Polymer chemistry
Polymer chemistry is a sub-discipline of chemistry that focuses on the structures of chemicals, chemical synthesis, and chemical and physical properties of polymers and macromolecules. The principles and methods used within polymer chemistry are also applicable through a wide range of other chemistry sub-disciplines like organic chemistry, analytical chemistry, and physical chemistry. Many materials have polymeric structures, from fully inorganic metals and ceramics to DNA and other biological molecules. However, polymer chemistry is typically related to synthetic and organic compositions. Synthetic polymers are ubiquitous in commercial materials and products in everyday use, such as plastics, and rubbers, and are major components of composite materials. Polymer chemistry can also be included in the broader fields of polymer science or even nanotechnology, both of which can be described as encompassing polymer physics and polymer engineering.[1][2][3][4]
History
The work of
In 1884
- Leading figures in polymer chemistry
-
Hermann Staudinger, father of polymer chemistry
-
Wallace Carothers, inventor of nylon.
-
Stephanie Kwolek, inventor of Kevlar.
Teaching and research programs in polymer chemistry were introduced in the 1940s. An Institute for Macromolecular Chemistry was founded in 1940 in Freiburg, Germany under the direction of Staudinger. In America, a Polymer Research Institute (PRI) was established in 1941 by
Polymers and their properties

The formation and properties of polymers have been rationalized by many theories including

The study of polymer thermodynamics helps improve the material properties of various polymer-based materials such as
Viscosity
As polymers get longer and their molecular weight increases, their viscosity tend to increase. Thus, the measured viscosity of polymers can provide valuable information about the average length of the polymer, the progress of reactions, and in what ways the polymer branches.[11]

Classification
Polymers can be classified in many ways. Polymers, strictly speaking, comprise most solid matter: minerals (i.e. most of the Earth's crust) are largely polymers, metals are 3-d polymers, organisms, living and dead, are composed largely of polymers and water. Often polymers are classified according to their origin:
- biopolymers
- synthetic polymers
- inorganic polymers
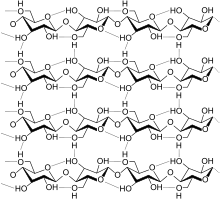
Biopolymers are the structural and functional materials that comprise most of the organic matter in organisms. One major class of biopolymers are proteins, which are derived from amino acids. Polysaccharides, such as cellulose, chitin, and starch, are biopolymers derived from sugars. The polynucleic acids DNA and RNA are derived from phosphorylated sugars with pendant nucleotides that carry genetic information.
See also
References
- ^ "The Macrogalleria: A Cyberwonderland of Polymer Fun". www.pslc.ws. Retrieved 2018-08-01.
- ISBN 0-412-22170-5
- OCLC 54781987.
- ^ "The Early Years of Artificial Fibres". The Plastics Historical Society. Retrieved 2011-09-05.
- )
- ^ "History of Cellophane". about.com. Archived from the original on June 29, 2012. Retrieved 2011-09-05.
- ^ "The Nobel Prize in Chemistry 2000". Retrieved 2009-06-02.
- S2CID 4328634.
- ^ X Zhang, X Peng, SW Zhang. "7 - Synthetic biodegradable medical polymers: Polymer blends" Science and Principles of Biodegradable and Bioresorbable Medical Polymers, 2017. 217-254.
- ^ "Viscosity of Polymer Solutions". polymerdatabase.com. Retrieved 2019-03-05.






