Hinokitiol
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Hydroxy-6-(propan-2-yl)cyclohepta-2,4,6-trien-1-one | |||
| Other names
β-Thujaplicin; 4-Isopropyltropolone
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.007.165 | ||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C10H12O2 | |||
| Molar mass | 164.204 g·mol−1 | ||
| Appearance | Colorless to pale yellow crystals | ||
| Melting point | 50 to 52 °C (122 to 126 °F; 323 to 325 K) | ||
| Boiling point | 140 °C (284 °F; 413 K) at 10 mmHg | ||
| 1.2 g/L (0 °C) | |||
| Solubility in ethanol | 20 g/L[2] | ||
| Solubility in dimethyl sulfoxide | 30 g/L[2] | ||
| Solubility in dimethylformamide | 12.5 g/L[2] | ||
| Hazards | |||
| Flash point | 140 °C (284 °F; 413 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hinokitiol (β-thujaplicin) is a natural
History
Hinokitiol was discovered by a Japanese chemist
Woods that are rich in hinokitiol were used by people of ancient
Natural occurrence
Hinokitiol has been found in the heartwood of the conifer trees of the
Its concentration in the trees are 0.1-0.2% in
There are three naturally found thujaplicins: α-thujaplicin, β-thujaplicin (hinokitiol) and γ-thujaplicin. Hinokitiol is the most common isomer and it appears to be the only isomer that exerts all biological activities attributed to thujaplicins.[19][20]
Chemical synthesis
There are different pathways to synthesize thujaplicins. Hinokitiol, as other thujaplicins, can be synthesized by
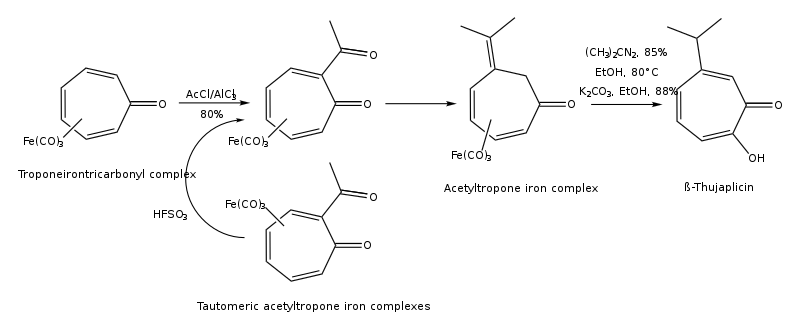
(1) Synthesis of hinokitiol from troponeirontricarbonyl complex:
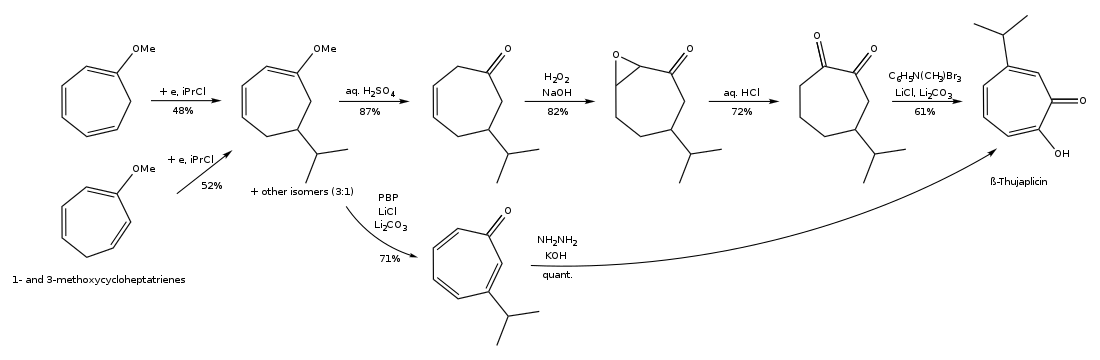
(2) Synthesis of hinokitiol by electro-reductive alkylation of substituted cycloheptatrienes:
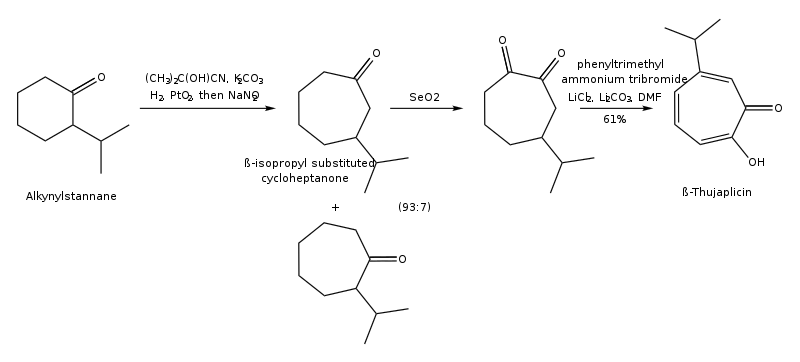
(3) Synthesis of hinokitiol through ring expansion of 2-isopropylcyclohexanone:
(4) Synthesis of hinokitiol through oxyallyl cation [4+3] cyclization (Noyori's synthesis):

Chemistry
Hinokitiol is a tropolone derivative containing an unsaturated seven-membered carbon ring. It is a monoterpenoid – cyclohepta-2,4,6-trien-1-one substituted by a hydroxy group at position 2 and an isopropyl group at position 4.[26][27][28] It is a enol and a cyclic ketone. It derives from a hydride of a cyclohepta-1,3,5-triene. Thujaplicins are soluble in organic solvents and aqueous buffers.[2] Hinokitiol provides acetone on vigorous oxidation and gives the saturated monocyclic diol upon catalytic hydrogenation.[7] It is stable to alkali and acids, forming salts or remaining unchanged, but does not convert to catechol derivatives. Hinokitiol, as other thujaplicins and tropolones, reversibly binds metal ions. It forms complex salts with metal ions.
Ionophore
Hinokitiol, as other tropolones, reversibly binds metal ions (i.e. Zn2+, Fe2+, Cu2+, Co2+, Mn2+, Ag2+) and form complex salts. It is considered as a broad-spectrum metallophore, and an efficient iron-chelating agent.[17] The iron complex with hinokitiol with the formula (C10H11O2)3Fe is called hinokitin. Hinoki oil is rich in hinokitin which has an appearance of dark red crystals.[7] The complexes made of iron and tropolones display high thermodynamic stability and has shown to have a stronger binding constant than the transferrin-iron complex.[29] It is believed that metal-binding activity may be the principal mechanism of action underlying the most part of its biological activities, especially binding iron, zinc, and copper ions.[20] By binding different metal ions and serving as an ionophore, it accelerates the intracellular uptake of those ions and increases their intracellular levels, thus influencing on different biological activities. It is shown that a synergistic effect in some biological activities and settings may occur when ionophores are combined with the ions they bind.[30] As an ionophore, its molecule has an hydrophilic center and a hydrophobic part. The hydrophobic part interacts with biological membranes. The hydrophilic center binds metal ions and form ionophore-ion complexes.
Biological properties
Hinokitiol and other thujaplicins have been mainly investigated in in-vitro studies and animal models for their possible biological properties, such as antimicrobial, antifungal, antiviral, antiproliferative, anti-inflammatory, antiplasmodial effects.[10][17][20] However, no evidence exists from clinical studies to support these findings. It has also shown to have insecticidal, pesticidal and antibrowning effects. The vast majority of these properties are thought to be due to the metal ion-binding activity. Hinokitiol appeared to exert all in-vitro activities attributed to thujaplicins.[20]
Hinokitiol has been shown to possess inhibitory effects on Chlamydia trachomatis and may be clinically useful as a topical drug.[31][10]
Safety
The safety of hinokitiol has been tested in rats and no carcinogenic effect to rats was found.[32] In 2006, hinokitiol was categorized under the Domestic substances list (DSL) in Canada as non-persistent, non-bioaccumulative and non-toxic to aquatic organisms.[33]
Uses
Skin and oral care products
Hinokitiol is used in a range of consumer products intended for skin care, such as
In April 2020, Advance Nanotek, an Australian producer of zinc oxide, filed a joint patent application with AstiVita Limited, for an anti-viral composition that included oral care products.[37]
Insect repellent
Hinokitiol is found to have
Food preservative
In experimental studies hinokitiol has been shown to act against
Wood preservative
Hinokitiol is one of the chemical compounds isolated from trees, known as
Research directions
Iron transport
Researchers screening a library of small biomolecules for signs of iron transport found that hinokitiol restored cell functionality. Further work by the team suggested a mechanism by which hinokitiol restores or reduces cell iron.[43]
Cancer research
Different in-vitro studies have investigated the effects of hinokitiol on various tumor cell lines.
See also
- Thujaplicins
- Tropolone
- Ionophore
- Cupressaceae
References
- ^ β-Thujaplicin Archived 2011-07-16 at the Wayback Machine at Sigma-Aldrich
- ^ a b c d "Hinokitiol - Product Information" (PDF). www.caymanchem.com. Cayman Chemical.
- PMID 19483786.
- ^ a b c "Hinokitiol | 499-44-5". www.chemicalbook.com.
- ^ .
- ^ a b "The Japan Food chemical Research Faundation". www.ffcr.or.jp.
- ^ .
- PMID 23737463.
- ISBN 9780521030854.
- ^ a b c d "Hinokitiol". American Chemical Society.
- ISBN 1-84246-068-4.
- PMID 16789546.
- ^ .
- ^ Okabe, T; Saito, K (1994). "Antibacterial and preservative effects of natural Hinokitiol (beta-Thujaplicin) extracted from wood". Acta Agriculturae Zhejiangensis. 6 (4): 257–266.
- ^ PMID 15187442.
- PMID 31766678.
- ^ .
- ^ PMID 25078835.
- ^ PMID 18250899.
- ^ a b c d Falcone, Eric (5 October 2016). "Investigating the Antiproliferative Activity of Synthetic Troponoids". Doctoral Dissertations.
- .
- PMID 25400298.
- S2CID 25767209.
- S2CID 8355694.
- S2CID 95994935.
- ^ "2,4,6-Cycloheptatrien-1-one, 2-hydroxy-3-(1-methylethyl)-". pubchem.ncbi.nlm.nih.gov. PubChem.
- ^ "Hinokitiol". pubchem.ncbi.nlm.nih.gov. PubChem.
- ^ "gamma-Thujaplicin". pubchem.ncbi.nlm.nih.gov. PubChem.
- S2CID 43256591.
- PMID 19859983.
- ISBN 978-3-8383-4661-8.
- PMID 17077589.
- ^ Secretariat, Treasury Board of Canada. "Detailed categorization results of the Domestic Substances List - Open Government Portal". open.canada.ca. Retrieved 2020-06-17.
- S2CID 11746050.
- S2CID 12971210.
- PMID 31825092.
- ^ "IP Australia: AusPat". Australian Government - IP Australia. Retrieved 2020-05-20.
- PMID 10963310.
- S2CID 208583176.
- PMID 17950018.
- ISBN 9780367397289.
- S2CID 16934998.
- PMID 28495746.
External links
- Hinokitiol at PubChem
- β-Thujaplicin at Sigma-Aldrich
- Hinokitiol at ChemicalBook


