GLUT4
| glucose transporter, type 4 | ||||||
|---|---|---|---|---|---|---|
| Identifiers | ||||||
Ensembl |
|
| ||||
| UniProt |
| |||||
| RefSeq (mRNA) |
|
| ||||
| RefSeq (protein) |
|
| ||||
| Location (UCSC) | n/a | n/a | ||||
| PubMed search | n/a | n/a | ||||
| View/Edit Human | |||
Glucose transporter type 4 (GLUT4), also known as solute carrier family 2, facilitated glucose transporter member 4, is a
At the cell surface, GLUT4 permits the facilitated diffusion of circulating glucose down its concentration gradient into muscle and fat cells. Once within cells, glucose is rapidly
Structure

Like all proteins, the unique amino acid arrangement in the
Other GLUT proteins
There are 14 total GLUT proteins separated into 3 classes based on sequence similarities. Class 1 consists of GLUT 1-4 and 14, class 2 contains GLUT 5, 7, 9 and 11, and class 3 has GLUT 6, 8, 10, 12 and 13.
Although there are some sequence differences between all GLUT proteins, they all have some basic structural components. For example, both the N and C termini in GLUT proteins are exposed to the cytoplasm of the cell, and they all have 12 transmembrane segments.[8]
Tissue distribution
Skeletal muscle
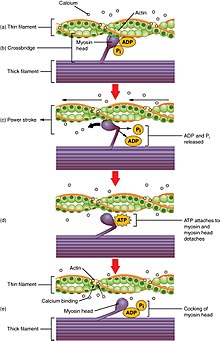
In striated skeletal muscle cells, GLUT4 concentration in the plasma membrane can increase as a result of either exercise or muscle contraction.
During exercise, the body needs to convert glucose to ATP to be used as energy. As G-6-P concentrations decrease, hexokinase becomes less inhibited, and the glycolytic and oxidative pathways that make ATP are able to proceed. This also means that muscle cells are able to take in more glucose as its intracellular concentrations decrease. In order to increase glucose levels in the cell, GLUT4 is the primary transporter used in this facilitated diffusion.[10]
Although muscle contractions function in a similar way and also induce the translocation of GLUT4 into the plasma membrane, the two skeletal muscle processes obtain different forms of intracellular GLUT4. The GLUT4 carrier vesicles are either transferrin positive or negative, and are recruited by different stimuli. Transferrin-positive GLUT4 vesicles are utilized during muscle contraction while the transferrin-negative vesicles are activated by insulin stimulation as well as by exercise.[11][12]
Cardiac muscle
Cardiac muscle is slightly different from skeletal muscle. At rest, they prefer to utilize fatty acids as their main energy source. As activity increases and it begins to pump faster, the cardiac muscles begin to oxidize glucose at a higher rate.[13]
An analysis of mRNA levels of GLUT1 and GLUT4 in cardiac muscles show that GLUT1 plays a larger role in cardiac muscles than it does in skeletal muscles.[14] GLUT4, however, is still believed to be the primary transporter for glucose.[15]
Much like in other tissues, GLUT4 also responds to insulin signaling, and is transported into the plasma membrane to facilitate the diffusion of glucose into the cell. [16][17]
Adipose tissue
Adipose tissue, commonly known as fat,[18] is a depository for energy in order to conserve metabolic homeostasis. As the body takes in energy in the form of glucose, some is expended, and the rest is stored as glycogen (primarily in the liver, muscle cells), or as triglyceride in adipose tissue.[19]
An imbalance in glucose intake and energy expenditure has been shown to lead to both adipose cell hypertrophy and hyperplasia, which lead to obesity.[20] In addition, mutations in GLUT4 genes in adipocytes can also lead to increased GLUT4 expression in adipose cells, which allows for increased glucose uptake and therefore more fat stored. If GLUT4 is over-expressed, it can actually alter nutrient distribution and send excess glucose into adipose tissue, leading to increased adipose tissue mass.[20]
Regulation
Insulin
Insulin is released from the pancreas and into the bloodstream in response to increased glucose concentration in the blood.
Under conditions of low insulin, most GLUT4 is sequestered in intracellular vesicles in muscle and fat cells. As the vesicles fuse with the plasma membrane, GLUT4 transporters are inserted and become available for transporting glucose, and glucose absorption increases.[23] The genetically engineered muscle insulin receptor knock‐out (MIRKO) mouse was designed to be insensitive to glucose uptake caused by insulin, meaning that GLUT4 is absent. Mice with diabetes or fasting hyperglycemia, however, were found to be immune to the negative effects of the insensitivity.[24]

The mechanism for GLUT4 is an example of a
Knockout mice that are heterozygous for GLUT4 develop insulin resistance in their muscles as well as diabetes.[29]
Muscle contraction
Muscle contraction stimulates muscle cells to translocate GLUT4 receptors to their surfaces. This is especially true in cardiac muscle, where continuous contraction increases the rate of GLUT4 translocation; but is observed to a lesser extent in increased skeletal muscle contraction.[30] In skeletal muscle, muscle contractions increase GLUT4 translocation severalfold,[31] and this is likely regulated by RAC1[32][33] and AMP-activated protein kinase.[34]
Muscle stretching
Muscle stretching also stimulates GLUT4 translocation and glucose uptake in rodent muscle via RAC1.[35]
Interactions
GLUT4 has been shown to interact with
In addition, recent reports demonstrated the presence of GLUT4 gene in central nervous system such as the hippocampus. Moreover, impairment in insulin-stimulated trafficking of GLUT4 in the hippocampus result in decreased metabolic activities and plasticity of hippocampal neurons, which leads to depressive like behaviour and cognitive dysfunction.[37][38][39]
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "GlycolysisGluconeogenesis_WP534".
References
- S2CID 4237493.
- S2CID 4285627.
- S2CID 20359706.
- PMID 2568955.
- PMID 15082519.
- ^ PMID 11243799.
- PMID 17403369.
- PMID 23506862.
- ISBN 978-0-7167-3706-3.
- PMID 23899560.
- PMID 9744875.
- PMID 23072821.
- S2CID 32458568.
- PMID 9321820.
- S2CID 7766813.
- PMID 1881917.
- S2CID 171571.
- ^ "Adipose tissue". ScienceDaily. Retrieved 2017-05-24.
- ^ PMID 25299671.
- ^ PMID 8226728.
- ^ "Insulin Synthesis and Secretion". www.vivo.colostate.edu. Retrieved 2017-05-24.
- PMID 22974359.
- PMID 6989818.
- PMID 10927996.
- S2CID 39756994.
- PMID 17259384.
- PMID 24216610.
- ^ PMID 23423567.
- S2CID 8643507.
- PMID 7597034.
- PMID 25353002.
- PMID 25239922.
- PMID 23274900.
- PMID 11389854.
- PMID 25416624.
- PMID 11842083.
- S2CID 10955351.
- S2CID 38081413.
- PMID 20438663.
External links
- GLUT4+Protein at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- USCD—Nature molecule pages: The signaling pathway", "GLUT4"; contains a high-resolution network map. Accessed 25 December 2009.

