Plasmodium knowlesi
| Plasmodium knowlesi | |
|---|---|
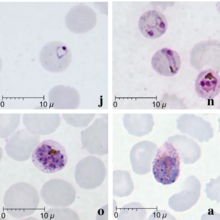
| |
| Giemsa-stained smears of Plasmodium knowlesi infecting human red blood cells | |
| Scientific classification | |
| Domain: | Eukaryota |
| Clade: | Diaphoretickes |
| Clade: | SAR |
| Clade: | Alveolata |
| Phylum: | Apicomplexa |
| Class: | Aconoidasida |
| Order: | Haemospororida |
| Family: | Plasmodiidae |
| Genus: | Plasmodium |
| Species: | P. knowlesi
|
| Binomial name | |
| Plasmodium knowlesi Sinton and Mulligan 1932
| |
Plasmodium knowlesi is a parasite that causes malaria in humans and other primates. It is found throughout Southeast Asia, and is the most common cause of human malaria in Malaysia. Like other Plasmodium species, P. knowlesi has a life cycle that requires infection of both a mosquito and a warm-blooded host. While the natural warm-blooded hosts of P. knowlesi are likely various Old World monkeys, humans can be infected by P. knowlesi if they are fed upon by infected mosquitoes. P. knowlesi is a eukaryote in the phylum Apicomplexa, genus Plasmodium, and subgenus Plasmodium. It is most closely related to the human parasite Plasmodium vivax as well as other Plasmodium species that infect non-human primates.
Humans infected with P. knowlesi can develop
P. knowlesi was first described as a distinct species and as a potential cause of human malaria in 1932. It was briefly used in the early 20th century to cause fever as a treatment for neurosyphilis. In the mid-20th century, P. knowlesi became popular as a tool for studying Plasmodium biology and was used for basic research, vaccine research, and drug development. P. knowlesi is still used as a laboratory model for malaria, as it readily infects the model primate the rhesus macaque, and can be grown in cell culture in human or macaque blood.
Life cycle

Like other Plasmodium parasites, P. knowlesi has a life cycle that requires it be passed back and forth between mammalian hosts and insect hosts. Primates are infected through the bite of an infected
A mosquito ingests gametocytes when it takes a
Cell biology

P. knowlesi largely resembles other
As an
Evolution and taxonomy
Despite its morphological similarity to P. malariae, P. knowlesi is most closely related to P. vivax as well as other Plasmodium species that infect non-human primates.[9] The last common ancestor of all modern P. knowlesi strains lived an estimated 98,000 to 478,000 years ago.[9] Among human parasites, P. knowlesi is most closely related to P. vivax, from which it diverged between 18 million and 34 million years ago.[5][10] A phylogenetic tree comparing the Plasmodium species that infect humans is shown below:[5]
| Subgenus Plasmodium |
| ||||||
The population of P. knowlesi parasites is more genetically diverse than that of P. falciparum or P. vivax. Within P. knowlesi there are three genetically distinct subpopulations.[5] Two are present in the same areas of Malaysian Borneo and may infect different mosquitoes.[5] The third has been found only in laboratory isolates originating from other parts of Southeast Asia.[5] Populations of P. knowlesi isolated from macaques are genetically indistinguishable from those isolated from human infections, suggesting the same parasite populations can infect humans and macaques interchangeably.[11]
Three subspecies of P. knowlesi have been described based on differences in their appearance in stained blood films: P. knowlesi edesoni, P. knowlesi sintoni, and P. knowlesi arimai, which were isolated from Malaysia, Java, and Taiwan respectively.[2][12] The relationship between these described subspecies and the populations described in the modern literature is not clear.[2]
Distribution
Plasmodium knowlesi is found throughout
Role in human disease

P. knowlesi can cause both
At least 10% of people infected with P. knowlesi develop severe malaria.
Diagnosis
Malaria is traditionally diagnosed by examining
Due to the morphological similarity among Plasmodium species, misdiagnosis of P. knowlesi infection as P. falciparum, P. malariae, or P. vivax is common.
Treatment
Because P. knowlesi takes only 24 hours to complete its erythrocytic cycle, it can rapidly result in very high levels of parasitemia with fatal consequences.
Epidemiology
P. knowlesi is the most common cause of malaria in Malaysia,[5] and cases of P. knowlesi malaria have been reported in most countries of Southeast Asia as well as travelers from the region.[11]
Infection with P. knowlesi is associated with socioeconomic and lifestyle factors that bring people into the dense forests where the mosquito hosts are commonly found.[14] In particular, those who work in the forest or at its margin such as farmers, hunters, and loggers are at increased risk for infection.[14] Likely for this reason, males are infected more frequently than females, and adults are infected more frequently than children.[14]
Research
P. knowlesi has long been used as a research model for studying the interaction between parasite and host, and developing antimalarial vaccines and drugs.[10] Its utility as a research model is partly due to its ability to infect rhesus macaques, a common laboratory model primate. Rhesus macaques are highly susceptible to P. knowlesi and can be infected by mosquito bite, injection of sporozoites, or injection of blood-stage parasites.[2][10] Infected monkeys develop some hallmarks of human malaria including anemia and enlargement of the spleen and liver.[2] Infection is typically fatal if untreated, with the cause of death seemingly circulatory failure characterized by adhesion of infected red blood cells to the blood vessel walls.[2] Monkeys can be cured of infection by treatment with antimalarials; repeated infection followed by cure results in the monkeys developing some immunity to infection, a topic that has also been the subject of substantial research.[2]
P. knowlesi is also used for
History

The Italian physician
Soon thereafter, in 1935 C. E. Van Rooyen and George R. Pile reported using P. knowlesi infection to treat
Through the 1960s and 1970s, scientific research groups used P. knowlesi as a research model to make seminal discoveries in malaria. In 1965 and 1972, several groups characterized how P. knowlesi antigenic variation contributed to immune evasion and chronic infection.[24] In 1975, Louis H. Miller and others showed that P. knowlesi required Duffy factor on the surface of red blood cells in order to invade them (they would go on to show the same requirement for P. vivax a year later).[24]
Work on P. knowlesi as a human malaria parasite was revitalized in 2004, when Balbir Singh and others used PCR to show that over half of a group of humans diagnosed with P. malariae malaria in Malaysian Borneo were actually infected with P. knowlesi.[24][28] Over the following decade, several investigators used molecular detection methods capable of distinguishing P. knowlesi from morphologically similar parasites to attribute an increasing proportion of malaria cases to P. knowlesi throughout Southeast Asia.[29] Work with archival samples has shown that infection with this parasite has occurred in Malaysia at least since the 1990s.[30]
References
- ^ PMID 22149265.
- ^ S2CID 13209647.
- ^ a b c d e f Coatney GR, Collins WE, Warren M, Contacos PG (1971). The Primate Malarias. Atlanta, GA: Division of Parasitic Diseases, U.S. Centers for Disease Control. pp. 317–334. Retrieved 26 August 2019.
- PMID 27639778.
- ^ S2CID 53228888.
- PMID 23570755.
- PMID 23186105.
- PMID 23927894.
- ^ PMID 21490952.
- ^ S2CID 9261727.
- ^ PMID 25843504.
- PMID 13960457.
- PMID 35674419.
- ^ PMID 23554413.
- S2CID 27841173.
- ^ PMID 27707609.
- ^ a b c d "DPDx Malaria - Image Gallery". U.S. Centers for Disease Control and Prevention. 29 December 2017. Retrieved 20 January 2020.
- ^ Despommier DD, Griffin DO, Gwadz RW, Hotez PJ, Knirsch CA. Parasitic Diseases (PDF) (7 ed.). Parasites Without Borders. p. 112.
- ISBN 978-92-4-154912-7. Archived from the originalon April 25, 2015. Retrieved 22 January 2020.
- S2CID 19786233.
- ^ Franchini G (1927) Su di un plasmodio pigmentato di una scimmia. Arch Ital Sci Med Colon 8:187–90
- PMID 21890752.
- S2CID 26553543. Archived from the original(PDF) on 2019-02-28. Retrieved 27 August 2019.
- ^ PMID 23088834.
- PMID 29010910.
- PMID 13467997.
- PMID 26307748.
- S2CID 7776536.
- PMID 18710577.
- PMID 19358848.
