of imidazol-2-ylidenes with
pKa values for the conjugate acids of several NHC families have been examined in aqueous solution. pKa values of triazolium ions lie in the range 16.5–17.8,[50] around 3 pKa units more acidic than related imidazolium ions.[51]
Dimerisation
At one time, stable carbenes were thought to reversibly
Chen and Taton

If this dimer existed as a dicarbene, the electron
On the other hand, heteroamino carbenes (such as R2N–C–OR or R2N–C–SR) and non-aromatic carbenes such as diaminocarbenes (such as R2N–C–NR2) have been shown to dimerise,[53] albeit quite slowly. This has been presumed to be due to the high barrier to singlet state dimerisation:

Diaminocarbenes do not truly dimerise, but rather form the dimer by reaction via
Unlike the dimerisation of
Reactivity
The chemistry of stable carbenes has not been fully explored. However, Enders et al.[39][54][55] have performed a range of organic reactions involving a triazol-5-ylidene. These reactions are outlined below and may be considered as a model for other carbenes.

| a | 3,6-diphenyl-1,2,4,5-tetrazine, toluene | 92% | e | 2 equiv., PhNCO, toluene, reflux | 92% | |
|---|---|---|---|---|---|---|
| b | RXH, RT | 95–97% | f | CS2, toluene, or PhNCS, THF, RT | 71–90% | |
| c | O2, S8, or Se, toluene, reflux | 54–68% | g | Maleimide, THF, RT | 47–84% | |
| d | R1CH=CHR2, THF, RT | 25–68% | h | Dimethylacetylene dicarboxylate, THF, reflux | 21% |
These carbenes tend to behave in a
The reported stable isothiazole carbene (2b) derived from an isothiazolium perchlorate (1)[56] was questioned.[57] The researchers were only able to isolate 2-imino-2H-thiete (4). The intermediate 3 was proposed through a rearrangement reaction. The carbene 2b is no longer considered as stable.[58]

Carbene complexation
Imidazol-2-ylidenes, triazol-5-ylidenes (and less so, diaminocarbenes) have been shown to coordinate to a plethora of elements, from
Group →
|
1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ↓ Period
|
||||||||||||||||||||
| 1 | 1 H |
2 He | ||||||||||||||||||
| 2 | 3 Li |
4 Be |
5 B |
6 C |
7 N |
8 O |
9 F |
10 Ne | ||||||||||||
| 3 | 11 Na |
12 Mg |
13 Al |
14 Si |
15 P |
16 S |
17 Cl |
18 Ar | ||||||||||||
| 4 | 19 K |
20 Ca |
21 Sc |
22 Ti |
23 V |
24 Cr |
25 Mn |
26 Fe |
27 Co |
28 Ni |
29 Cu |
30 Zn |
31 Ga |
32 Ge |
33 As |
34 Se |
35 Br |
36 Kr | ||
| 5 | 37 Rb |
38 Sr |
39 Y |
40 Zr |
41 Nb |
42 Mo |
43 Tc |
44 Ru |
45 Rh |
46 Pd |
47 Ag |
48 Cd |
49 In |
50 Sn |
51 Sb |
52 Te |
53 I |
54 Xe | ||
| 6 | 55 Cs |
56 Ba |
71 Lu |
72 Hf |
73 Ta |
74 W |
75 Re |
76 Os |
77 Ir |
78 Pt |
79 Au |
80 Hg |
81 Tl |
82 Pb |
83 Bi |
84 Po |
85 At |
86 Rn | ||
| 7 | 87 Fr |
88 Ra |
103 Lr |
104 Rf |
105 Db |
106 Sg |
107 Bh |
108 Hs |
109 Mt |
110 Ds |
111 Rg |
112 Cn |
113 Nh |
114 Fl |
115 Mc |
116 Lv |
117 Ts |
118 Og | ||
| 57 La |
58 Ce |
59 Pr |
60 Nd |
61 Pm |
62 Sm |
63 Eu |
64 Gd |
65 Tb |
66 Dy |
67 Ho |
68 Er |
69 Tm |
70 Yb | |||||||
| 89 Ac |
90 Th |
91 Pa |
92 U |
93 Np |
94 Pu |
95 Am |
96 Cm |
97 Bk |
98 Cf |
99 Es |
100 Fm |
101 Md |
102 No | |||||||
- Legend
- Carbene complex with element known
- No carbene complex with element known
Carbenes in organometallic chemistry & catalysis
Carbenes can be stabilised as organometallic species. These transition metal carbene complexes fall into two categories:[citation needed]
- Fischer carbenes in which carbenes are tethered to a metal and an electron-withdrawing group (usually a carbonyl),
- electron-donating group. The reactions that such carbenes participate in are very different from those in which organic carbenes participate.
Triplet state carbene chemistry
Persistent triplet state carbenes are likely to have very similar reactivity as other non-persistent triplet state
Physical properties
Those carbenes that have been isolated to date tend to be colorless solids with low melting points. These carbenes tend to sublime at low temperatures under high vacuum.
One of the more useful physical properties is the diagnostic chemical shift of the carbenic carbon atom in the 13C-
Upon coordination to metal centers, the 13C carbene resonance usually shifts highfield, depending on the Lewis acidity of the complex fragment. Based on this observation, Huynh et al. developed a new methodology to determine ligand donor strengths by 13C NMR analysis of trans-palladium(II)-carbene complexes. The use of a 13C-labeled N-heterocyclic carbene ligand also allows for the study of mixed carbene-phosphine complexes, which undergo trans-cis-isomerization due to the trans effect.[65]
Applications

NHCs are widely used as
Preparation methods
NHCs are often strongly
In these cases, strong unhindered nucleophiles are avoided whether they are generated in situ or are present as an impurity in other reagents (such as LiOH in BuLi).
Several approaches have been developed in order to prepare stable carbenes, these are outlined below.
Deprotonation
Deprotonation of carbene precursor salts with strong bases has proved a reliable route to almost all stable carbenes:
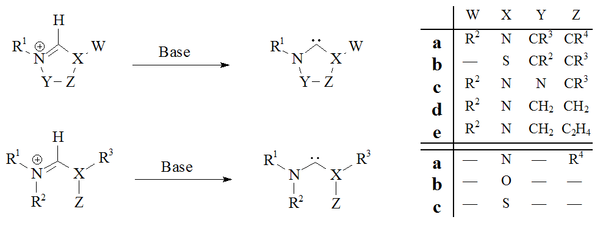
Imidazol-2-ylidenes and dihydroimidazol-2-ylidenes, such
Several bases and reaction conditions have been employed with varying success. The degree of success has been principally dependent on the nature of the precursor being deprotonated. The major drawback with this method of preparation is the problem of isolation of the free carbene from the metals ions used in their preparation.
Metal hydride bases
One might believe that sodium or
Deprotonation with sodium or potassium hydride in a mixture of liquid ammonia/THF at −40 °C has been reported[36] for imidazole-based carbenes. Arduengo and coworkers[33] managed to prepare a dihydroimidazol-2-ylidene using NaH. However, this method has not been applied to the preparation of diaminocarbenes. In some cases, potassium tert-butoxide can be employed without the addition of a metal hydride.[26]
Alkyllithiums
The use of
Amides bases
Lithium amides like the
Metal-free carbene preparation
The preparation of stable carbenes free from metal cations has been keenly sought to allow further study of the carbene species in isolation from these metals. Separating a carbene from a carbene-metal complex can be problematic due to the stability of the complex. Accordingly, it is preferable to make the carbene free from these metals in the first place. Indeed, some metal ions, rather than stabilising the carbene, have been implicated in the catalytic dimerisation of unhindered examples.
Shown right is an X-ray structure showing a complex between a diaminocarbene and potassium
Metal free carbenes have been prepared in several ways as outlined below:
Dechalcogenation
Another approach of preparing carbenes has relied on the

The
Vacuum pyrolysis
Vacuum pyrolysis, with the removal of neutral volatile byproducts i.e. methanol or chloroform, has been used to prepare dihydroimidazole and triazole based carbenes. Historically the removal of chloroform by

Bis(trimethylsilyl)mercury
For example:- (CH3)3Si−Hg−Si(CH3)3 + R2N=C(Cl)−NR+
2Cl− → R2N−C−NR2 + Hg(l) + 2(CH3)3SiCl
Photochemical decomposition
Persistent triplet state carbenes have been prepared by
Purification
Stable carbenes are very reactive, and so the minimum amount of handling is desirable using
References
- S2CID 672379.
- Chem. Ind.26: 893.
- .
- .
- PMID 23081675.
- ^ Chemists Approach Elusive Breslow Intermediate Carmen Drahl
- .
- ^ .
- ^ .
- .
- .
- .
- PMID 11093196.
- ^ .
- .
- ^ .
- ^ .
- ^ .
- PMID 19281270.
- .
- ^ .
- PMID 10796999.
- PMID 11397943.
- .
- ^ Rzepa, Henry (11 Sep 2016). "What's in a name? Carbenes: a reality check". Chemistry with a Twist. Retrieved 15 Feb 2024.
- ^ .
- ^ .
- ^ .
- ^ .
- ^ PMID 15467857.
- ^ doi:10.1039/a703610h.
- ^ .
- ^ .
- ^ .
- ^ PMID 16614171.
- ^ .
- ^ .
- ^ .
- ^ .
- ^ doi:10.1039/a808951e.
- .
- ^ .
- ^ .
- .
- ^ PMID 16028925.
- S2CID 4373216.
- .
- PMID 16417387.
- ^ a b R. W. Alder; P. R. Allen; S. J. Williams (1995). "Stable carbenes as strong bases". .
- PMID 23173841.
- S2CID 205757477.
- .
- PMID 15457494.
- .
- ^ a b Enders, D.; Breuer, K.; Teles, J.H.; Ebel, K. (1997). "1,3,4-Triphenyl-4,5-dihydro-1H-1,2,4-triazol-5-ylidene – applications of a stable carbene in synthesis and catalysis". .
- PMID 17372997.
- ^ PMID 17661300.
- .
- ^ S2CID 97336589.
- .
- ^ .
- ^ a b .
- ^ a b .
- ^ M. Scholl; T. M. Trnka; J. P. Morgan; R. H. Grubbs (1999). "Increased ring closing metathesis activity of ruthenium-based olefin metathesis catalysts coordinated with imidazolin-2-ylidene ligands". .
- ^ Han Vinh Huynh; et al. (2009). "13C NMR Spectroscopic Determination of Ligand Donor Strengths Using N-Heterocyclic Carbene Complexes of Palladium(II)". .
- ISBN 3-527-31400-8
- ISBN 3-540-36929-5
- S2CID 206902952.
- S2CID 43090499.
- doi:10.1039/b104110j.
- .
- .
- PMID 14746458.
Further reading
Reviews on persistent carbenes:
- Hopkinson, M. N.; Richter, C.; Schedler, M.; Glorius, F. (2014). "An Overview of N-Heterocyclic Carbenes". S2CID 672379..
- Carbene Chemistry: From Fleeting Intermediates to Powerful Reagents, (Chapter 4, Hideo Tomioka (triplet state); Chapter 5 (singlet state), Roger W. Alder) - ed. Guy Bertrand
- Reactive Intermediate Chemistry By Robert A. Moss, Matthew Platz, Maitland Jones (Chapter 8, Stable Singlet Carbenes, Guy Bertrand)
- R. W. Alder, in 'Diaminocarbenes: exploring structure and reactivity', ed. G. Bertrand, New York, 2002
- M. Regitz (1996). "Stable Carbenes—Illusion or Reality?". .
For a review on the physico-chemical properties (electronics, sterics, ...) of N-heterocyclic carbenes:
- T. Dröge; F. Glorius (2010). "The Measure of All Rings - N-Heterocyclic Carbenes". PMID 20715233.
