Triazofos
| |
| Names | |
|---|---|
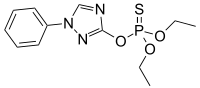
| Preferred IUPAC name
O,O-Diethyl O-(1-phenyl-1H-1,2,4-triazol-3-yl) phosphorothioate | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.041.791 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H16N3O3PS | |
| Molar mass | 313.31 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Triazofos is a chemical compound used in acaricides, insecticides, and nematicides.
History
Triazofos has been registered in the
Synthesis and available forms
Synthesis
Triazofos can be synthesized through various reactions.
A method of manufacturing triazofos produces the substance in the presence of triethylamine by reacting 1-phenyl-3-hydroxy-1H-1,2,4-triazole suspended in acetone with diethoxythiophosphoryl chloride.[4]
Another method produces the substance by the reaction of
An improved process for manufacturing triazofos uses
Available forms
Triazofos is available in several forms; as an emulsifiable concentrate (40%), wettable concentrates, wettable powders (30%), ultralow-volume liquids (25%, 40%) and granules (5%) at various concentrations.[6]
Chemical properties
Structure and reactivity
Triazofos is an
Metabolism and mechanisms of action
Metabolism
The metabolic fate of triazofos has been studied in rats and dogs.
23 female
The maximum blood drug concentration (
From all the tissues that were analysed, the highest concentration of
The metabolic fate of triazofos was also examined in two female
Altogether, the metabolic fate of triazofos in dogs was similar to that in rats .[8] The urine consisted of the same three metabolites as in rats. However, there was one other metabolite found only in the dog's urine representing 11% of the administered dose. It was considered to be another sulfate ester conjugate of the 1-phenyl-3-hydroxy-(1H)-1,2,4-triazole metabolite. There was no unchanged triazofos found in the urine of the dogs. The faeces contained low concentrations of triazofos and the free 1-phenyl-3-hydroxy-(1H)-1,2,4-triazole metabolite as well as five unidentified metabolites at about 0.7, 0.3 and 7.3% of the administered dose, respectively.
Effects on animals
The signal of oral poisoning similarly happen in mice, rats and dogs, characterized by tremors, abdominal position, squatting, jerky respiration, lachrymation, salivation, saltatory spasm, tonic convulsions.[9][10]
- Acute Toxicity: In mice, rats and guinea-pigs, the acute LD50 value of triazofos ranged from 26–82 mg/kg body weight, while dogs have higher values up to 500 mg/kg body weight. Deaths occurred within minutes to several days after oral administration. Resulting in WHO's consideration of triazofos as a highly hazardous compound.
- Genotoxicity: In the study of toxicity and carcinogenicity effects in mice and rats, triazofos induced no significant or consistent increase in any tumour types.
- Reproductive toxicity: Signs of toxicity such as aggressive behaviour and decreased body weight and food consumption were seen only in F1 parent. Hence, there is no significant effect observed for the reproductive toxicity
Mechanism of action
Triazofos interacts with several enzymes and signalling pathways according to various bio-assay results:[11][4]
- inhibits vitamin D receptor (VDR).
- acts as a small molecule disruptors of the mitochondrial membrane potential.
- acts as a disruptor of small molecule activators of the human pregnane X receptor (PXR) signalling pathway.
- acts as a genotoxic compound against isogenic chicken DT40 cell lines.
- acts as a small molecule antagonist of the oestrogen receptor alpha (Er-alpha) signalling pathway.
- acts as a small molecule that activates the Aryl (AHR) hydrocarbon receptor signalling pathway.
- acts as a small molecule antagonist of the constitutive androstane receptor (CAR) signalling pathway.
Efficacy and toxicity
Efficacy
The use of triazofos as an insecticide in many Asian countries such China, India and Indonesia is widely known due to many insects and pests playing important roles in the market production of staple plant food production.[12] Among various constraints, leafhoppers (Amarasca devastans) and whiteflies (Bemisia tabaci) are one of the major factors in cultivation problems due to their capability to suck the cell sap of plants.
An experiment was conducted by Horticultural Ecosystem in India about the efficacy of triazofos as an insecticide for leafhoppers and whiteflies on Brinjal (Solamum melongena L), one of the prominent crops in India.[13] The investigation was arranged with various market names of triazofos with varying concentrations. The analysis was established after 20 days of transplanting and observing the pest incidence.
Before spraying the insecticide, there were no observations of significant numbers between leafhoppers and whiteflies with the respect to leaf samples.[13]
The visual observations were also constructed in assessing phytotoxic symptoms such as injury on leaf tips or surface, wilting, etc. Nonetheless, no phytotoxic symptoms were observed on the plants with the treatment. In conclusion, the triazofos of 1250 ml/ ha was most effective against leafhoppers, whiteflies and shoot and fruit borer of brinjal.[13]
Toxicity
Triazofos (O,O-diethyl O-1-phenyl-1H-1,2,4-triazol-3-yl phosphorothioate) is considered an
Toxicological evaluation revealed the maximum level of triazofos which causes no toxicological effect and the maximum level of exposure considered acceptable for humans. The estimated acceptable daily intake for humans is 0–0.001 mg/kg bodyweight.(See Table 2 and 3)
| Mouse | 30 ppm in the diet, equal to 4.5 mg/kg bw/day (2 year of study) |
|---|---|
| Rat | 3 ppm in the diet, equal to 0.17 mg/kg bw/day (2 year of study) |
| Dog | 4 ppm in the diet, equal to 0.12 mg/kg bw/day (1 year of study) |
| Human | 0.0125 mg/kg bw/day (3 week study) |
| Summary | Value | Study | Safety Factor |
|---|---|---|---|
| ADI | 0-0.001 mg/kg bw | 3 weeks, humans | 10 |
| Acute RfD | 0.001 mg/kg bw | 3 weeks, humans | 10 |
Adverse effects and health hazards in humans
Acute exposure to triazofos may produce the following signs and symptoms:
Antidote
The following
References
- ^ Brandt, P. Franz, H. Holzman, A. (2009). Berichte zu Pflanzenschutzmitteln 2009. Retrieved on March 17th 2017, from website: http://www.bvl.bund.de/SharedDocs/Downloads/04_Pflanzenschutzmittel/bericht_WirkstoffeI%20nPSM_2009.pdf?__blob=publicationFile&v=3
- ^ Commission Regulation (EC) No 1336/2003 of 25 July 2003 amending Regulation (EC) No 2076/2002 as regards the continued use of the substances listed in Annex II
- ^ CBG. (Undated). Coalition against BAYER dangers. Retrieved on March 17th 2017, from website: http://www.cbgnetwork.org/4047.html
- ^ a b c Toxnet. (Undated). HSDB: Triazophos: Methods of Manufacturing. Retrieved on March 10th 2017, from website: https://toxnet.nlm.nih.gov/cgibin/sis/search2/r?dbs+hsdb:@term+@rn+@rel+24017-47-8
- ^ Sambhaji, P.S Murgyappa, S.A. Shivaji, B.C. Bhairu, K.V. Gopal, M.S. Pratap, S.M & Kumar, K.V. (2008). An improved process for preparation of triazophos. Retrieved on March 10th 2017, from website: http://www.allindianpatents.com/patents/220854-an-improved-process-for-preparationof-triazophos
- ^ PubChem Compound Database. (Undated). Triazophos: Formulations/Preparations. Retrieved on March 17th 2017, from website: https://pubchem.ncbi.nlm.nih.gov/compound/Triazophos#section=Formulations-Preparations
- ^ a b Cameo Chemicals. (Undated). Triazofos. Retrieved on March 17th 2017 from website: https://cameochemicals.noaa.gov/chemical/5222
- ^ a b c InChem. (Undated). Triazophos. Retrieved on March 17th from website: http://www.inchem.org/documents/jmpr/jmpmono/v86pr18.htm
- ^ a b Inchem. (1982). Pesticide Residues in food – 1982. Retrieved on March 17th 2017, from website: http://www.inchem.org/documents/jmpr/jmpmono/v82pr33.htm
- ^ a b Hamernik, K.L. (Undated). Pesticide residues in food – 2002- Joint FAO/WHO meeting on pesticide residues: Triazophos. Retrieved on March 17th 2017, from website: http://www.inchem.org/documents/jmpr/jmpmono/2002pr14.htm
- ^ a b PubChem Compound Database. (Undated). Triazophos: Biological Test Results (Bioassay results). Retrieved on March 17th 2017, from website:https://pubchem.ncbi.nlm.nih.gov/compound/Triazophos#section=BioAssay-Results
- ^ Lal, R. Jat, B.J. "Bio-efficacy of insecticides and biorationals against the incidence of whitefly, Bemisia tabaci (Genn.) and yellow mosaic virus in mungbean. Departments of Entomology, CCS Haryana Agricultural University, India. African Journal of Agricultural Research. Vol. !0 (10) pp. 1050-1056.
- ^ a b c Kumar, P. (2010). "Efficacy of Triazophos 40 EC against pest complex of brinjal". Kittur Rani Channamma College of Hrticulture, Karnataka, India. Pest Management in Horicultural Ecosystems. Vol. 16 (1). Pp 87-89.
