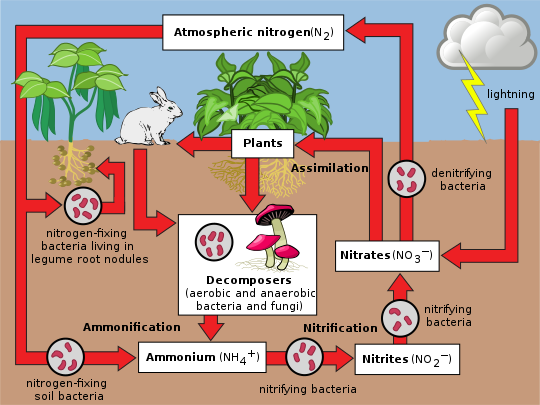
Nitrogen cycle

The nitrogen cycle is the
The nitrogen cycle is of particular interest to
Processes
| Part of a series on |
| Biogeochemical cycles |
|---|
 |
Nitrogen is present in the environment in a wide variety of chemical forms including organic nitrogen, ammonium (NH+4), nitrite (NO−2), nitrate (NO−3), nitrous oxide (N2O), nitric oxide (NO) or inorganic nitrogen gas (N2). Organic nitrogen may be in the form of a living organism, humus or in the intermediate products of organic matter decomposition. The processes in the nitrogen cycle is to transform nitrogen from one form to another. Many of those processes are carried out by microbes, either in their effort to harvest energy or to accumulate nitrogen in a form needed for their growth. For example, the nitrogenous wastes in animal urine are broken down by nitrifying bacteria in the soil to be used by plants. The diagram alongside shows how these processes fit together to form the nitrogen cycle.
Nitrogen fixation
The conversion of nitrogen gas (N2) into nitrates and nitrites through atmospheric, industrial and biological processes is called nitrogen fixation. Atmospheric nitrogen must be processed, or "
Assimilation
Plants can absorb nitrate or ammonium from the soil by their root hairs. If nitrate is absorbed, it is first reduced to nitrite ions and then ammonium ions for incorporation into amino acids, nucleic acids, and chlorophyll. In plants that have a symbiotic relationship with rhizobia, some nitrogen is assimilated in the form of ammonium ions directly from the nodules. It is now known that there is a more complex cycling of amino acids between Rhizobia bacteroids and plants. The plant provides amino acids to the bacteroids so ammonia assimilation is not required and the bacteroids pass amino acids (with the newly fixed nitrogen) back to the plant, thus forming an interdependent relationship.
Ammonification
When a plant or animal dies or an animal expels waste, the initial form of nitrogen is
- GS: Gln Synthetase (cytosolic & plastic)
- GOGAT: Glu 2-oxoglutarate aminotransferase (Ferredoxin & NADH-dependent)
- GDH: Glu Dehydrogenase:
- Minor role in ammonium assimilation.
- Important in amino acid catabolism.

Nitrification
The conversion of ammonium to nitrate is performed primarily by soil-living bacteria and other nitrifying bacteria. In the primary stage of nitrification, the oxidation of ammonium (NH+4) is performed by bacteria such as the
Due to their very high
Denitrification
Denitrification is the reduction of nitrates back into nitrogen gas (N
2), completing the nitrogen cycle. This process is performed by bacterial species such as Pseudomonas and Paracoccus, under anaerobic conditions. They use the nitrate as an electron acceptor in the place of oxygen during respiration. These facultatively (meaning optionally) anaerobic bacteria can also live in aerobic conditions. Denitrification happens in anaerobic conditions e.g. waterlogged soils. The denitrifying bacteria use nitrates in the soil to carry out respiration and consequently produce nitrogen gas, which is inert and unavailable to plants. Denitrification occurs in free-living microorganisms as well as obligate symbionts of anaerobic ciliates.[29]
-
Classical representation of nitrogen cycle
-
Flow of nitrogen through the ecosystem. Bacteria are a key element in the cycle, providing different forms of nitrogen compounds able to be assimilated by higher organisms
-
Simple representation of the nitrogen cycle. Blue represent nitrogen storage, green is for processes moving nitrogen from one place to another, and red is for the bacteria involved
Dissimilatory nitrate reduction to ammonium
Dissimilatory nitrate reduction to ammonium (DNRA), or nitrate/nitrite ammonification, is an anaerobic respiration process. Microbes which undertake DNRA oxidise organic matter and use nitrate as an electron acceptor, reducing it to nitrite, then ammonium (NO−3 → NO−2 → NH+4).[30] Both denitrifying and nitrate ammonification bacteria will be competing for nitrate in the environment, although DNRA acts to conserve bioavailable nitrogen as soluble ammonium rather than producing dinitrogen gas.[31]
Anaerobic ammonia oxidation
The ANaerobic AMMonia OXidation process is also know, as the
2) gas and two water molecules. This process makes up a major proportion of nitrogen conversion in the oceans. The stoichiometrically balanced formula for the ANAMMOX chemical reaction can be written as following, where an ammonium ion includes the ammonia molecule, its conjugated base
- NH+4 + NO−2 → N2 + 2 H2O (ΔG° = −357 kJ⋅mol−1).[32]
This an exergonic process (here also an exothermic reaction) releasing energy, as indicated by the negative value of ΔG°, the difference in Gibbs free energy between the products of reaction and the reagents.
Other processes
Though nitrogen fixation is the primary source of plant-available nitrogen in most ecosystems, in areas with nitrogen-rich bedrock, the breakdown of this rock also serves as a nitrogen source.[33][34][35] Nitrate reduction is also part of the iron cycle, under anoxic conditions Fe(II) can donate an electron to NO−3 and is oxidized to Fe(III) while NO−3 is reduced to NO−2, N2O, N2, and NH+4 depending on the conditions and microbial species involved.[36] The fecal plumes of cetaceans also act as a junction in the marine nitrogen cycle, concentrating nitrogen in the epipelagic zones of ocean environments before its dispersion through various marine layers, ultimately enhancing oceanic primary productivity.[37]
Marine nitrogen cycle


2 fixation (red), nitrification (light blue), nitrate reduction (violet), DNRA (magenta), denitrification (aquamarine), N-damo (green), and anammox (orange). Black curved arrows represent physical processes such as advection and diffusion.[38]
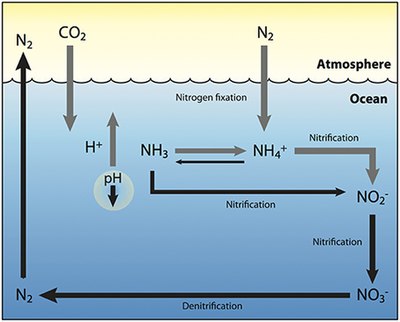
The nitrogen cycle is an important process in the ocean as well. While the overall cycle is similar, there are different players[40] and modes of transfer for nitrogen in the ocean. Nitrogen enters the water through the precipitation, runoff, or as N
2 from the atmosphere. Nitrogen cannot be utilized by phytoplankton as N
2 so it must undergo nitrogen fixation which is performed predominately by cyanobacteria.[41] Without supplies of fixed nitrogen entering the marine cycle, the fixed nitrogen would be used up in about 2000 years.[42] Phytoplankton need nitrogen in biologically available forms for the initial synthesis of organic matter. Ammonia and urea are released into the water by excretion from plankton. Nitrogen sources are removed from the euphotic zone by the downward movement of the organic matter. This can occur from sinking of phytoplankton, vertical mixing, or sinking of waste of vertical migrators. The sinking results in ammonia being introduced at lower depths below the euphotic zone. Bacteria are able to convert ammonia to nitrite and nitrate but they are inhibited by light so this must occur below the euphotic zone.[43] Ammonification or Mineralization is performed by bacteria to convert organic nitrogen to ammonia. Nitrification can then occur to convert the ammonium to nitrite and nitrate.[44] Nitrate can be returned to the euphotic zone by vertical mixing and upwelling where it can be taken up by phytoplankton to continue the cycle. N
2 can be returned to the atmosphere through denitrification.
Ammonium is thought to be the preferred source of fixed nitrogen for phytoplankton because its assimilation does not involve a redox reaction and therefore requires little energy. Nitrate requires a redox reaction for assimilation but is more abundant so most phytoplankton have adapted to have the enzymes necessary to undertake this reduction (nitrate reductase). There are a few notable and well-known exceptions that include most Prochlorococcus and some Synechococcus that can only take up nitrogen as ammonium.[42]
The nutrients in the ocean are not uniformly distributed. Areas of upwelling provide supplies of nitrogen from below the euphotic zone. Coastal zones provide nitrogen from runoff and upwelling occurs readily along the coast. However, the rate at which nitrogen can be taken up by phytoplankton is decreased in
Nitrate is depleted in near-surface water except in upwelling regions. Coastal upwelling regions usually have high nitrate and
Ammonium and nitrite show a maximum concentration at 50–80 m (lower end of the
New vs. regenerated nitrogen
Nitrogen entering the euphotic zone is referred to as new nitrogen because it is newly arrived from outside the productive layer.[41] The new nitrogen can come from below the euphotic zone or from outside sources. Outside sources are upwelling from deep water and nitrogen fixation. If the organic matter is eaten, respired, delivered to the water as ammonia, and re-incorporated into organic matter by phytoplankton it is considered recycled/regenerated production.
New production is an important component of the marine environment. One reason is that only continual input of new nitrogen can determine the total capacity of the ocean to produce a sustainable fish harvest.[45] Harvesting fish from regenerated nitrogen areas will lead to a decrease in nitrogen and therefore a decrease in primary production. This will have a negative effect on the system. However, if fish are harvested from areas of new nitrogen the nitrogen will be replenished.
Future acidification
As illustrated by the diagram on the right, additional carbon dioxide (CO2) is absorbed by the ocean and reacts with water, carbonic acid (H
2CO
3) is formed and broken down into both bicarbonate (HCO−3) and hydrogen (H+
) ions (gray arrow), which reduces bioavailable carbonate (CO2−3) and decreases ocean pH (black arrow). This is likely to enhance nitrogen fixation by diazotrophs (gray arrow), which utilize H+
ions to convert nitrogen into bioavailable forms such as ammonia (NH
3) and ammonium ions (NH+4). However, as pH decreases, and more ammonia is converted to ammonium ions (gray arrow), there is less oxidation of ammonia to nitrite (NO–
2), resulting in an overall decrease in nitrification and denitrification (black arrows). This in turn would lead to a further build-up of fixed nitrogen in the ocean, with the potential consequence of eutrophication. Gray arrows represent an increase while black arrows represent a decrease in the associated process.[39]
Human influences on the nitrogen cycle


As a result of extensive cultivation of legumes (particularly
Generation of Nr, reactive nitrogen, has increased over 10 fold in the past century due to global industrialisation.[2][47] This form of nitrogen follows a cascade through the biosphere via a variety of mechanisms, and is accumulating as the rate of its generation is greater than the rate of denitrification.[48]
Ammonia (NH
3) in the atmosphere has tripled as the result of human activities. It is a reactant in the atmosphere, where it acts as an aerosol, decreasing air quality and clinging to water droplets, eventually resulting in nitric acid (HNO3) that produces acid rain. Atmospheric ammonia and nitric acid also damage respiratory systems.
The very high temperature of lightning naturally produces small amounts of NO
x, NH
3, and HNO
3, but high-temperature combustion has contributed to a 6- or 7-fold increase in the flux of NO
x to the atmosphere. Its production is a function of combustion temperature - the higher the temperature, the more NO
x is produced. Fossil fuel combustion is a primary contributor, but so are biofuels and even the burning of hydrogen. However, the rate that hydrogen is directly injected into the combustion chambers of internal combustion engines can be controlled to prevent the higher combustion temperatures that produce NO
x.
Ammonia and nitrous oxides actively alter
Decreases in
Consequence of human modification of the nitrogen cycle

Impacts on natural systems
Increasing levels of nitrogen deposition are shown to have a number of negative effects on both terrestrial and aquatic ecosystems.[52][53] Nitrogen gases and aerosols can be directly toxic to certain plant species, affecting the aboveground physiology and growth of plants near large point sources of nitrogen pollution. Changes to plant species may also occur, as accumulation of nitrogen compounds increase its availability in a given ecosystem, eventually changing the species composition, plant diversity, and nitrogen cycling. Ammonia and ammonium – two reduced forms of nitrogen – can be detrimental over time due to an increased toxicity toward sensitive species of plants,[54] particularly those that are accustomed to using nitrate as their source of nitrogen, causing poor development of their roots and shoots. Increased nitrogen deposition also leads to soil acidification, which increases base cation leaching in the soil and amounts of aluminum and other potentially toxic metals, along with decreasing the amount of nitrification occurring and increasing plant-derived litter. Due to the ongoing changes caused by high nitrogen deposition, an environment's susceptibility to ecological stress and disturbance – such as pests and pathogens – may increase, thus making it less resilient to situations that otherwise would have little impact to its long-term vitality.
Additional risks posed by increased availability of inorganic nitrogen in aquatic ecosystems include water acidification;
Ammonia (NH
3) is highly toxic to fish and the level of ammonia discharged from wastewater treatment facilities must be closely monitored. To prevent fish deaths, nitrification via aeration prior to discharge is often desirable. Land application can be an attractive alternative to the aeration.
Impacts on human health: nitrate accumulation in drinking water
Leakage of Nr (reactive nitrogen) from human activities can cause nitrate accumulation in the natural water environment, which can create harmful impacts on human health. Excessive use of N-fertilizer in agriculture has been one of the major sources of nitrate pollution in groundwater and surface water.[59][60] Due to its high solubility and low retention by soil, nitrate can easily escape from the subsoil layer to the groundwater, causing nitrate pollution. Some other non-point sources for nitrate pollution in groundwater are originated from livestock feeding, animal and human contamination and municipal and industrial waste. Since groundwater often serves as the primary domestic water supply, nitrate pollution can be extended from groundwater to surface and drinking water in the process of potable water production, especially for small community water supplies, where poorly regulated and unsanitary waters are used.[61]
The
Impacts on human health: air quality
Human activities have also dramatically altered the global nitrogen cycle via production of nitrogenous gases, associated with the global atmospheric nitrogen pollution. There are multiple sources of atmospheric
2 can be oxidized to nitric acid (HNO
3), and it can further react with NH
3 to form ammonium nitrate (NH4NO3), which facilitates the formation of particulate nitrate. Moreover, NH
3 can react with other acid gases (sulfuric and hydrochloric acids) to form ammonium-containing particles, which are the precursors for the secondary organic aerosol particles in photochemical smog.[65]
See also
- Planetary boundaries – Limits not to be exceeded if humanity wants to survive in a safe ecosystem
- Phosphorus cycle – Biogeochemical movement
References
- PMID 23713126.
- ^ (PDF) from the original on 2011-11-08. Retrieved 2019-09-23.
- PMID 23713117.
- ^ PMID 23713119.
- ^ PMID 23713126.
- S2CID 154935568.
- PMID 23713124.
- ISSN 0148-0227.
- PMID 23713128.
- S2CID 839759.
- ^ S2CID 11204131.
- PMID 21576477.
- OCLC 228429704.
- OCLC 690090202.
- S2CID 10804715.
- ISBN 978-0-88192-611-8.
- S2CID 3948918.
- S2CID 98109580.
- ISSN 1748-9326.
- PMID 22852755.
- PMID 28462990.
- ISBN 978-1-904455-86-8.
- ISBN 9781402036118.
- ISBN 9780716750796.
- ISBN 978-0-07-337526-7.
- PMID 24141308.
- PMID 22842521.
- ^ ISSN 1051-0761.
- PMID 33658719.
- PMID 21329208.
- PMID 25127459.
- ^ "Anammox". Anammox - MicrobeWiki. MicrobeWiki. Archived from the original on 2015-09-27. Retrieved 5 July 2015.
- ^ "Nitrogen Study Could 'Rock' A Plant's World". NPR.org. 2011-09-06. Archived from the original on 2011-12-05. Retrieved 2011-10-22.
- S2CID 2946571.
- S2CID 4352571.
- ISSN 1540-9309.
- PMID 20949007.
- doi:10.3389/fmars.2019.00739..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ^ doi:10.3389/fmars.2016.00047..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ISSN 1540-9295.
- ^ ISBN 978-0-632-05536-4.
- ^ ISBN 978-0-12-372522-6.
- ISBN 978-0-632-05536-4.
- ^ Boyes, Elliot, Susan, Michael. "Learning Unit: Nitrogen Cycle Marine Environment". Archived from the original on 15 April 2012. Retrieved 22 October 2011.
{{cite web}}: CS1 maint: multiple names: authors list (link) - ^ ISBN 978-0-7506-3384-0.)
{{cite book}}: CS1 maint: multiple names: authors list (link - S2CID 189917368.
- PMID 22852755.
- S2CID 3356400.
- ISBN 0-387-95443-0.
- ^ Proceedings of the Scientific Committee on Problems of the Environment (SCOPE) International Biofuels Project Rapid Assessment, 22–25 September 2008, Gummersbach, Germany, R. W. Howarth and S. Bringezu, editors. 2009 Executive Summary, p. 3 Archived 2009-06-06 at the Wayback Machine
- S2CID 34882407.
- (PDF) from the original on 2019-09-30. Retrieved 2019-09-30.
- PMID 20828899.
- .
- PMID 16781774.
- JSTOR 3069262.
- .
- ^ New York State Environmental Conservation, Environmental Impacts of Acid Deposition: Lakes [1] Archived 2010-11-24 at the Wayback Machine
- ISSN 0167-8809.
- ISSN 0167-8809.
- PMID 15471727.
- OCLC 50144984.
- S2CID 133944481
- S2CID 38513536.
- PMID 23713116.