Histone deacetylase
| Histone deacetylase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Histone deacetylase superfamily | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | Hist_deacetyl | ||||||||
SCOP2 | 1c3s / SCOPe / SUPFAM | ||||||||
| |||||||||
Histone deacetylases (EC 3.5.1.98, HDAC) are a class of enzymes that remove acetyl groups (O=C-CH3) from an ε-N-acetyl lysine amino acid on both histone and non-histone proteins.[2] HDACs allow histones to wrap the DNA more tightly.[3] This is important because DNA is wrapped around histones, and DNA expression is regulated by acetylation and de-acetylation. HDAC's action is opposite to that of histone acetyltransferase. HDAC proteins are now also called lysine deacetylases (KDAC), to describe their function rather than their target, which also includes non-histone proteins.[4] In general, they suppress gene expression.[5]
HDAC super family
Together with the acetylpolyamine amidohydrolases and the acetoin utilization proteins, the histone deacetylases form an ancient protein superfamily known as the histone deacetylase superfamily.[6]
Classes of HDACs in higher eukaryotes
HDACs, are classified in four classes depending on sequence homology to the yeast original enzymes and domain organization:[7]
| Class | Members | Catalytic sites | Subcellular localization | Tissue distribution | Substrates | Binding partners | Knockout phenotype |
|---|---|---|---|---|---|---|---|
| I | HDAC1 | 1 | Nucleus | Ubiquitous | Androgen receptor, SHP, p53, MyoD, E2F1, STAT3 | – | Embryonic lethal, increased histone acetylation, increase in p27
|
HDAC2 |
1 | Nucleus | Ubiquitous | Glucocorticoid receptor, YY1, BCL6, STAT3 | – | Cardiac defect | |
| HDAC3 | 1 | Nucleus | Ubiquitous | SHP, YY1, GATA1, RELA, STAT3, MEF2D | NCOR1[8] |
– | |
| HDAC8 | 1 | Nucleus/cytoplasm | Ubiquitous? | – | EST1B | – | |
| IIA | HDAC4 | 1 | Nucleus / cytoplasm | heart, skeletal muscle, brain | GCMA, GATA1, HP1 | RFXANK | Defects in chondrocyte differentiation |
HDAC5 |
1 | Nucleus / cytoplasm | heart, skeletal muscle, brain | SMAD7, HP1 |
REA, estrogen receptor | Cardiac defect | |
| HDAC7 | 1 | Nucleus / cytoplasm / mitochondria | heart, skeletal muscle, pancreas, placenta | PLAG1, PLAG2 | ACTN4, androgen receptor, Tip60 |
Maintenance of vascular integrity, increase in MMP10 | |
| HDAC9 | 1 | Nucleus / cytoplasm | brain, skeletal muscle | – | FOXP3 | Cardiac defect | |
| IIB | HDAC6 | 2 | Mostly cytoplasm | heart, liver, kidney, placenta | SMAD7 |
RUNX2 | – |
| HDAC10 | 1 | Mostly cytoplasm | liver, spleen, kidney | – | – | – | |
| III | SIRT7 ) |
– | – | – | – | – | – |
Sir2 in the yeast S. cerevisiae |
– | – | – | – | – | – | |
| IV | HDAC11 | 2 | Nucleus / cytoplasm | brain, heart, skeletal muscle, kidney | – | – | – |
HDAC (except class III) contain zinc and are known as Zn2+-dependent histone deacetylases.[9] They feature a classical arginase fold and are structurally and mechanistically distinct from sirtuins (class III), which fold into a Rossmann architecture and are NAD+ dependent.[10]
Subtypes
HDAC proteins are grouped into four classes (see above) based on function and DNA sequence similarity. Class I, II and IV are considered "classical" HDACs whose activities are inhibited by
Evolution
HDACs are conserved across evolution, showing orthologs in all eukaryotes and even in Archaea. All upper eukaryotes, including vertebrates, plants and arthropods, possess at least one HDAC per class, while most vertebrates carry the 11 canonical HDACs, with the exception of bone fish, which lack HDAC2 but appears to have an extra copy of HDAC11, dubbed HDAC12. Plants carry additional HDACs compared to animals, putatively to carry out the more complex transcriptional regulation required by these sessile organisms. HDACs appear to be deriving from an ancestral acetyl-binding domain, as HDAC homologs have been found in bacteria in the form of Acetoin utilization proteins (AcuC) proteins.[3]

Subcellular distribution
Within the Class I HDACs, HDAC 1, 2, and 3 are found primarily in the nucleus, whereas HDAC8 is found in both the nucleus and the cytoplasm, and is also membrane-associated. Class II HDACs (HDAC4, 5, 6, 7 9, and 10) are able to shuttle in and out of the nucleus, depending on different signals.[14][15]
HDAC6 is a cytoplasmic, microtubule-associated enzyme. HDAC6 deacetylates tubulin, Hsp90, and cortactin, and forms complexes with other partner proteins, and is, therefore, involved in a variety of biological processes.[16]
Function
Histone modification
Histone tails are normally positively charged due to
Histone deacetylase is involved in a series of pathways within the living system. According to the Kyoto Encyclopedia of Genes and Genomes (KEGG), these are:
- Environmental information processing; signal transduction; notch signaling pathway PATH:ko04330
- Cellular processes; cell growth and death; cell cycle PATH:ko04110
- Human diseases; cancers; chronic myeloid leukemia PATH:ko05220
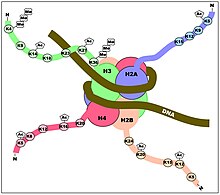

Histone acetylation plays an important role in the regulation of gene expression. Hyperacetylated chromatin is transcriptionally active, and hypoacetylated chromatin is silent. A study on mice found that a specific subset of mouse genes (7%) was deregulated in the absence of HDAC1.
Non-histone effects
It is a mistake to regard HDACs solely in the context of regulating gene transcription by modifying histones and chromatin structure, although that appears to be the predominant function. The function, activity, and stability of proteins can be controlled by
- aggresomes. Misfolded protein aggregates are tagged by ubiquitination and removed from the cytoplasm by dynein motors via the microtubule network to an organelle termed the aggresome. HDAC 6 binds polyubiquitinated misfolded proteins and links to dynein motors, thereby allowing the misfolded protein cargo to be physically transported to chaperones and proteasomes for subsequent destruction.[20] HDAC6 is important regulator of HSP90 function and its inhibitor proposed to treat metabolic disorders.[21]
- APE1/Ref-1 (APEX1) is a multifunctional protein possessing both DNA repair activity (on abasic and single-strand break sites) and transcriptional regulatory activity associated with oxidative stress. APE1/Ref-1 is acetylated by PCAF; on the converse, it is stably associated with and deacetylated by Class I HDACs. The acetylation state of APE1/Ref-1 does not appear to affect its DNA repair activity, but it does regulate its transcriptional activity such as its ability to bind to the PTH promoter and initiate transcription of the parathyroid hormone gene.[24][25]
- NF-κB is a key transcription factor and effector molecule involved in responses to cell stress, consisting of a p50/p65 heterodimer. The p65 subunit is controlled by acetylation via PCAF and by deacetylation via HDAC3 and HDAC6.[26]
These are just some examples of constantly emerging non-histone, non-chromatin roles for HDACs.
HDAC inhibitors
Histone deacetylase inhibitors (HDIs) have a long history of use in psychiatry and neurology as mood stabilizers and anti-epileptics, for example,
HDAC inhibitors have effects on non-histone proteins that are related to acetylation. HDIs can alter the degree of acetylation of these molecules and, therefore, increase or repress their activity. For the four examples given above (see Function) on HDACs acting on non-histone proteins, in each of those instances the HDAC inhibitor
The
Histone deacetylase inhibitors may modulate the latency of some viruses, resulting in reactivation.
Histone deacetylase inhibitors have shown activity against certain Plasmodium species and stages which may indicate they have potential in malaria treatment. It has been shown that HDIs accumulate acetylated histone H3K9/H3K14, a downstream target of class I HDACs.[42]
See also
- Histone acetyltransferase (HAT)
- Histone deacetylase inhibitor
- Histone methyltransferase (HMT)
- Histone-modifying enzymes
- RNA polymerase control by chromatin structure
References
- PMID 18614528.
- PMID 24691964.
- ^ PMID 32429325.
- S2CID 206520776.
- PMID 25746103.
- PMID 9278492.
- PMID 17951399.
- PMID 23292142.
- PMID 19459166.
- PMID 30534609.
- S2CID 2967911.
- PMID 18292778.
- PMID 22963873.
- PMID 12429021.
- PMID 16532030.
- PMID 18472263.
- PMID 16940178.
- PMID 17961987.
- PMID 16289629.
- PMID 18413721.
- PMID 22723278.
- PMID 18757404.
- PMID 18385463.
- PMID 14633989.
- PMID 18324520.
- PMID 17657723.
- S2CID 14344174.
- ^ "Scientists 'reverse' memory loss". BBC News. 2007-04-29. Retrieved 2007-07-08.
- ^ S2CID 248527466.
- PMID 20536416.
- PMID 14613312.
- S2CID 39017666.
- ^ Depletion of Latent HIV in CD4 Cells - Full Text View - ClinicalTrials.gov
- PMID 19345475.
- PMID 23547652.
- PMID 20669972.
- PMID 20614936.
- PMID 15822187.
- PMID 17694074.
- PMID 23223453.
- PMID 21458587.
- PMID 30151334.
External links
- Histone+deacetylase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Animation at Merck
