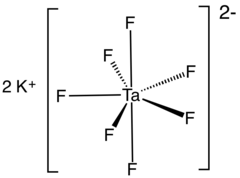
Potassium heptafluorotantalate
 | |
| Names | |
|---|---|
| IUPAC name
Dipotassium heptafluorotantalate
| |
| Systematic IUPAC name
Dipotassium heptafluorotantalum(2-) | |
| Other names
Potassium heptafluorotantalate(V)
Potassium fluorotantalate | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.037.245 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
SMILES
| |
| Properties | |
| K2[TaF7] | |
| Molar mass | 392.13 g/mol |
| Appearance | white solid |
| Density | 4.56 g/mL at 25 °C |
| Melting point | 630 to 820 °C (1,166 to 1,508 °F; 903 to 1,093 K) |
0.5 g/100 mL (15 °C)[1]
| |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H301, H315, H319, H331, H335 | |
| P261, P264, P270, P271, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P311, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
110 mg/kg (Oral: rat) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Potassium heptafluorotantalate is an inorganic compound with the formula K2[TaF7]. It is the potassium salt of the heptafluorotantalate anion [TaF7]2−. This white, water-soluble solid is an intermediate in the purification of tantalum from its ores and is the precursor to the metal.[2]
Preparation
Industrial
Potassium heptafluorotantalate is an intermediate in the industrial production of metallic tantalum. Its production involves leaching tantalum ores, such as columbite and tantalite, with hydrofluoric acid and sulfuric acid to produce the water-soluble hydrogen heptafluorotantalate.[2]
- Ta2O5 + 14 HF → 2 H2[TaF7] + 5 H2O
This solution is subjected to a number of liquid-liquid extraction steps to remove metallic impurities (most importantly niobium) before being treated with potassium fluoride to produce K2[TaF7]
Lab-scale
Hydrofluoric acid is both corrosive and toxic, making it unappealing to work with; as such a number of alternative processes have been developed for small-scale syntheses. Potassium heptafluorotantalate can be produced by both anhydrous and wet methods. The anhydrous method involves the reaction of tantalum oxide with potassium bifluoride or ammonium bifluoride according to the following equation:[1][3]
- Ta2O5 + 4 KHF2 + 6 HF → 2 K2[TaF7] + 5 H2O
The method was originally reported by
K2[TaF7] can also be precipitated from solutions in hydrofluoric acid provided that the concentration of HF is below about 42%. Solutions having higher concentrations of HF yield potassium hexafluorotantalate [KTaF6]. The K-salt can be also precipitated from a solution in
- 5 HF + 2 KF + TaCl5 → K2[TaF7] + 5 HCl
Structure
Potassium heptafluorotantalate exists in at least two
At temperatures above 230 °C this converts to β-K2[TaF7], which is
Reactions
K2[TaF7] is primarily used to produce metallic tantalum by
- K2[TaF7] + 5 Na → Ta + 5 NaF + 2 KF
K2[TaF7] is susceptible to hydrolysis. For example, a boiling aqueous solution of K2[TaF7] yields potassium oxyfluorotantalate (K2Ta2O3F6), known as “Marignac’s salt”. In order to prevent hydrolysis and co-precipitation of potassium oxyfluorotantalate, a small excess of HF is added to the solution.
References
- ^ a b W. Kwasnik (1963). "Potassium Heptafluorotantalate (V)". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry, 2nd Ed. Vol. 2pages=256. NY, NY: Academic Press.
- ^ ISBN 9780080529028.
- ^ J. J. Berzelius Pogg. Ann. 4, 6 (1825#.
- .
- .
- .
