Porphyria cutanea tarda
| Porphyria cutanea tarda | |
|---|---|
| Other names | PCT |
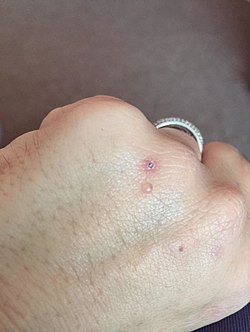 | |
| Blister on the hand of a person with porphyria cutanea tarda | |
| Specialty | Endocrinology |
Porphyria cutanea tarda is the most common subtype of porphyria.[1] The disease is named because it is a porphyria that often presents with skin manifestations later in life. The disorder results from low levels of the enzyme responsible for the fifth step in heme production. Heme is a vital molecule for all of the body's organs. It is a component of hemoglobin, the molecule that carries oxygen in the blood.
Symptoms and signs
Porphyria cutanea tarda (PCT) is recognized as the most prevalent subtype of porphyritic diseases.[3]
PCT is characterized by
Patients who are diagnosed with PCT typically seek treatment following the development of
Vitamin, mineral, and enzyme deficiencies
Certain vitamin and minerals deficiencies are common in people with porphyria cutanea tarda. The most common deficiencies are
Porphyrins interact with iron, absorbing photons to create reactive oxygen species is the mechanism of action causing the itchy, painful blisters of PCT.[7] The reactive oxygen species consume the skin antioxidants beta-carotene, vitamin E, and vitamin C. Supplementation of these three vitamins reduces the oxidation and potentially diminishes the severity of blister formation.[10] No single one of the three vitamins can inhibit the damaging effects of oxidized porphyrins, specifically uroporphyrins and coproporphyrins, but all three working together synergistically are capable of neutralizing their damaging effects.[citation needed]
Genetics

Inherited mutations in the
Nongenetic factors such as excess
The
In the 20% of cases where porphyria cutanea tarda is inherited, it is inherited in an
Other
While inherited deficiencies in uroporphyrinogen decarboxylase often lead to the development of PCT, there are a number of risk factors that can both cause and exacerbate the symptoms of this disease. One of the most common risk factors observed is infection with the Hepatitis C virus.[11] One review of a collection of PCT studies noted Hepatitis C infection in 50% of documented cases of PCT. Additional risk factors include alcohol use disorder, excess iron (from iron supplements as well as cooking on cast iron skillets), and exposure to chlorinated cyclic hydrocarbons and Agent Orange.[citation needed]
It can be a
Exacerbating factors
Pathogenesis
Porphyria cutanea tarda is primarily caused by uroporphyrinogen decarboxylase deficiency (UROD). Uroporphyrinogen decarboxylase occurs in nature as a homodimer of two subunits. It participates in the fifth step in heme synthesis pathway, and is active in the cytosol. This enzymatic conversion results in coproporphyrinogen III as the primary product. This is accomplished by the clockwise removal of the four carboxyl groups present in the cyclic uroporphyrinogen III molecule. Therefore, a deficiency in this enzyme causes the aforementioned buildup of uroporphyrinogen and hepta-carboxylic porphyrinogen, and to a lesser extent hexa-carboxylic porphyrinogen, and penta-carboxylic porphyrinogen in the urine, which can be helpful in the diagnosis of this disorder.[16][17]
The dermatological symptoms of PCT that include blistering and lesions on sun-exposed areas of the skin are caused by a buildup of porphyrin compounds (specifically uroporphyrinogen) close to the surface of the skin that have been oxidized by free radicals or sunlight.
The strong association of PCT with Hepatitis C virus infection is not entirely understood. Studies have suggested that the cytopathic effect of the virus on hepatocytes can lead to the release of free iron. This iron can disrupt the activity of cytochrome p450, releasing activated oxygen species. These can oxidize the UROD substrate uroporphyrinogen, which can result in the inhibition of UROD and lead to deficient activity of this key enzyme.[22]
Excess alcohol use is frequently associated with both inducing PCT[23] and aggravating a preexisting diagnosis of the disorder. It is thought to do so by causing oxidative damage to liver cells, resulting in oxidized species of uroporphyrinogen that inhibit the activity of hepatic UROD. It is also felt to increase the uptake of iron in liver cells, leading to further oxidation of uroporphyrinogen by the release of activated oxygen species. Additionally, exposure to chlorinated cyclic hydrocarbons can lead to a deficiency in the activity of uroporphyrinogen decarboxylase, causing the buildup of excess uroporphyrinogen. Additionally, alcohol has been shown to increase the activity of the delta-aminolevulinic acid synthetase (ALA synthetase), the rate-limiting enzymatic step in heme synthesis in the mitochondria, in rats.[24] Therefore, alcohol consumption may increase the production of uroporphyrinogen, exacerbating symptoms in individuals with porphyria cutanea tarda.[citation needed]
Diagnosis
While the most common symptom of PCT is the appearance of skin lesions and blistering, their appearance is not conclusive. Laboratory testing commonly reveals high levels of
Classification
Some sources divide PCT into two types: sporadic and familial.[2] Other sources include a third type,[25] but this is less common.
| Type | OMIM
|
Description |
|---|---|---|
| Type I ("sporadic") | 176090 | Type I porphyria cutanea tarda, the sporadic form, is indicated by UROD deficiency that is observed only in hepatic cells and nowhere else in the body. Genetically, these individuals will not exhibit deficiency in the UROD gene, although other genetic factors such as HFE deficiency (resulting in hemochromatosis and the buildup of iron in the liver) are thought to play a key role. Typically in these individuals, a variety of risk factors such as alcohol use disorder and Hepatitis C virus infection co-occur to result in the clinical manifestation of PCT. |
| Type II ("familial") | 176100 | Patients exhibiting Type II PCT have a specific deficiency in the UROD gene, passed down in an autosomal dominant pattern. Those possessing this deficiency are heterozygous for the UROD gene. They do not show a complete lack of functional uroporphyrinogen decarboxylase, only a deficient form of the enzyme that is marked by reduced conversion of uroporphyrinogen to coproporphyrinogen. Therefore, the expression of uroporphyrinogen decarboxylase will be reduced throughout the body of these individuals, while it is isolated to the liver in Type I patients. While this genetic deficiency is the main distinction between Type I and Type II PCT, the risk factors mentioned before are often seen in patients presenting with Type II PCT. In fact, many people who possess the deficient UROD gene often go their entire lives without having a clinical manifestation of PCT symptoms. |
| Type III | - | The least common is Type III, which is no different from Type I insofar as the patients possess normal UROD genes. Despite this, Type III PCT is observed in more than one family member, indicating a genetic component unrelated to the expression of uroporphyrinogen decarboxylase. |
One study used 74% as the cutoff for UROD activity, with those patients under that number being classified as type II, and those above classified as type III if there was a family history, and type I if there was not.[26]
Genetic variants associated with
Treatment
Since PCT is a chronic condition, a comprehensive management of the disease is the most effective means of treatment. Primarily, it is key that patients diagnosed with PCT avoid alcohol consumption, iron supplements, excess exposure to sunlight (especially in the summer), as well as estrogen and chlorinated cyclic hydrocarbons, all of which can potentially exacerbate the disorder. Additionally, the management of excess iron (due to the commonality of hemochromatosis in PCT patients) can be achieved through phlebotomy, whereby blood is systematically drained from the patient. A borderline iron deficiency has been found to have a protective effect by limiting heme synthesis. In the absence of iron, which is to be incorporated in the porphyrin formed in the last step of the synthesis, the mRNA of erythroid 5-aminolevulinate synthase (ALAS-2) is blocked by attachment of an iron-responsive element (IRE) binding cytosolic protein, and transcription of this key enzyme is inhibited.[27]
Low doses of
Epidemiology
PCT
]Society and culture
Porphyria cutanea tarda is implicated in the origin of vampire myths.[original research?] This is because people with the disease tend to avoid the sun due to photosensitivity and may develop disfigurement that eats away their noses, eyelids, lips, and gums giving their teeth a fang-like appearance. It has also been suggested[citation needed] they may have developed a craving for healthy blood to replace their own in a self medicated treatment in prior centuries.
Some folklore scholars[who?] claim that this is a mistake, first suggested in the 1990s,[citation needed] because vampires of myth did not have photosensitivity, nor were they described as looking like the modern incarnation of vampires. They were described as unintelligent roaming beings who fed on their victims to the point that they became reddened and heavily bloated, fattened on blood. Fangs were very rarely mentioned. Photosensitivity was not added to the vampire mythology until the 1922 film Nosferatu. Count Dracula of Bram Stroker's novel could walk about freely in daylight unharmed but not as powerful in the book.[citation needed]
Porphyria cutanea tarda is the name of a song by the rock band AFI on their fourth album Black Sails in the Sunset, released on May 18, 1999.[citation needed]
Porphyria cutanea tarda is the disease that both Dabney Pratt and Brother Rush suffer from in Virginia Hamilton's children's novel Sweet Whispers, Brother Rush.[citation needed]
References
- PMID 17360334.
- ^ a b "porphyria cutanea tarda" at Dorland's Medical Dictionary
- PMID 17516469.
- PMID 993332.
- S2CID 23741767.
- ^ Goljan, E. F. (2011). Pathology (3rd ed., rev. reprint.). Philadelphia, PA: Mosby/Elsevier.[page needed]
- ^ S2CID 44998779.
- PMID 10669994.
- ^ S2CID 23170672.
- PMID 11809377.
- PMID 18855993.
- PMID 17891697.
- ^ S2CID 1787139.
- PMID 10189391.
- ^ "Porphyria Cutanea Tarda (PCT)". 2020-01-12.
- ^ "Porphyrin Tests". 7 May 2020.
- PMID 1008499.
- PMID 8274157.
- PMID 1791486.
- PMID 2488874.
- PMID 22510500.
- PMID 22510500.
- ^ Porphyria Cutanea Tarda at eMedicine
- PMID 838185.
- S2CID 43785629.
- PMID 12699242.
- S2CID 8470078.
- PMID 22985607.
- ^ a b c "Enforcement Reports". www.accessdata.fda.gov.
- PMID 22276975.
- PMID 22079977.
- PMID 20864177.
- PMID 14278818.
- PMID 4744026.
- S2CID 35585037.
- ISBN 978-1-4051-3400-2.[page needed]
External links
- Porphyria cutanea tarda at NIH's Office of Rare Diseases
