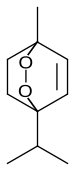
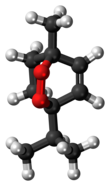
Ascaridole
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
1-Methyl-4-(1-methylethyl)-2,3-dioxabicyclo[2.2.2]oct-5-ene
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 121382 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.007.408 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties[1] | |||
| C10H16O2 | |||
| Molar mass | 168.23 g/mol | ||
| Appearance | Colorless liquid | ||
| Density | 1.010 g/cm3 | ||
| Melting point | 3.3 °C (37.9 °F; 276.4 K) | ||
| Boiling point | 40 °C (104 °F; 313 K) at 0.2 mmHg | ||
| Hazards | |||
| GHS labelling: | |||
 
| |||
| Danger | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Ascaridole is a natural
History
Ascaridole was the first, and for a long time only, discovered naturally occurring
The first laboratory synthesis was demonstrated in 1944 by Günther Schenck and Karl Ziegler and might be regarded as mimicking the natural production of ascaridole. The process starts from α-terpinene which reacts with oxygen under the influence of chlorophyll and light. Under these conditions singlet oxygen is generated which reacts in a Diels–Alder reaction with the diene system in the terpinene.[8][9][10] Since 1945, this reaction has been adopted into the industry for large-scale production of ascaridole in Germany. It was then used as an inexpensive drug against intestinal worms.[11]
Properties
Ascaridole is a colorless liquid that is soluble in most organic solvents. It is toxic and has a pungent, unpleasant smell and taste. Like other pure, low molecular weight
Occurrence

The specific flavor of the Chilean tree
Applications
Ascaridole is mainly used as an anthelmintic drug that expels parasitic worms (helminths) from the human body and plants. This property gave the name to the chemical, after Ascaris – a genus of the large intestinal roundworm. In the early 1900s, it was a major remedy against intestinal parasites in humans, cats, dogs, goats, sheep, chickens, horses, and pigs, and it is still used in livestock, particularly in the Central American countries. The dosage was specified by the ascaridole content in the oil, which was traditionally determined with an assay developed by Nelson in 1920. It was later substituted with modern gas chromatography and mass spectrometry methods.[19] The worms and their larvae were killed by immersion in a solution of ascaridole in water (about 0.015 vol%) for 18 hours at 50 °F (10 °C) or 12 hours at 60 °F (16 °C) or 6 hours at 65 to 70 °F (18 to 21 °C). Meanwhile, such immersion did not damage the roots and stems of plants such as Iris, Phlox, Sedum and others at 70 °F (21 °C) for 15 hours or longer.[12]
The wormseed plant itself (Mexican tea) is traditionally used in
Health issues
The usage of wormseed oil on humans is limited by the toxicity of ascaridole and has therefore been discouraged. In high doses, wormseed oil causes
References
- ^ ISBN 978-0-470-18024-2.
- ^ Hüthig (April 1908). "Commercial notes and scientific information on essential oils". Semi-annual Report of Schimmel & Co.: 12–120. Ascaridole is discussed in the section titled "Wormseed oil, American", pp. 109–119. Ascaridole is named on p. 111; its empirical formula is stated on p. 114; the "explosive character" of the decomposition on heating is mentioned on p. 115. Some of the text can be seen about nine minutes into this video.
- ^ (Hüthig, April 1908), p. 116.
- S2CID 250895582.
- . See p. 1412.
- ^ .
- .
- ^ a b Nelson, E. K. (1947). "Chapter 5: Oxides: Ascaridole". The Terpenes. Vol. 2. CUP Archive. pp. 446–452.
- S2CID 24081588.
- S2CID 8655604.
- ISBN 978-0-495-38857-9.
- ^ a b US Department of Agriculture (1972). "Technical Bulletin". Technical Bulletin. 1441: 65.
- )
- ISBN 978-9968-31-235-6.
- ISBN 978-970-722-113-0.
- .
- ^ ISBN 978-0-444-82590-2.
- ^ ISBN 978-0-660-19073-0.
- ^ a b "Chenopodium ambrosioides". Medicinal Plants for Livestock. Ithaca, NY: Cornell University, Department of Animal Science. Archived from the original on 2006-02-21.