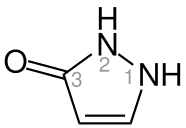Pyrazolone
Pyrazolone is 5-membered
Structure and synthesis
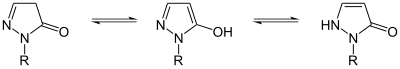
Pyrazolone can exist in two isomers: 3-pyrazolone and 4-pyrazolone.
These isomers can interconvert via lactam–lactim and imine–enamine
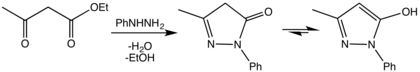
The first synthesis of pyrazolones was reported in 1883 by Ludwig Knorr, via a condensation reaction between ethyl acetoacetate and phenylhydrazine.[2]
Many pyrazolones are produced by functionalization of preformed pyrazolones.
Applications
Pharmaceuticals
This section is missing information about safety profile of analgesics (which ones are withdrawn and whether there is a common theme in adverse effects). (August 2022) |
Pyrazolones are amongst the oldest synthetic pharmaceuticals, starting with the introduction of
The basic structure has been also used in a number of newer drugs of other effects.[5] Edaravone is useful for prevention and/or therapy of arterial wall injury.[6] Eltrombopag is used to address low blood platelet count.
Dyes
Pyrazolone groups are present in several important dyes. They are commonly used in combination with
Ligands
Pyrazolones have been studied as ligands.[7]
References
- ^ ISBN 0471238961.
- .
- ^ S2CID 2761563.
- .
- ^ PMID 31761383.
- S2CID 214766793.
- .
External links
- Pyrazolones at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Pubchem - 3-Pyrazolone
- Pubchem - 4-Pyrazolone