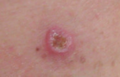
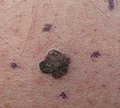
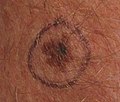
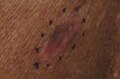
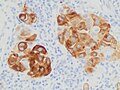
Melanoma
| Melanoma | |
|---|---|
| Other names | Malignant melanoma |
Five-year survival rates in US 99% (localized), 25% (disseminated)[4] | |
| Frequency | 3.1 million (2015)[5] |
| Deaths | 59,800 (2015)[6] |
Melanoma is the most dangerous type of skin cancer; it develops from the melanin-producing cells known as melanocytes.[1] It typically occurs in the skin, but may rarely occur in the mouth, intestines, or eye (uveal melanoma).[1][2] In women, melanomas most commonly occur on the legs; while in men, on the back.[2] Melanoma is frequently referred to as malignant melanoma. However, the medical community stresses that there is no such thing as a 'benign melanoma' and recommends that the term 'malignant melanoma' should be avoided as redundant.[7][8][9]
About 25% of melanomas develop from moles.[2] Changes in a mole that can indicate melanoma include increase—especially rapid increase—in size, irregular edges, change in color, itchiness, or skin breakdown.[1]
The primary cause of melanoma is
Avoiding UV light and using
Melanoma is the most dangerous type of skin cancer.
Signs and symptoms
Early signs of melanoma are changes to the shape or color of existing
- Asymmetry
- Borders (irregular with edges and corners)
- Colour (variegated)
- Diameter (greater than 6 mm (0.24 in), about the size of a pencil eraser)
- Evolving over time
This classification does not apply to nodular melanoma, which has its own classifications:[16]
- Elevated above the skin surface
- Firm to the touch
- Growing
Metastatic melanoma may cause nonspecific paraneoplastic symptoms, including loss of appetite, nausea, vomiting, and fatigue. Metastasis (spread) of early melanoma is possible, but relatively rare; less than a fifth of melanomas diagnosed early become metastatic. Brain metastases are particularly common in patients with metastatic melanoma.[17] It can also spread to the liver, bones, abdomen, or distant lymph nodes.[citation needed]
Cause
Melanomas are usually caused by DNA damage resulting from exposure to UV light from the sun. Genetics also play a role.[18][19] Melanoma can also occur in skin areas with little sun exposure (i.e. mouth, soles of feet, palms of hands, genital areas).[20] People with dysplastic nevus syndrome, also known as familial atypical multiple mole melanoma, are at increased risk for the development of melanoma.[21]
Having more than 50 moles indicates an increased risk in melanoma might arise. A weakened immune system makes cancer development easier due to the body's weakened ability to fight cancer cells.[18]
UV radiation
UV radiation exposure from tanning beds increases the risk of melanoma.[22] The International Agency for Research on Cancer finds that tanning beds are "carcinogenic to humans" and that people who begin using tanning devices before the age of thirty years are 75% more likely to develop melanoma.[23]
Those who work in airplanes also appear to have an increased risk, believed to be due to greater exposure to UV.[24]
Exposure to radiation (UVA and UVB) is a major contributor to developing melanoma.
Possible significant elements in determining risk include the intensity and duration of sun exposure, the age at which sun exposure occurs, and the degree of
Incurring multiple severe sunburns increases the likelihood that future sunburns develop into melanoma due to cumulative damage.[18] UV-high sunlight and tanning beds are the main sources of UV radiation that increase the risk for melanoma[32] and living close to the equator increases exposure to UV radiation.[18]
Genetics
A number of rare mutations, which often run in families, greatly increase melanoma susceptibility.[33] Several genes increase risks. Some rare genes have a relatively high risk of causing melanoma; some more common genes, such as a gene called MC1R that causes red hair, have a relatively lower elevated risk. Genetic testing can be used to search for the mutations.[citation needed]
One class of mutations affects the gene
Familial melanoma is genetically heterogeneous,
Other mutations confer lower risk, but are more common in the population. People with mutations in the
Fair- and red-haired people, persons with multiple atypical
A family history of melanoma greatly increases a person's risk, because mutations in several genes have been found in melanoma-prone families.[39][18] People with a history of one melanoma are at increased risk of developing a second primary tumor.[40]
Fair skin is the result of having less melanin in the skin, which means less protection from UV radiation exists.[18]
Pathophysiology


The earliest stage of melanoma starts when melanocytes begin out-of-control growth. Melanocytes are found between the outer layer of the skin (the
When the tumor cells start to move in a different direction – vertically up into the epidermis and into the
The next step in the evolution is the invasive radial growth phase, in which individual cells start to acquire invasive potential. From this point on, melanoma is capable of spreading.[
The vertical growth phase (VGP) following is invasive melanoma. The tumor becomes able to grow into the surrounding tissue and can spread around the body through blood or
The host elicits an immunological reaction against the tumor during the VGP,
A cause common to most cancers is damage to DNA.
If unrepaired, cyclobutane pyrimidine dimer (CPD) photoproducts can lead to mutations by inaccurate
The entire genomes of 25 melanomas were sequenced.[48] On average, about 80,000 mutated bases (mostly C>T transitions) and about 100 structural rearrangements were found per melanoma genome. This is much higher than the roughly 70 mutations across generations (parent to child).[49][50] Among the 25 melanomas, about 6,000 protein-coding genes had missense, nonsense, or splice site mutations. The transcriptomes of over 100 melanomas has also been sequenced and analyzed. Almost 70% of all human protein-coding genes are expressed in melanoma. Most of these genes are also expressed in other normal and cancer tissues, with some 200 genes showing a more specific expression pattern in melanoma compared to other forms of cancer. Examples of melanoma specific genes are tyrosinase, MLANA, and PMEL.[51][52]
UV radiation causes
Cancer stem cells may also be involved.[59]
Gene mutations
Large-scale studies, such as The Cancer Genome Atlas, have characterized recurrent somatic alterations likely driving initiation and development of cutaneous melanoma. The Cancer Genome Atlas study has established four subtypes : BRAF mutant, RAS mutant, NF1 mutant, and triple wild-type.[60]
The most frequent mutation occurs in the 600th codon of
In some cases (3-7%) mutated versions of BRAF and NRAS undergo
Metastasis
The research done by Sarna's team proved that heavily pigmented melanoma cells have Young's modulus about 4.93, when in non-pigmented ones it was only 0.98.[67] In another experiment they found that elasticity of melanoma cells is important for its metastasis and growth: non-pigmented tumors were bigger than pigmented and it was much easier for them to spread. They shown that there are both pigmented and non-pigmented cells in melanoma tumors, so that they can both be drug-resistant and metastatic.[67]
Diagnosis


Looking at or visually inspecting the area in question is the most common method of suspecting a melanoma.[68] Moles that are irregular in color or shape are typically treated as candidates. To detect melanomas (and increase survival rates), it is recommended to learn to recognize them (see "ABCDE" mnemonic), to regularly examine moles for changes (shape, size, color, itching or bleeding) and to consult a qualified physician when a candidate appears.[69][70] In-person inspection of suspicious skin lesions is more accurate than visual inspection of images of suspicious skin lesions.[71] When used by trained specialists, dermoscopy is more helpful to identify malignant lesions than use of the naked eye alone.[72] Reflectance confocal microscopy may have better sensitivity and specificity than dermoscopy in diagnosing cutaneous melanoma but more studies are needed to confirm this result.[73]
However, many melanomas present as lesions smaller than 6 mm in diameter, and all melanomas are malignant when they first appear as a small dot. Physicians typically examine all moles, including those less than 6 mm in diameter. Seborrheic keratosis may meet some or all of the ABCD criteria, and can lead to false alarms. Doctors can generally distinguish seborrheic keratosis from melanoma upon examination or with dermatoscopy.[citation needed]
Some advocate replacing "enlarging" with "evolving": moles that change and evolve are a concern. Alternatively, some practitioners prefer "elevation". Elevation can help identify a melanoma, but lack of elevation does not mean that the lesion is not a melanoma. Most melanomas in the US are detected before they become elevated. By the time elevation is visible, they may have progressed to the more dangerous invasive stage.[citation needed]
-
Melanoma in skin biopsy with H&E stain – this case may represent superficial spreading melanoma.
-
Lymph node with almost complete replacement by metastatic melanoma. The brown pigment is a focal deposition of melanin.
-
Adermatoscope
-
Malignant Melanoma, right posterior thigh
-
Melanoma in situ, vertex scalp marked for biopsy
-
Malignant Melanoma in situ, evolving, right clavicle marked for biopsy
-
Malignant Melanoma, vertex scalp marked for biopsy
-
Malignant Melanoma, right medial thigh marked for biopsy
-
Malignant Melanoma, right posterior shoulder circled for biopsy
-
Malignant Melanoma, left forearm marked for biopsy
-
Malignant Melanoma left forearm post excision with purse-string closure
-
Melanoma in situ, right forehead marked for biopsy
-
Melanoma in situ, dermatoscope image, right forehead marked for biopsy
-
Malignant Melanoma in situ, evolving, a medial right temple with adjacent sebaceous hyperplasia, lateral
-
Malignant Melanoma in situ, left anterior shoulder marked for biopsy
-
Malignant Melanoma in situ, right anterior shoulder marked for biopsy
-
Malignant Melanoma in situ, left upper inner arm
-
Malignant Melanoma in situ marked for biopsy, left forearm
-
Malignant Melanoma in situ, right upper medial back, marked for biopsy
-
Malignant Melanoma, mid frontal scalp
-
Malignant melanoma, left mid-back marked for biopsy
-
Malignant melanoma, left mid-back marked for biopsy, through dermatoscope
-
Gross pathology of melanoma metastasis, which is pigment-forming in a vast majority of cases, giving it a dark appearance.
-
Histopathology of a metastatic melanoma to a lymph node, H&E stain, showing poorly differentiated cells
-
Metastatic melanoma onMelan-A, which helps in diagnosing uncertain cases
-
Metastatic melanoma on immunohistochemistry for SOX10, another helpful stain in uncertain cases
Ugly duckling
One method is the "
Amelanotic melanomas and melanomas arising in fair-skinned individuals are very difficult to detect, as they fail to show many of the characteristics in the ABCD rule, break the "ugly duckling" sign, and are hard to distinguish from acne scarring, insect bites,
Biopsy
Following a visual examination and a dermatoscopic exam,
Total body photography, which involves photographic documentation of as much body surface as possible, is often used during follow-up for high-risk patients. The technique has been reported to enable early detection and provides a cost-effective approach (with any digital camera), but its efficacy has been questioned due to its inability to detect macroscopic changes.[68] The diagnosis method should be used in conjunction with (and not as a replacement for) dermoscopic imaging, with a combination of both methods appearing to give extremely high rates of detection.
Histopathologic types
Melanoma is a type of neuroectodermal neoplasm.[81] There are four main types of melanoma:[82]
| SN | Type | Features | Incidence[82][notes 1] | Photograph | Micrograph |
|---|---|---|---|---|---|
| 1. | Superficial spreading melanoma | Melanoma cells with nest formation along the dermo-epidermal junction. | 70% | 
|

|
| 2. | Nodular melanoma | Grows relatively more in depth than in width. | 15% - 20% | 
|

|
| 3. | Lentigo maligna melanoma | Linear spread of atypical epidermal melanocytes as well as invasion into the dermis.[83] | 5% - 10% | 
|

|
| 4. | Acral lentiginous melanoma | Continuous proliferation of atypical melanocytes at the dermoepidermal junction.[84] | 7% - 10% | 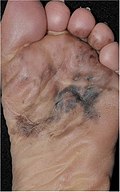
|

|
Other histopathologic types are:

- Mucosal melanoma; When melanoma occurs on mucous membranes.
- Desmoplastic melanoma
- Melanoma with small nevus-like cells
- Melanoma with features of a Spitz nevus
- Uveal melanoma
- Vaginal melanoma
- Polypoid melanoma, a subclass of nodular melanoma.
In situ or invasive
A melanoma in situ has not invaded beyond the basement membrane, whereas an invasive melanoma has spread beyond it.
Some histopathological types of melanoma are inherently invasive, including nodular melanoma and lentigo maligna melanoma, where the in situ counterpart to lentigo maligna melanoma is lentigo maligna.[85] Lentigo maligna is sometimes classified as a very early melanoma,[86] and sometimes a precursor to melanoma.[87]
Superficial spreading melanomas and acral lentiginous melanomas can be either in situ or invasive,[88] but acral lentiginous melanomas are almost always invasive.[89]
Staging
Further context on cancer staging is available at TNM.

Metastatic melanomas can be detected by X-rays, CT scans, MRIs, PET and PET/CTs, ultrasound, LDH testing and photoacoustic detection.[90] However, there is lack of evidence in the accuracy of staging of people with melanoma with various imaging methods.[91]
Melanoma stages according to AJCC, 8th edition:[92]
- TX: Primary tumor thickness cannot be assessed (such as a diagnosis by curettage)
- T0: No evidence of primary tumor (such as unknown primary or completely regressed melanoma)
| Stage | T category[92] | Thickness[92] | Ulceration[92] |
|---|---|---|---|
| Stage 0 | Melanoma in situ | ||
| Stage I | T1a | Less than 0.8 mm | No |
| T1b | Less than 0.8 mm | Yes | |
| >0.8 to 1.0 mm | Yes or no | ||
| T2a | >1.0 to 2.0 mm | No | |
| Stage II | T2b | >1.0 to 2.0 mm | Yes |
| T3a | >2.0 to 4.0 mm | No | |
| T3b | >2.0 to 4.0 mm | Yes | |
| T4a | >4.0 mm | No | |
| T4b | >4.0 mm | Yes | |
Stage 1 and 2 require an N (lymph node) class of:
- N0 – No regional metastases.[92]
| Stage | N category | Number of tumor-involved regional lymph nodes | Presence of in-transit, satellite, and/or microsatellite metastases |
|---|---|---|---|
| N/A | NX | Regional nodes not assessed (such as sentinel lymph node biopsy not performed, or regional nodes previously removed for another reason)[notes 2] | |
| Stage III | N1 | One involved lymph node, or any number of in-transit, satellite, and/or microsatellite metastases with no tumor-involved nodes. | |
| N1a | One clinically occult (that is, detected by sentinel node biopsy) | No | |
| N1b | One clinically detected | No | |
| N1c | No regional lymph node disease | Yes | |
| N2 | Two or 3 tumor‐involved nodes or any number of in‐transit, satellite, and/or microsatellite metastases with one tumor‐involved node | ||
| N2a | Two or 3 clinically occult (that is, detected by sentinel node biopsy) | No | |
| N2b | Two or 3, at least one of which was clinically detected | No | |
| N2c | One clinically occult or clinically detected | Yes | |
| N3 | Four or more tumor‐involved nodes or any number of in‐transit, satellite, and/or microsatellite metastases with 2 or more tumor‐involved nodes, or any number of matted nodes without or with in‐transit, satellite, and/or microsatellite metastases | ||
| N3a | Four or more clinically occult (that is, detected by sentinel node biopsy) | No | |
| N3b | Four or more, at least one of which was clinically detected, or the presence of any number of matted nodes | No | |
| N3c | Two or more clinically occult or clinically detected and/or presence of any number of matted nodes | Yes | |
Stage 1, 2 and 3 require an M (metastasis status) of:
- M0: No evidence of distant metastasis
| Stage | M category | Anatomic site | lactate dehydrogenase (LDH) level |
|---|---|---|---|
| Stage IV | M1 | Evidence of distant metastasis | |
| M1a | Distant metastasis to the skin, soft tissue including muscle, and/or non-regional lymph node | Not recorded or unspecified | |
| M1a(0) | Not elevated | ||
| M1a(1) | Elevated | ||
| M1b | Distant metastasis to lung with or without metastasis at M1a sites | Not recorded or unspecified | |
| M1b(0) | Not elevated | ||
| M1b(1) | Elevated | ||
| M1c | Distant metastasis to non‐CNS visceral sites, with or without metastasis to M1a or M1b sites | Not recorded or unspecified | |
| M1c(0) | Not elevated | ||
| M1c(1) | Elevated | ||
| M1d | Distant metastasis to CNS, with or without metastasis to M1a, M1b, or M1c sites | Not recorded or unspecified | |
| M1d(0) | Not elevated | ||
| M1d(1) | Elevated | ||
Older systems include "

Laboratory
Lactate dehydrogenase (LDH) tests are often used to screen for metastases, although many patients with metastases (even end-stage) have a normal LDH; extraordinarily high LDH often indicates the metastatic spread of the disease to the liver.
It is common for patients diagnosed with melanoma to have chest X-rays and an LDH test, and in some cases
HMB-45 is a monoclonal antibody that reacts against an antigen present in melanocytic tumors such as melanomas. It is used in anatomic pathology as a marker for such tumors. The antibody was generated to an extract of melanoma. It reacts positively against melanocytic tumors but not other tumors, thus demonstrating specificity and sensitivity. The antibody also reacts positively against junctional nevus cells but not intradermal nevi, and against fetal melanocytes but not normal adult melanocytes.
HMB-45 is nonreactive with almost all non-melanoma human malignancies, with the exception of rare tumors showing evidence of melanogenesis (e.g., pigmented schwannoma, clear cell sarcoma) or tumors associated with tuberous sclerosis complex (angiomyolipoma and lymphangiomyoma).
Prevention
There is no evidence to support or refute adult population screening for melanoma.[93]
Ultraviolet radiation
Minimizing exposure to sources of ultraviolet radiation (the sun and sunbeds),[94] following sun protection measures and wearing sun protective clothing (long-sleeved shirts, long trousers, and broad-brimmed hats) can offer protection.
Using artificial light for tanning was once believed to help prevent skin cancers, but it can actually lead to an increased incidence of melanomas.[95]
UV nail lamps, which are used in nail salons to dry nail polish, are another common and widespread source of UV radiation that could be avoided.[96][97] Although the risk of developing skin cancer through UV nail lamp use is low, it is still recommended to wear fingerless gloves and/or apply SPF 30 or greater sunscreen to the hands before using a UV nail lamp.[96][97]
The body uses UV light to generate vitamin D so there is a need to balance getting enough sunlight to maintain healthy vitamin D levels and reducing the risk of melanoma; it takes around a half-hour of sunlight for the body to generate its vitamin D for the day and this is about the same amount of time it takes for fair-skinned people to get a sunburn. Exposure to sunlight can be intermittent instead of all at one time.[98]
Sunscreen
Concerns have been raised that sunscreen might create a false sense of security against sun damage.[101]
Medications
A 2005 review found tentative evidence that statin and fibrate medication may decrease the risk of melanoma.[102] A 2006 review however did not support any benefit.[103]
Treatment

Confirmation of the clinical diagnosis is done with a skin biopsy. This is usually followed up with a wider excision of the scar or tumor. Depending on the stage, a sentinel lymph node biopsy may be performed. Controversy exists around trial evidence for sentinel lymph node biopsy;[104] with unclear evidence of benefit as of 2015.[105] Treatment of advanced malignant melanoma is performed from a multidisciplinary approach.
Surgery
Excisional biopsies may remove the tumor, but further surgery is often necessary to reduce the risk of recurrence. Complete surgical excision with adequate
Mohs surgery has been reported with cure rate as low as 77%[109] and as high as 98.0% for melanoma-in-situ.[110] CCPDMA and the "double scalpel" peripheral margin controlled surgery is equivalent to Mohs surgery in effectiveness on this "intra-epithelial" type of melanoma.
Melanomas that spread usually do so to the
Biopsy of sentinel lymph nodes is a widely used procedure when treating cutaneous melanoma.[112][113]
Neither sentinel lymph node biopsy nor other diagnostic tests should be performed to evaluate early, thin melanoma, including melanoma in situ, T1a melanoma or T1b melanoma ≤ 0.5mm.[114] People with these conditions are unlikely to have the cancer spread to their lymph nodes or anywhere else and have a 5-year survival rate of 97%.[114] Because of these considerations, sentinel lymph node biopsy is considered unnecessary health care for them.[114] Furthermore, baseline blood tests and radiographic studies should not be performed only based on identifying this kind of melanoma, as there are more accurate tests for detecting cancer and these tests have high false-positive rates.[114] To potentially correct false positives, gene expression profiling may be used as auxiliary testing for ambiguous and small lesions.[115][116]
Sentinel lymph node biopsy is often performed, especially for T1b/T2+ tumors, mucosal tumors, ocular melanoma and tumors of the limbs.[
If a lymph node is positive, depending on the extent of lymph node spread, a radical lymph node dissection will often be performed. If the disease is completely resected, the patient will be considered for adjuvant therapy. Excisional
Add on treatment
Adjuvant treatment after surgery may reduce the risk of recurrence after surgery, especially in high-risk melanomas. Routines vary in different countries, but today (2024) the most common adjuvant treatment is immune checkpoint inhibitor treatment for up to a year post-surgery.[118]
In the early 2000's, a relatively common strategy was to treat patients with high risk of recurrence with up to a year of high-dose interferon treatment, which has severe side effects, but may improve the patient's prognosis slightly.[119] A 2013 meta-analysis suggested that the addition of interferon alpha increased disease-free and averall survival for people with AJCC TNM stage II-III cutaneous melanoma.[120] A 2011 meta-analysis showed that interferon could lengthen the time before a melanoma comes back but increased survival by only 3% at 5 years. The unpleasant side effects also greatly decrease quality of life.[121] In the European Union, interferon is usually not used outside the scope of clinical trials.[122][123]
Chemotherapy
Chemotherapy drugs such as Dacarbazine have been the backbone of metastatic melanoma treatment since FDA approval in 1975; however, its efficacy in terms of survival has never been proven in an RCT.[124] Since the approval of immune checkpoint inhibitors, dacarbazine and its oral counterpart temozolomide constitute potential treatment options in later lines of therapy.[125]
In people with locally advanced cutaneous malignancies and sarcoma, isolated limb infusion (ILI) has been found to be a minimally invasive and well-tolerated procedure for delivering regional chemotherapy.[126][127]
Targeted therapy
Melanoma cells have mutations that allow them to survive and grow indefinitely in the body.
A number of treatments improve survival over traditional chemotherapy.[124] Biochemotherapy (chemotherapy with cytokines IL-2 and IFN-α) combined with BRAF inhibitors improved survival for people with BRAF positive melanoma.[124] Biochemotherapy alone did not improve overall survival and had higher toxicity than chemotherapy.[124] Combining multiple chemotherapy agents (polychemotherapy) did not improve survival over monochemotherapy.[124] Targeted therapies result in relatively short progression-free survival (PFS) times. The therapy combination of dabrafenib and trametinib has a 3-year PFS of 23%, and 5-year PFS of 13%.[132]
Lifileucel (Amtagvi) is a tumor-derived autologous T cell immunotherapy that was approved for medical use in the United States in February 2024.[133][134]
Immunotherapy
Immunotherapy is aimed at stimulating the person's immune system against the tumor, by enhancing the body's own ability to recognize and kill cancer cells.[135] The current approach to treating melanoma with immunotherapy includes three broad categories of treatments including cytokines, immune check point inhibitors, and adoptive cell transfer.[135] These treatment options are most often used in people with metastatic melanoma and significantly improves overall survival.[124] However, these treatments are often costly. For example, one immune check point inhibitor treatment, pembrolizumab, costs $10,000 to US$12,000 for a single dose administered every 3 weeks.[136]
Cytokine therapies used for melanoma include
Immune check point inhibitors include anti-
Ongoing research is looking at treatment by adoptive cell transfer.[140] Adoptive cell transfer refers to the application of pre-stimulated, modified T cells or dendritic cells and is presently used to minimize complications from graft-versus-host disease.[137][141]
The combination nivolumab/relatlimab (Opdualag) was approved for medical use in the United States in March 2022.[142]
Lentigo maligna
Standard excision is still being done by most surgeons. Unfortunately, the recurrence rate is exceedingly high (up to 50%). This is due to the ill-defined visible surgical margin, and the facial location of the lesions (often forcing the surgeon to use a narrow surgical margin). The narrow surgical margin used, combined with the limitation of the standard "bread-loafing" technique of fixed tissue histology – result in a high "false negative" error rate, and frequent recurrences. Margin control (peripheral margins) is necessary to eliminate the false negative errors. If bread loafing is used, distances from sections should approach 0.1 mm to assure that the method approaches complete margin control. A meta-analysis of the literature in 2014 found no randomized controlled trials of surgical interventions to treat lentigo maligna or melanoma in-situ, even though surgery is the most widely used treatment.[143]
Mohs surgery has been done with cure rate reported to be as low as 77%,[109] and as high as 95% by another author.[110] The "double scalpel" peripheral margin controlled excision method approximates the Mohs method in margin control, but requires a pathologist intimately familiar with the complexity of managing the vertical margin on the thin peripheral sections and staining methods.[144]
Some melanocytic nevi, and melanoma-in-situ (lentigo maligna) have resolved with an experimental treatment, imiquimod (Aldara) topical cream, an immune enhancing agent. Some derma-surgeons are combining the 2 methods: surgically excising the cancer and then treating the area with Aldara cream postoperatively for three months. While some studies have suggested the adjuvant use of topical tazarotene, the current evidence is insufficient to recommend it and suggests that it increases topical inflammation, leading to lower patient compliance.[143]
Radiation
Radiation therapy is often used after surgical resection for patients with locally or regionally advanced melanoma or for patients with un-resectable distant metastases. Kilovoltage x-ray beams are often used for these treatments and have the property of the maximum radiation dose occurring close to the skin surface.[145] It may reduce the rate of local recurrence but does not prolong survival.[146] Radioimmunotherapy of metastatic melanoma is currently under investigation. Radiotherapy has a role in the palliation of metastatic melanoma.[147]
Prognosis


Factors that affect prognosis include:
- tumor thickness in millimeters (Breslow's depth),
- depth related to skin structures (Clark level),
- type of melanoma,
- presence of ulceration,
- presence of lymphatic/perineural invasion,
- presence of tumor-infiltrating lymphocytes (if present, prognosis is better),
- location of lesion,
- presence of satellite lesions, and
- presence of regional or distant metastasis.[148]
Certain types of melanoma have worse prognoses but this is explained by their thickness. Less invasive melanomas even with lymph node metastases carry a better prognosis than deep melanomas without regional metastasis at time of staging. Local recurrences tend to behave similarly to a primary unless they are at the site of a wide local excision (as opposed to a staged excision or punch/shave excision) since these recurrences tend to indicate lymphatic invasion.
When melanomas have spread to the lymph nodes, one of the most important factors is the number of nodes with malignancy. Extent of malignancy within a node is also important; micrometastases in which malignancy is only microscopic have a more favorable prognosis than macrometastases. In some cases micrometastases may only be detected by special staining, and if malignancy is only detectable by a rarely employed test known as the polymerase chain reaction (PCR), the prognosis is better. Macro-metastases in which malignancy is clinically apparent (in some cases cancer completely replaces a node) have a far worse prognosis, and if nodes are matted or if there is extracapsular extension, the prognosis is worse still. In addition to these variables, expression levels and copy number variations of a number of relevant genes may be used to support assessment of malignant melanoma prognosis.[115][116]
Stage IV melanoma, in which it has metastasized, is the most deadly skin malignancy: five-year survival is 22.5%.[132] When there is distant metastasis, the cancer is generally considered incurable. The five-year survival rate is less than 10%.[149] The median survival is 6–12 months. Treatment is palliative, focusing on life extension and quality of life. In some cases, patients may live many months or even years with metastatic melanoma (depending on the aggressiveness of the treatment). Metastases to skin and lungs have a better prognosis. Metastases to brain, bone and liver are associated with a worse prognosis. Survival is better with metastasis in which the location of the primary tumor is unknown.[150]
There is not enough definitive evidence to adequately stage, and thus give a prognosis for, ocular melanoma and melanoma of soft parts, or mucosal melanoma (e.g., rectal melanoma), although these tend to metastasize more easily. Even though regression may increase survival, when a melanoma has regressed, it is impossible to know its original size and thus the original tumor is often worse than a pathology report might indicate.
About 200 genes are prognostic in melanoma, with both unfavorable genes where high expression is correlated to poor survival and favorable genes where high expression is associated with longer survival times. Examples of unfavorable genes are MCM6 and TIMELESS; an example of a favorable gene is WIPI1.[51][52]
An increased neutrophil-to-lymphocyte ratio is associated with worse outcomes.[151][152][153]
Epidemiology

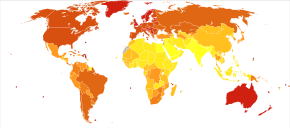
Globally, in 2012, melanoma occurred in 232,000 people and resulted in 55,000 deaths.[2] Australia and New Zealand have the highest rates of melanoma in the world.[2] It has become more common in the last 20 years in areas that are mostly Caucasian.[2]
The rate of melanoma has increased in the recent years, but it is not clear to what extent changes in behavior, in the environment, or in early detection are involved.[155]
Australia
Australia has a very high – and increasing – rate of melanoma. In 2012, deaths from melanoma occurred in 7.3–9.8 per 100,000 population. In Australia, melanoma is the third most common cancer in either sex; indeed, its incidence is higher than for lung cancer, although the latter accounts for more deaths. It is estimated that in 2012, more than 12,000 Australians were diagnosed with melanoma: given Australia's modest population, this is better expressed as 59.6 new cases per 100,000 population per year; >1 in 10 of all new cancer cases were melanomas.[156] Melanoma incidence in Australia is matter of significance, for the following reasons:
- Australian melanoma incidence has increased by more than 30 per cent between 1991 and 2009.
- Australian melanoma age-standardized incidence rates were, as of 2008, at least 12 times higher than the world average.
- Australian melanoma incidence is, by some margin, the highest in the world.
- Overall age-standardized cancer incidence in Australia is the highest in the world, and this is attributable to melanoma alone. Age-standardized overall cancer incidence is similar to New Zealand, but there is a statistically significant difference between Australia and all other parts of the developed world including North America, Western Europe, and the Mediterranean.
United States
In the United States, about 9,000 people die from melanoma a year.[158] In 2011, it affected 19.7 per 100,000, and resulted in death in 2.7 per 100,000.[158]
In 2013:
- 71,943 people in the United States were diagnosed with melanomas of the skin, including 42,430 men and 29,513 women.
- 9,394 people in the United States died from melanomas of the skin, including 6,239 men and 3,155 women.[159]
The American Cancer Society's estimates for melanoma incidence in the United States for 2017 are:
- About 87,110 new melanomas will be diagnosed (about 52,170 in men and 34,940 in women).
- About 9,730 people are expected to die of melanoma (about 6,380 men and 3,350 women).
Melanoma is more than 20 times more common in whites than in African Americans. Overall, the lifetime risk of getting melanoma is about 2.5% (1 in 40) for whites, 0.1% (1 in 1,000) for African Americans, and 0.5% (1 in 200) for Mexicans.
The risk of melanoma increases as people age. The average age of people when the disease is diagnosed is 63.[160]
History
Although melanoma is not a new disease, evidence for its occurrence in antiquity is rather scarce. However, one example lies in a 1960s examination of nine
The French physician René Laennec was the first to describe melanoma as a disease entity. His report was initially presented during a lecture for the Faculté de Médecine de Paris in 1804 and then published as a bulletin in 1806.[163]
The first English-language report of melanoma was presented by an English general practitioner from Stourbridge, William Norris in 1820.[164] In his later work in 1857 he remarked that there is a familial predisposition for development of melanoma (Eight Cases of Melanosis with Pathological and Therapeutical Remarks on That Disease). Norris was also a pioneer in suggesting a link between nevi and melanoma and the possibility of a relationship between melanoma and environmental exposures, by observing that most of his patients had pale complexions.[165] He also described that melanomas could be amelanotic and later showed the metastatic nature of melanoma by observing that they can disseminate to other visceral organs.
The first formal acknowledgment of advanced melanoma as untreatable came from Samuel Cooper in 1840. He stated that the only chance for a cure depends upon the early removal of the disease (i.e., early excision of the malignant mole) ...'[166]
More than one and a half centuries later this situation remains largely unchanged.
Terminology
The word melanoma came to English from 19th-century
Research
Pharmacotherapy research for un-resectable or metastatic malignant melanoma is ongoing.[171]
Targeted therapies
In clinical research, adoptive cell therapy and gene therapy, are being tested.[172]
Two kinds of experimental treatments developed at the National Cancer Institute (NCI), have been used in metastatic melanoma with tentative success.[42]
The first treatment involves adoptive cell therapy (ACT) using TILs immune cells (tumor-infiltrating lymphocytes) isolated from a person's own melanoma tumor.[130] These cells are grown in large numbers in a laboratory and returned to the patient after a treatment that temporarily reduces normal T cells in the patient's body. TIL therapy following lymphodepletion can result in durable complete response in a variety of setups.[173][174]
The second treatment, adoptive transfer of genetically altered autologous lymphocytes, depends on delivering genes that encode so called
A cancer vaccine showed modest benefit in late-stage testing in 2009 against melanoma.[176][177]
BRAF inhibitors
About 60% of melanomas contain a mutation in the B-Raf gene. Early clinical trials suggested that B-Raf inhibitors including Plexxicon's vemurafenib could lead to substantial tumor regression in a majority of patients if their tumor contain the B-Raf mutation.[178] In June 2011, a large clinical trial confirmed the positive findings from those earlier trials.[179][180]
In August 2011, Vemurafenib received FDA approval for the treatment of late-stage melanoma. In May 2013 the US FDA approved dabrafenib as a single agent treatment for patients with BRAF V600E mutation-positive advanced melanoma.[181]
Some researchers believe that combination therapies that simultaneously block multiple pathways may improve efficacy by making it more difficult for the tumor cells to mutate before being destroyed. In October 2012 a study reported that combining Dabrafenib with a MEK inhibitor trametinib led to even better outcomes. Compared to Dabrafenib alone, progression-free survival was increased to 41% from 9%, and the median progression-free survival increased to 9.4 months versus 5.8 months. Some side effects were, however, increased in the combined study.[182][183]
In January 2014, the FDA approved the combination of dabrafenib and trametinib for the treatment of people with BRAF V600E/K-mutant metastatic melanoma.[184] In June 2018, the FDA approved the combination of a BRAF inhibitor encorafenib and a MEK inhibitor binimetinib for the treatment of un-resectable or metastatic melanoma with a BRAF V600E or V600K mutation.[185]
Eventual resistance to BRAF and MEK inhibitors may be due to a cell surface protein known as EphA2 which is now being investigated.[186]
Ipilimumab
At the
Ipilimumab was approved by the FDA in March 2011 to treat patients with late-stage melanoma that has spread or cannot be removed by surgery.[190][191][192]
In June 2011, a clinical trial of ipilimumab plus dacarbazine combined this immune system booster with the standard chemotherapy drug that targets cell division. It showed an increase in median survival for these late stage patients to 11 months instead of the 9 months normally seen. Researchers were also hopeful of improving the five year survival rate, though serious adverse side-effects were seen in some patients. A course of treatment costs $120,000. The drug's brandname is Yervoy.[179][193]
Surveillance methods
Advances in high resolution ultrasound scanning have enabled surveillance of metastatic burden to the sentinel lymph nodes.[194] The Screening and Surveillance of Ultrasound in Melanoma trial (SUNMEL) is evaluating ultrasound as an alternative to invasive surgical methods.[195]
Oncolytic virotherapy
In some countries oncolytic virotherapy methods are studied and used to treat melanoma. Oncolytic virotherapy is a promising branch of virotherapy, where oncolytic viruses are used to treat diseases; viruses can increase metabolism, reduce anti-tumor immunity and disorganize vasculature.[196] Talimogene laherparepvec (T-VEC) (which is a herpes simplex virus type 1–derived oncolytic immunotherapy), was shown to be useful against metastatic melanoma in 2015 with an increased survival of 4.4 months.[197][12]
Antivirals
Antiretrovirals have been tested in vitro against melanoma. The rationale behind this lies in their potential to inhibit human endogenous retroviruses, whose activity has been associated with the development of melanoma.[198][199] The results on malignant cells have shown moderate efficacy, and further developments are awaited through animal model testing.
Notes
References
- ^ a b c d e f g h i j k l m "Melanoma Treatment – for health professionals". National Cancer Institute. 26 June 2015. Archived from the original on 4 July 2015. Retrieved 30 June 2015.
- ^ ISBN 978-92-832-0429-9. Archived(PDF) from the original on 30 May 2014.
- PMID 11310650.
- ^ a b "SEER Stat Fact Sheets: Melanoma of the Skin". NCI. Archived from the original on 6 July 2014.
- ^ PMID 27733282.
- ^ PMID 27733281.
- ^ Schwartzman RM, Orkin M (1962). A Comparative Study of Diseases of Dog and Man. Springfield, IL: Thomas. p. 85.
The term 'melanoma' in human medicine indicates a malignant growth; the prefix 'malignant' is redundant.
- ^ Bobonich M, Nolen ME (2015). Dermatology for Advanced Practice Clinicians. Philadelphia: Wolters Kluwer. p. 106.
The term malignant melanoma is becoming obsolete because the word 'malignant' is redundant as there are no benign melanomas.
- ^ Farlex Partner Medical Dictionary. 2012. Archived from the original on 10 June 2022. Retrieved 4 March 2021.
Avoid the redundant phrase malignant melanoma.
- ^ PMID 22123420.
- ^ PMID 25245960.
- ^ PMID 29208439.
- ^ "USCS Data Visualizations". gis.cdc.gov. Archived from the original on 17 March 2020. Retrieved 7 March 2020.
Need to select "melanoma"
- ^ "CDC - What Are the Symptoms of Skin Cancer?". www.cdc.gov. 26 June 2018. Archived from the original on 7 December 2018. Retrieved 1 February 2019.
- PMID 25741397.
- ^ "The EFG of Nodular Melanomas | MoleMap New Zealand". The EFG of Nodular Melanomas | MoleMap New Zealand. Archived from the original on 2 February 2019. Retrieved 1 February 2019.
- PMID 10862180.
- ^ a b c d e f "Melanoma Risk factors". Mayo Clinic. Archived from the original on 10 April 2017. Retrieved 10 April 2017.
- ^ S2CID 32817426.
- ^ PMID 26601870.
- PMID 26034853.
- PMID 22833605.
- PMID 19655431.
- PMID 25188246.
- S2CID 25209863.
- S2CID 7655216.
- PMID 16326797.
- PMID 7426301.
- S2CID 196354681.
- ^ "WHO | The World Health Organization recommends that no person under 18 should use a sunbed". WHO. Archived from the original on 16 June 2009.
- PMID 1632422.
- ^ "Can we get skin cancer from tanning beds?". CuradermBCC. 1 September 2022. Archived from the original on 3 September 2022. Retrieved 3 September 2022.
- PMID 26892650.
- S2CID 29876528.
- ^ "CDKN2A cyclin-dependent kinase inhibitor 2A (melanoma, p16, inhibits CDK4)". U.S. National Library of Medicine. Archived from the original on 17 November 2004. Retrieved 4 September 2017.
- PMID 26892650.
- PMID 19318491.
- S2CID 9539601.
- PMID 16822996.
- PMID 3312689.
- PMID 20656740.
- ^ PMID 21234353.
- from the original on 25 July 2011.
- PMID 20697348.
- ISBN 0-87969-404-1– via Internet Archive.
- S2CID 45189513.
- PMID 23303275.
- PMID 22622578.
- PMID 20220176.
- PMID 23001126.
- ^ a b "The human pathology proteome in melanoma – The Human Protein Atlas". www.proteinatlas.org. Archived from the original on 2 October 2017. Retrieved 2 October 2017.
- ^ S2CID 206659235.
- PMID 29596782.
- S2CID 23515325.
- PMID 18397884.
- PMID 15010535.
- S2CID 678541.
- PMID 18269766.
- PMID 26978405.
- ^ PMID 26091043.
- PMID 22554099.
- PMID 31618628.
- PMID 30835257.
- PMID 24445538.
- PMID 27477320.
- PMID 26013381.
- ^ PMID 31243305.
- ^ .
- ^ "Prevention: ABCD's of Melanoma". American Melanoma Foundation. Archived from the original on 23 April 2003.
- S2CID 20787489.
- PMID 30521684.
- PMID 30521682.
- PMID 30521681.
- ^ PMID 9828892.
- ^ a b "Introduction to Dermoscopy". DermNet New Zealand. Archived from the original on 7 May 2009.
- PMID 19155361.
- S2CID 2510803.
- S2CID 3431248.
- PMID 16307945.
- PMID 24665419.
- S2CID 34498802.
- ^ OCLC 1040695302. Archived from the originalon 4 August 2020.
- from the original on 8 March 2021. Retrieved 13 February 2020 – via National Center for Biotechnology Information. Last Update: 18 May 2019.
- PMID 26838538.
- S2CID 8312676.
- ^ "Precancerous conditions of the skin". Canadian Cancer Society. Archived from the original on 3 August 2020. Retrieved 26 February 2020.
- S2CID 71464540.
- ^ Hale CS. "Skin melanocytic tumor - Melanoma - Melanoma in situ". pathology Outlines. Archived from the original on 26 February 2020. Retrieved 26 February 2020. Topic Completed: 1 May 2013. Revised: 23 May 2019
- PMID 24281086.
- PMID 17001379.
- PMID 31260100.
- ^ PMID 29028110., citing
Amin MB, Edge SB, Greene FL, et al, eds. AJCC Cancer Staging Manual. 8th ed. New York: Springer International Publishing; 2017:563‐585). - PMID 31157404.
- S2CID 6091309.
- PMID 18273687.
- ^ S2CID 46958616.
- ^ S2CID 206984768.
- PMID 26096748.
- ^ "Can Melanoma Be Prevented?". Archived from the original on 27 June 2006.
- S2CID 29173997.
- S2CID 1477485.
- from the original on 4 July 2018. Retrieved 3 July 2018.
- PMID 17077356.
- ^ "The Sentinel Node Biopsy Procedure in Melanoma does not offer a survival advantage". Malignant Melanoma. 8 January 2008. Archived from the original on 11 July 2012. Retrieved 13 August 2012.
- PMID 25978975.
- PMID 18596673.
- PMID 8373269.
- PMID 19821334.
- ^ ISBN 978-0-7216-3415-9. Archivedfrom the original on 7 January 2016.
- ^ S2CID 23386371..
Cure rate as high as 98% for small melanoma in situ, and as high as 95% noted for lentigo maligna variant of melanona in situ has been reported with Mohs surgery
- ^ "The Screening and Surveillance of Ultrasound in Melanoma trial (SUNMEL)". Archived from the original on 6 January 2009.
- PMID 24005142.
- PMID 23608449.
- ^ ABIM Foundation, American Academy of Dermatology, archived from the originalon 1 December 2013, retrieved 5 December 2013, which cites:
- Bichakjian CK, Halpern AC, Johnson TM, Foote Hood A, Grichnik JM, Swetter SM, et al. (American Academy of Dermatology) (November 2011). "Guidelines of care for the management of primary cutaneous melanoma. American Academy of Dermatology". Journal of the American Academy of Dermatology. 65 (5): 1032–1047. PMID 21868127.
- American Joint Committee on Cancer (2010). Edge SB (ed.). AJCC cancer staging manual (7th ed.). Springer. ISBN 978-0-387-88440-0.
- National Comprehensive Cancer Network (2012), National Comprehensive Cancer Network clinical practice guidelines in oncology (NCCN Guidelines): melanoma (PDF), Fort Washington, Pennsylvania: National Comprehensive Cancer Network, archived from the original (PDF) on 28 December 2013, retrieved 5 December 2013

- Bichakjian CK, Halpern AC, Johnson TM, Foote Hood A, Grichnik JM, Swetter SM, et al. (American Academy of Dermatology) (November 2011). "Guidelines of care for the management of primary cutaneous melanoma. American Academy of Dermatology". Journal of the American Academy of Dermatology. 65 (5): 1032–1047.
- ^ PMID 30190820.
- ^ S2CID 20257709.
- PMID 12380054.
- S2CID 231943836.
- S2CID 24020903.
- from the original on 12 June 2018. Retrieved 11 June 2018.
- .
- PMID 10856105.
- S2CID 33866115.
- ^ PMID 29405038.
- PMID 38158376.
- S2CID 28907006.
- PMID 23990731.
- PMID 29600692.
- PMID 30288368.
- ^ PMID 25520039.
- S2CID 207369519.
- ^ .
- ^ "FDA Approves First Cellular Therapy to Treat Patients with Unresectable or Metastatic Melanoma". U.S. Food and Drug Administration (Press release). 16 February 2024. Retrieved 18 February 2024.
- ^ "Iovance's Amtagvi (lifileucel) Receives U.S. FDA Accelerated Approval for Advanced Melanoma" (Press release). Iovance Biotherapeutics Inc. 16 February 2024. Retrieved 18 February 2024 – via GlobeNewswire.
- ^ PMID 24651672.
- S2CID 5079628.
- ^ PMID 26182315.
- ^ "Interleukin-2 (IL2) for Metastatic Melanoma". Melanoma Research Alliance. Retrieved 5 October 2022.
- PMID 15609471.
- PMID 25838374.
- PMID 24912947.
- ^ "U.S. Food and Drug Administration Approves First LAG-3-Blocking Antibody Combination, Opdualag (nivolumab and relatlimab-rmbw), as Treatment for Patients with Unresectable or Metastatic Melanoma" (Press release). Bristol Myers Squibb. 18 March 2022. Archived from the original on 19 March 2022. Retrieved 19 March 2022 – via Business Wire.
- ^ PMID 25526608.
- PMID 9366823.
- S2CID 18082594.
- PMID 15707701.
- ^ Ford MB, Mitchell MF, Boyer KL, Ford MB, Judkins AF, Levin B (1999). "Cancer Epidemiology". Primary Care Oncology. pp. 1–27.
- PMID 16258493.
- PMID 11504745. Archived from the originalon 5 March 2006. Retrieved 31 July 2006.
- PMID 25440435.
- S2CID 44155588.
- PMID 30066226.
- PMID 31974724.
- ^ "CANCERMondial (GLOBOCAN)". GLOBOCAN. 2010. Archived from the original on 17 February 2012. Retrieved 12 August 2010.
- PMID 16368510.
- ^ "Cancer in Australia: an overview 2012". AIHW. Archived from the original on 2 June 2014. Retrieved 1 June 2014.
- ^ U.S. Cancer Statistics Working Group. U.S. Cancer Statistics Data Visualizations Tool, based on 2020 submission data (1999-2018): U.S. Department of Health and Human Services, Centers for Disease Control and Prevention and National Cancer Institute; www.cdc.gov/cancer/dataviz, released in June 2021.
- ^ from the original on 31 May 2017.
- ^ "CDC – Skin Cancer Statistics". www.cdc.gov. Archived from the original on 10 April 2017. Retrieved 10 April 2017.
- ^ "Key Statistics for Melanoma Skin Cancer". www.cancer.org. Archived from the original on 10 April 2017. Retrieved 10 April 2017.
- PMID 5326247.
- PMID 5698493.
- ^ Laennec RT (1806). "Sur les melanoses". Bulletin de la Faculté de Médecine de Paris. 1: 24–26.
- PMID 30332089.
- ^ Norris W. Eight cases of Melanosis with pathological and therapeutical remarks on that disease. London: Longman; 1857.
- ^ Cooper S (1844). The First Lines of the Theory and Practice of Surgery: Including the Principle Operations. S.S. and W. Wood. Archived from the original on 4 August 2021. Retrieved 14 December 2017.
- ^ Merriam-Webster, Merriam-Webster's Collegiate Dictionary, Merriam-Webster, archived from the original on 10 October 2020, retrieved 20 July 2018
- ^ Liddell HG, Scott R. "μέλας". A Greek-English Lexicon. Perseus. Archived from the original on 5 June 2011.
- ^ "Dorland's Illustrated Medical Dictionary". Elsevier. Archived from the original on 11 January 2014. Retrieved 20 July 2018.
- ^ "The American Heritage Dictionary of the English Language". Houghton Mifflin Harcourt. Archived from the original on 25 September 2015. Retrieved 20 July 2018.
- S2CID 13247048. Archived from the originalon 1 January 2013. Retrieved 12 June 2012.
- PMID 11907465.
- PMID 18809613.
- S2CID 32568. Retrieved 15 August 2011.
- ^ "New Method of Gene Therapy Alters Immune Cells for Treatment of Advanced Melanoma; Technique May Also Apply to Other Common Cancers". 30 December 2015. Archived from the original on 28 September 2006.
- ^ "Immune System Taught To Fight Melanoma". CBSNews. 30 May 2009. Archived from the original on 29 October 2010.
- PMID 21631324.
- ^ Harmon A (21 February 2010). "A Roller Coaster Chase for a Cure". The New York Times. Archived from the original on 10 February 2017.
- ^ a b Pollack A (5 June 2011). "Drugs Show Promise Slowing Advanced Melanoma". The New York Times. Archived from the original on 31 January 2017.
- PMID 21639808.
- ^ "GSK melanoma drugs add to tally of U.S. drug approvals". Reuters. 30 May 2013. Archived from the original on 24 September 2015.
- ^ "Combination of dabrafenib and trametinib delays development of treatment resistance in MM patients". News Medical. 1 October 2012. Archived from the original on 14 May 2013.
- PMID 23020132.
- ^ "Dabrafenib/Trametinib Combination Approved for Advanced Melanoma". OncLive. 9 January 2014. Archived from the original on 25 January 2014. Retrieved 4 February 2015.
- S2CID 52020890.
- ^ "Counteracting Drug Resistance in Melanoma". 2015. Archived from the original on 4 February 2015.
- ^ "Bristol drug cuts death risk in advanced melanoma". Reuters. 5 June 2010. Archived from the original on 9 November 2010.
- ^ "The Risk For Bristol". Forbes. Archived from the original on 15 March 2011.
- ^ "Phase 3 clinical study: Ipilimumab boosts, sustains immune system responses against melanoma tumors". News-medical.net. 9 June 2010. Archived from the original on 19 October 2012. Retrieved 13 August 2012.
- U.S. Food and Drug Administration (FDA). Archivedfrom the original on 27 March 2011. Retrieved 25 March 2011.
- ^ Pollack A (25 March 2011). "Approval for Drug That Treats Melanoma". The New York Times. Archived from the original on 1 April 2011. Retrieved 27 March 2011.
- ^ "Yervoy". Drugs.com. Archived from the original on 9 August 2011.
- PMID 21639810.
- PMID 20065175.
- ^ "malignant-melanoma.org". Archived from the original on 14 October 2011.
- PMID 24062768.
- S2CID 23663167.
- PMID 37834078.
- PMID 35163254.
External links
- Melanoma at Curlie