Organoboron chemistry

Organoboron chemistry or organoborane chemistry studies organoboron compounds, also called organoboranes. These
Organoboranes and -borates enable many
Properties of the B-C bond
The C-B bond has low polarity (electronegativity 2.55 for carbon and 2.04 for boron). Alkyl boron compounds are in general stable, though easily oxidized.
Boron often forms
Classes of organoboron compounds
Organoboranes and hydrides

The most-studied class of organoboron compounds has the formula BRnH3−n. These compounds are catalysts, reagents, and synthetic intermediates. The trialkyl and triaryl derivatives feature a
Borinic and boronic acids and esters (BRn(OR)3-n)
Compounds of the type BRn(OR)3-n are called
Boron clusters
Boron is renowned for cluster
Bora-substituted aromatic compounds
In borabenzene, boron replaces one CH center in benzene. Borabenzene and derivatives invariably appear as adducts, e.g., C5H5B-pyridine.
The cyclic compound borole, a structural analog of pyrrole, has not been isolated, but substituted derivatives (boroles) are known.
The cyclic compound borepin is aromatic.
Boryl compounds
Strong bases do not deprotonate boranes R2BH. Instead these reactions afford the octet-complete adduct R2HB-base.[7]
Compounds

Alkylideneboranes
Alkylideneboranes (RB=CRR) with a boron–carbon double bond are rare. One example is borabenzene. The parent compound, HB=CH2, can be detected at low temperature. The derivative CH3B=C(SiMe3)2 is fairly stable, but prone to cyclodimerisation.[10]
NHC adducts of boron
Diborenes
Boron-boron double bonds are extraordinarily rare. In 2007, the University of Georgia's Gregory Robinson presented the first neutral diborene (RHB=BHR):[12][13]

Each boron atom has an attached proton and is coordinated to a
A reported diboryne is based on similar chemistry.
Synthesis
From Grignard reagents
Simple organoboranes such as
addition will effect a borate (R4B−).Boronic acids RB(OH)2 react with potassium bifluoride K[HF2] to form trifluoroborate salts K[RBF3],[16] precursors to nucleophilic alkyl and aryl boron difluorides, ArBF2:[17]

From alkenes
In
Hydroboration with borane (BH3) equivalents converts only 33% of the starting olefin to product — boron-containing
By borylation
Metal-catalyzed borylation reactions produce an organoboron compound from aliphatic or aromatic C-H sigma bonds via a transition-metal catalyst. A common reagent is bis(pinacolato)diboron.
From other boron compounds
Carbon monoxide reacts with alkylboranes to form an unstable borane carbonyl. Then an alkyl substituent migrates from boron to the carbonyl carbon. For example, homologated primary alcohols result from organoboranes, carbon monoxide, and a reducing agent (here, sodium borohydride):[20]

Alkenylboranes
Alkynylboranes attack electrophiles to give trans alkenylboranes,[21] as in the first step of this olefin synthesis:

Reactions

The key property of organoboranes (R3B) and borates (R4B−, generated via addition of R− to R3B) is their susceptibility to reorganization. These compounds possess boron–carbon bonds polarized toward carbon. The boron-attached carbon is nucleophilic;[22] in borates, the nucleophicity suffices for intermolecular transfer to an electrophile.[23][3]
Boranes alone are generally not nucleophilic enough to transfer an R group intermolecularly. Instead, the group

An organic group's migration propensity depends on its ability to stabilize negative charge: alkynyl > aryl ≈ alkenyl > primary alkyl > secondary alkyl > tertiary alkyl.[24] Bis(norbornyl)borane and 9-BBN are often hydroboration reagents for this reason — only the hydroborated olefin is likely to migrate upon nucleophilic activation.
Migration retains configuration at the migrant carbon[25] and inverts it at the (presumably sp3-hybridized) terminus.[26] The resulting reorganized borane can then be oxidized or protolyzed to a final product.
Protonolysis
Organoboranes are unstable to Brønsted–Lowry acids, deboronating in favor of a proton. Consequently, organoboranes are easily removed from an alkane or alkene substrate, as in the second step of this olefin synthesis:[21]

Addition to halocarbonyls
α-Halo enolates are common nucleophiles in borane reorganization. After nucleophilic attack at boron, the resulting ketoboronate eliminates the halogen and tautomerizes to a neutral enolborane. A functionalized carbonyl compound then results from protonolysis,[27] or quenching with other electrophiles:

Because the migration is stereospecific, this method synthesizes enantiopure α-alkyl or -aryl ketones.[28]
α-Haloester enolates add similarly to boranes, but with lower yields:[29]
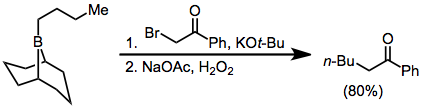

Diazoesters and diazoketones remove the requirement for external base.[30] α,α'-Dihalo enolates react with boranes to form α-halo carbonyl compounds that can be further functionalized at the α position.[31]
Addition to carbonyls
In allylboration, an
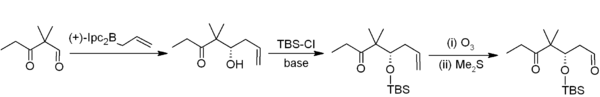
Trifluoroborate salts are stabler than boronic acids and selectively alkylate aldehydes:[34]

Oxygenation
The
Oxidation of an alkenylborane gives an boron-free enol.[35]
Halogenation
Organoborane activation with hydroxide or alkoxide and treatment with X2 yields haloalkanes. With excess base, two of the three alkyl groups attached to the boron atom may convert to halide, but disiamylborane permits only halogenation of the hydroborated olefin:[36]
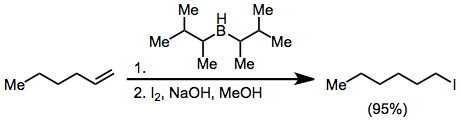
Treatment of an alkenylborane with iodine or bromine induces migration of a boron-attached organic group. Alkynyl groups migrate selectively, forming enynes after treatment with sodium acetate and hydrogen peroxide:[37]

Transmetalation and coupling
Organoboron compounds also
Reducing agents
Borane hydrides such as
Other synthetic applications
Alcohols
Homologated primary alcohols result from the treatment of organoboranes with carbon monoxide and a hydride:[39]

Tertiary alcohols with two identical groups attached to the alcohol carbon may be synthesized through an alkynylborane double migration:[35]

Carbonyl groups
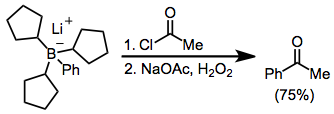
Organoborates anions reductively eliminate against acyl halides. Here, the borate was generated from tri(cyclopentyl)borane and phenyllithium; the three cyclopentyl groups do not significantly migrate:[40]
Applications
Organoboron chemistry is mainly of commercial value in the pharmaceutical industry.
Triethylborane was used to ignite the JP-7 fuel of the Pratt & Whitney J58 variable cycle engines powering the Lockheed SR-71 Blackbird.
Organoboron compounds have long been discussed for use as boron delivery agents in neutron capture therapy of cancer.[41]
References
Further reading
- Thomas, Susan E. The Roles of Boron and Silicon. Oxford Chemistry Primers No. 1; 1991.
Footnotes
- ^ Thomas 1991
- ISBN 3-527-29390-6– Wiley-VCH, Weinheim
- ^
- .
- ISBN 0-471-11280-1.
- ISBN 9780128019054.
- ISBN 3-527-30991-8
- S2CID 21040230.
- ^ Halford, Bethany. "Attacks: Electropositive element pressed into action as nucleophilic boryllithium" Chemical & Engineering News 2006; Volume 84 (41): 11
- .
- PMID 21898724.
- PMID 17887683.
- ^ Dagani, Ron. "Neutral Diborene Is A First" Chemical & Engineering News October 1, 2007 Volume 85, Number 40 p. 10
- PMID 23362015.
- (PDF) from the original on July 22, 2018.
- .
- PMID 19899086.
- ISBN 0-306-41088-5
- ^ Jacob III, P.; Brown, H. C. J. Org. Chem. 1977, 42, 579.
- ^ Rathke, M. W.; Brown, H. C. J. Am. Chem. Soc. 1967, 89, 2740.
- ^ a b Corey, E. J.; Ravindranathan, T. J. Am. Chem. Soc. 1972, 94, 4013.
- ^ Allred, A. L.; Rochow, E. G. J. Inorg. Nucl. Chem. 1958, 5, 264.
- ^ a b Negishi E.-I. J. Organometal. Chem. 1976, 108, 281.
- ^ Miyaura M.; Sasaki N.; Itoh M.; Suzuki A. Tetrahedron Lett. 1977, 173.
- ^ Zweifel, G. in Aspects of Mechanism and Organometallic Chemistry, J. H. Bewster, Ed., Plenum, 1978, p. 229.
- ^ Midland, M. M.; Zolopa, A. R.; Halterman, R. L. J. Am. Chem. Soc. 1979, 101, 248.
- ^ Brown, H. C.; Rogic M. M.; Nambu H.; Rathke, M. W. J. Am. Chem. Soc. 1969, 91, 2147.
- ^ Nesmeyanov, A. N.; Sokolik, R. A. The Organic Compounds of Boron, Aluminium, Gallium, Indium, and Thallium, North-Holland, Amsterdam, 1967.
- ^ Brown, H. C.; Rogic, M. M.; Rathke, M. W.; Kabalka, G. W. J. Am. Chem. Soc. 1968, 90, 818.
- ^ Hooz, J.; Gunn, D. M. J. Am. Chem. Soc. 1969, 91, 6195.
- ^ Pasto, D. J.; Wojtkowski, P. W. J. Org. Chem. 1971, 36, 1790.
- ISBN 978-0471264187.
- .
- S2CID 49223984.
- ^ a b Midland, M. M.; Brown, H. C. J. Org. Chem. 1975, 40, 2845.
- ^ Brown, H. C.; Rathke, M. W.; Rogic, M. M. J. Am. Chem. Soc. 1968, 90, 5038.
- ^ Negishi, E.-i.; Lew, G.; Yoshida T. Chem. Commun. 1973, 874.
- S2CID 53050782.
- ^ Rathke, M. W.; Brown, H. C. J. Am. Chem. Soc. 1967, 89, 2740.
- ^ Negishi E.-I.; Abramovitch, A.; Merrill, R. E. Chem. Commun. 1975, 138.
- PMID 29914561.

![{\displaystyle {\ce {R1-BY2{}+R2-X->[{} \atop {\underset {\text{catalyst}}{\ce {Pd}}}][{\text{Base}}]R1-R2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/4242d81e9b075c35d4c3fb6635b7ecd3bb4a865b)