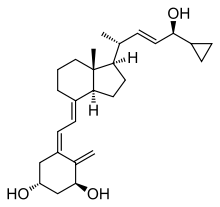
Calcipotriol
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Daivonex, Dovonex, Sorilux |
| Other names | calcipotriene (USAN US) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608018 |
| License data | |
| Pregnancy category |
|
Topical administration | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 5 to 6% |
| Metabolism | Liver |
| Excretion | Biliary |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Calcipotriol, also known as calcipotriene, is a synthetic derivative of calcitriol, a form of vitamin D. It is used in the treatment of psoriasis.[1] It is safe for long-term application in psoriatic skin conditions.[medical citation needed]
It was patented in 1985 and approved for medical use in 1991.[2] It is marketed under the trade name "Dovonex" in the United States, "Daivonex" outside North America, and "Psorcutan" in Germany.[citation needed]
It is on the World Health Organization's List of Essential Medicines.[3]
Calcipotriol is also available as
Medical uses
Chronic plaque psoriasis is the chief medical use of calcipotriol.[5] It has also been used successfully in the treatment of alopecia areata.[6]
Contraindications
Hypersensitivity, use on face, hypercalcaemia, or evidence of vitamin D toxicity are the only
Cautions include exposure to excessive natural or artificial light, due to the potential for calcipotriol to cause photosensitivity.[7]
Adverse effects
Adverse effects by frequency:[5][7][8][9]
- Very common (> 10% frequency)
- Burning
- Itchiness
- Skin irritation
- Common (1–10% frequency)
- Dermatitis
- Dry skin
- Erythema
- Peeling
- Worsening of psoriasis including facial/scalp
- Rash
- Uncommon (0.1–1% frequency)
- Exacerbation of psoriasis
- Rare (< 0.1% frequency)
- Allergic contact dermatitis
- Hypercalcaemia
- Photosensitivity
- Changes in pigmentation
- Skin atrophy
Interactions
No drug interactions are known.[7]
Pharmacology
Mechanism of action
The efficacy of calcipotriol in the treatment of psoriasis was first noticed by the observation of patients receiving various forms of vitamin D in an osteoporosis study. Unexpectedly, some patients who also had psoriasis experienced dramatic reductions in lesion counts.[10]
The precise mechanism of calcipotriol in remitting psoriasis is not well understood. However, it has been shown to have comparable affinity with calcitriol for the vitamin D receptor (VDR), while being less than 1% as active as the calcitriol in regulating calcium metabolism. The vitamin D receptor belongs to the steroid/thyroid receptor superfamily, and is found on the cells of many different tissues including the thyroid, bone, kidney, and T cells of the immune system. T cells are known to play a role in psoriasis, and it is thought that the binding of calcipotriol to the VDR modulates the T cells gene transcription of cell differentiation and proliferation related genes.
In mouse studies, topical calcipotriol administration to the ear and dorsal skin led to a dose-dependent increase in the production of the epithelial cell-derived cytokine
Pharmacokinetics
After application and systemic uptake, calcipotriol undergoes rapid
The metabolites of calcipotriol are less potent than the parent compound.
Chemistry
Calcipotriol is a white to almost white crystalline compound.
References
- ISBN 9780198729426.
- ISBN 9783527607495.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "Taclonex- calcipotriene and betamethasone dipropionate ointment". DailyMed. 21 May 2020. Retrieved 19 October 2020.
- ^ ISBN 978-0-9805790-9-3.
- PMID 22879719.
- ^ a b c d "Dovonex, Calcitrene Ointment (calcipotriene) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved 26 January 2014.
- ^ "CALCIPOTRIENE (calcipotriene) solution [E. FOUGERA & CO. A division of Fougera Pharmaceuticals Inc.]". DailyMed. E. FOUGERA & CO. A division of Fougera Pharmaceuticals Inc. May 2012. Retrieved 26 January 2014.
- ^ "PRODUCT INFORMATION DAIVONEX® CREAM Calcipotriol 50 microgram/g" (PDF). TGA eBusiness Services. LEO Pharma Pty Ltd. 28 April 2011. Retrieved 26 January 2014.
- PMID 4069059.
- PMID 16880407.
- PMID 23364144.
- ^ "Enstilar (calcipotriene and betamethasone dipropionate) Foam, 0.005%/0.064% for topical use. Full Prescribing Information" (PDF). Parsippany, NJ: LEO Pharma Inc. 2015. Archived from the original (PDF) on 2018-09-20. Retrieved 2015-11-21.
External links
- "Calcipotriene". Drug Information Portal. U.S. National Library of Medicine.
