Naturally occurring phenols
In biochemistry, naturally occurring phenols are natural products containing at least one phenol functional group.[1][2][3] Phenolic compounds are produced by plants and microorganisms.[4] Organisms sometimes synthesize phenolic compounds in response to ecological pressures such as pathogen and insect attack, UV radiation and wounding.[5] As they are present in food consumed in human diets and in plants used in traditional medicine of several cultures, their role in human health and disease is a subject of research.[1][5][6][7]: 104 Some phenols are germicidal and are used in formulating disinfectants.


Classification
Various
| Number of carbon atoms | Basic skeleton | Number of phenolic cycles | Class | Examples |
|---|---|---|---|---|
| 6 | C6 | 1 | Simple phenols, Benzoquinones | Catechol, Hydroquinone, 2,6-Dimethoxybenzoquinone |
| 7 | C6-C1 | 1 | Phenolic acids, Phenolic aldehydes | Gallic, salicylic acids |
| 8 | C6-C2 | 1 | Acetophenones, Tyrosine derivatives, Phenylacetic acids | p-Hydroxyphenylacetic acid, Homogentisic acid
|
| 9 | C6-C3 | 1 | Hydroxycinnamic acids, Allylbenzenes, Coumarins, Isocoumarins, Chromones | Caffeic, ferulic acids, Myristicin, Eugenol, Umbelliferone, aesculetin, Bergenon, Eugenin |
| 10 | C6-C4 | 1 | Naphthoquinones | Juglone, Plumbagin |
| 13 | C6-C1-C6 | 2 | Xanthonoids | Mangiferin |
| 14 | C6-C2-C6 | 2 | Anthraquinones
|
Resveratrol, Emodin |
| 15 | C6-C3-C6 | 2 | Chalconoids, Flavonoids, Isoflavonoids, Neoflavonoids | Quercetin, cyanidin, Genistein |
| 16 | C6-C4-C6 | 2 | Halogenated algal phenolic compounds | Kaviol A, colpol |
| 18 | (C6-C3)2 | 2 | Neolignans
|
Pinoresinol, Eusiderin |
| 30 | (C6-C3-C6)2 | 4 | Biflavonoids | Amentoflavone |
| many | (C6-C3)n, (C6)n, (C6-C3-C6)n |
n > 12 | Polyphenolic proteins,
Polyphenols |
Raspberry ellagitannin, Tannic acid |
C6-C7-C6 Diarylheptanoids are not included in this Harborne classification.
They can also be classified on the basis of their number of phenol groups. They can therefore be called simple phenols or monophenols, with only one phenolic group, or di- (bi-), tri- and oligophenols, with two, three or several phenolic groups respectively.
A diverse family natural phenols are the
The phenolic unit can be found dimerized or further polymerized, creating a new class of polyphenol. For example, ellagic acid is a dimer of gallic acid and forms the class of ellagitannins, or a catechin and a gallocatechin can combine to form the red compound theaflavin, a process that also results in the large class of brown thearubigins in tea.
Two natural phenols from two different categories, for instance a flavonoid and a lignan, can combine to form a hybrid class like the flavonolignans.
Nomenclature of polymers:
| Base Unit: | Gallic Acid
|
 Flavone |
 Cinnamic acid |
|---|---|---|---|
| Class/Polymer: | Hydrolyzable tannins
|
Flavonoid, Condensed tannins | Lignins |
Hybrid chemical classes
Plants in the genus Humulus and Cannabis produce terpenophenolic metabolites, compounds that are meroterpenes.[12][13] Phenolic lipids are long aliphatic chains bonded to a phenolic moiety.
Chirality
Many natural phenols are
UV visible absorbance
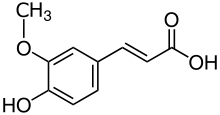
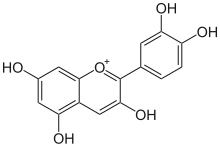
Natural phenols show optical properties characteristic of benzene, e.g. absorption near 270 nm. According to
As molecules with higher conjugation levels undergo this bathochromic shift phenomenon, a part of the visible spectrum is absorbed. The wavelengths left in the process (generally in red section of the spectrum) recompose the
Here is a series of UV visible spectra of molecules classified from left to right according to their conjugation level:
lambda max around 270 nanometers (nm) |
 |
 |
cyanidin-3-O-glucoside (chrysanthemin), with lambda max at 518 nm. |
 |
 |
 |
 |
The absorbance pattern responsible for the red color of anthocyanins may be complementary to that of green chlorophyll in photosynthetically active tissues such as young Quercus coccifera leaves.[16]
Oxidation
Natural phenols are
Browning associated with oxidation of phenolic compounds has also been given as the cause of cells death in calli formed in in vitro cultures. Those phenolics originate both from explant tissues and from explant secretions.
Phenolic compounds
Naturally occurring
Cannabinoids |
the active constituents of cannabis |
| Capsaicin | the pungent compound of chili peppers |
| Carvacrol | found in neuroprotectant[22]
|
| Cresol | found in coal tar and creosote |
| Estradiol | estrogen - hormones |
| Eugenol | the main constituent of the essential oil of clove |
| Gallic acid | found in galls
|
| Guaiacol | (2-methoxyphenol) - has a smokey |
| Methyl salicylate | the major constituent of the essential oil of wintergreen |
| Raspberry ketone | a compound with an intense raspberry smell |
| Salicylic acid | precursor compound to Aspirin (chemical synthesis is used in manufacturing) |
noradrenaline |
natural neurotransmitters
|
| Thymol | (2-Isopropyl-5-methyl phenol) - found in thyme; an antiseptic that is used in mouthwashes |
| Tyrosine | an amino acid |
| Sesamol | a naturally occurring compound found in sesame seeds
|
Synthetic
| Phenol | the parent compound, used as a disinfectant and for chemical synthesis |
| Bisphenol A | and other bisphenols produced from ketones and phenol / cresol |
| BHT | (butylated hydroxytoluene) - a fat-soluble antioxidant and food additive |
4-Nonylphenol |
a breakdown product of detergents and nonoxynol-9
|
Orthophenyl phenol |
a citrus fruits
|
| Picric acid | (trinitrophenol) - an explosive material
|
| Phenolphthalein | pH indicator |
| Xylenol | used in antiseptics & disinfectants |
Biosynthesis
Phenolics are formed by three different biosynthetic pathways: (i) the shikimate/chorizmate or succinylbenzoate pathway, which produces the phenyl propanoid derivatives (C6–C3); (ii) the acetate/malonate or polyketide pathway, which produces the side-chain-elongated phenyl propanoids, including the large group of flavonoids (C6–C3–C6) and some quinones; and (iii) the acetate/mevalonate pathway, which produces the aromatic terpenoids, mostly monoterpenes, by dehydrogenation reactions.
In plants, the phenolic units are
In olive oil, tyrosol forms esters with fatty acids.[25] In rye, alkylresorcinols are phenolic lipids.
Some acetylations involve terpenes like geraniol.[26] Those molecules are called meroterpenes (a chemical compound having a partial terpenoid structure).
Methylations can occur by the formation of an
Biodegradation
The white rot fungus
Applications
Tannins are used in the tanning industry.
Some natural phenols can be used as biopesticides. Furanoflavonoids like karanjin or rotenoids are used as acaricide or insecticide.[28]
Some phenols are sold as dietary supplements. Phenols have been investigated as drugs. For instance, Crofelemer (USAN trade name Fulyzaq) is a drug under development for the treatment of diarrhea associated with anti-HIV drugs. Additionally, derivatives have been made of phenolic compound, combretastatin A-4, an anticancer molecule, including nitrogen or halogens atoms to increase the efficacy of the treatment.[30]
Industrial processing and analysis
Phenol extraction
Phenol extraction is a processing technology used to prepare phenols as raw materials, compounds or additives for industrial wood processing and for chemical industries.
The recovery of natural phenols from biomass residue is part of biorefining.[31]
Separation
The
Analytical methods
Studies on evaluating antioxidant capacity can use
Detection can be made by recombinant
Profiling
Phenolic profiling can be achieved with liquid chromatography–mass spectrometry (LC/MS).[36]
Quantification
A method for phenolic content quantification is
Some methods for quantification of total phenolic content are based on
Lamaison and Carnet have designed a test for the determination of the total flavonoid content of a sample (AlCI3 method). After proper mixing of the sample and the reagent, the mixture is incubated for 10 minutes at ambient temperature and the absorbance of the solution is read at 440 nm. Flavonoid content is expressed in mg/g of quercetin.[37]
Quantitation results produced by the means of
Antioxidant effect assessment
- In vitro measurements
Other tests measure the antioxidant capacity of a fraction. Some make use of the 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) (ABTS) radical cation, which is reactive towards most antioxidants including phenolics, thiols and vitamin C.[38] During this reaction, the blue ABTS radical cation is converted back to its colorless neutral form. The reaction may be monitored spectrophotometrically. This assay is often referred to as the Trolox equivalent antioxidant capacity (TEAC) assay. The reactivity of the various antioxidants tested are compared to that of Trolox, which is a vitamin E analog.
Other antioxidant capacity assays that use Trolox as a standard include the
A cellular antioxidant activity (CAA) assay also exists. Dichlorofluorescin is a probe that is trapped within cells and is easily oxidized to fluorescent
Other methods include
- In vivo models
Larvae of the model animal
Genetic analysis
The phenolic biosynthetic and metabolic pathways and enzymes can be studied by means of
Natural occurrences
Phenols are found in the natural world, especially in the plant kingdom.
Occurrences in prokaryotes
The proteobacterium
Occurrences in fungi
- In yeasts
Aromatic alcohols (example: tyrosol) are produced by the yeast Candida albicans.[50] They are also found in beer.[51] These molecules are quorum sensing compounds for Saccharomyces cerevisiae.[52]
- Metabolism
Aryldialkylphosphatase (also known as organophosphorus hydrolase, phosphotriesterase, and paraoxon hydrolase) uses an aryl dialkyl phosphate and H2O to produce dialkyl phosphate and an aryl alcohol.
Occurrences in lichen
Gyrophoric acid, a depside, and orcinol are found in lichen.[53]
Occurrence in algae
The
Phenolic acids such as
Occurrence in land plants (embryophytes)
Occurrences in vascular plants
Phenolic compounds are mostly found in
In ferns, compounds such as
In conifers (Pinophyta), phenolics are stored in polyphenolic parenchyma cells, a tissue abundant in the phloem of all conifers.[61]
The
Occurrences in monocotyledons
Alkylresorcinols can be found in cereals.[citation needed]
2,4-Bis(4-hydroxybenzyl)phenol is a phenolic compound found in the orchids Gastrodia elata and Galeola faberi.[citation needed]
Occurrences in non-vascular plants
Phenolics can also be found in non-vascular land plants (bryophytes). Dihydrostilbenoids and bis(dibenzyls) can be found in liverworts (Marchantiophyta), for instance, the macrocycles cavicularin and riccardin C. Though lignin is absent in mosses (Bryophyta) and hornworts (Anthocerotophyta), some phenolics can be found in those two taxa.[63] For instance, rosmarinic acid and a rosmarinic acid 3'-O-β-D-glucoside can be found in the hornwort Anthoceros agrestis.[64]
Occurrences in other eukaryotes
Occurrences in insects
The hardening of the protein component of
Occurrences in mammals
In female elephants, the two compounds
Roles
In some cases of natural phenols, they are present in vegetative
Role in soils
In
In the soil, soluble phenols face four different fates. They might be degraded and mineralized as a carbon source by
Role in survival
Phenolic compounds can act as protective agents, inhibitors, natural animal toxicants and pesticides against invading organisms, i.e. herbivores, nematodes, phytophagous insects, and fungal and bacterial pathogens. The scent and pigmentation conferred by other phenolics can attract symbiotic microbes, pollinators and animals that disperse fruits.[23]
Defense against predators
Volatile phenolic compounds are found in plant
In the kelp species
Defense against infection
In
Stilbenes are produced in Eucalyptus sideroxylon in case of pathogens attacks. Such compounds can be implied in the hypersensitive response of plants. High levels of phenolics in some woods can explain their natural preservation against rot.[97]
In plants,
Role in allelopathic interactions
Natural phenols can be involved in
Phenolics, and in particular
Content in human food
Notable sources of natural phenols in
Natural phenols can also be found in fatty matrices like olive oil.[104] Unfiltered olive oil has the higher levels of phenols, or polar phenols that form a complex phenol-protein complex.
Phenolic compounds, when used in
Some advocates for
Human metabolism
In animals and humans, after ingestion, natural phenols become part of the
References
- ^ PMID 23429347.
- PMID 22505046.
- PMID 12720366. Archived from the originalon 2016-03-04. Retrieved 2018-12-02.
- ^ PMID 10802549.
- ^ PMID 21243436.
- PMID 21889825.
- ISBN 9781420006186.
- ^ ISBN 9781402051647.
- ^ Harborne, J. B. (1980). "Plant phenolics". In Bell, E. A.; Charlwood, B. V. (eds.). Encyclopedia of Plant Physiology, Volume 8: Secondary Plant Products. Berlin Heidelberg New York: Springer-Verlag. pp. 329–395.
- ISBN 9783642221446
- ISBN 9780443071935.
- ISBN 9780080451251.
- PMID 16458906.
- PMID 12358498.
- PMID 16452075.
- PMID 12717756.
- PMID 19053150.
- .
- S2CID 83665714.
- PMID 10552504.
- PMID 22438954.
- ^ PMID 20696007.
- PMID 11476485.
- PMID 20524658.
- PMID 17625893.
- PMID 19062164.
- PMID 25928224.
- ISBN 9781741998580. Archivedfrom the original on 2022-11-05.
- PMID 20933304.
- PMID 20593898.
- PMID 9098965.
- .
- PMID 20866027.
- PMID 16581105.
- S2CID 39140266.
- ^ "Teneurs en principaux flavonoides des fleurs de Cratageus monogyna Jacq et de Cratageus Laevigata (Poiret D.C.) en Fonction de la vegetation". J. L. Lamaison and A. Carnet, Plantes Medicinales Phytotherapie, 1991, XXV, pages 12–16
- PMID 19199590.
- .
- PMID 17902627.
- .
- PMID 17694562.
- PMID 8580908.
- PMID 20073469.
- ^ Production of phenolic compounds by Spirulina maxima microalgae and their protective effects in vitro toward hepatotoxicity model. Abd El-Baky Hanaa H., El Baz Farouk K. and El-Baroty Gamal S., Advances in food sciences, 2009, volume 31, number 1, pp. 8–16, INIST 21511068
- ^ S2CID 26084768.
- PMID 15826166.
- PMID 19425182.
- .
- PMID 18836025.
- .
- S2CID 3970864.
- ^ Robiquet (1829). "Essai analytique des lichens de l'orseille". Annales de chimie et de physique. 42: 236–257.
- PMID 19410410.
- PMID 11809064.
- .
- PMID 10913848.
- .
- ^ Flavonoids and a proanthrocyanidin from rhizomes of Selliguea feei. Baek Nam-In, Kennelly E. J., Kardono L. B. S., Tsauri S., Padmawinata K., Soejarto D. D. and Kinghorn A. D., Phytochemistry, 1994, vol. 36, no. 2, pp. 513–518, INIST 3300075
- PMID 18177906.
- ISBN 978-1-4020-8181-1.
- ^ .
- .
- S2CID 29302603.
- PMID 16748434.
- ^ Acetosyringone on www.pherobase.com, the pheromones data base
- .
- S2CID 34346907.
- S2CID 8561972.
- S2CID 5207502.
- ^ Semiochemical - 2-methoxy-4-vinylphenol, Pherobase.com
- S2CID 21599793.
- ^ Urinary, temporal gland, and breath odors from Asian elephants of Mudumalai National Park. L. E. L. Rasmussen and V. Krishnamurthy, Gajah, the Journal of the Asian Elephant Specialist Group, January 2001, Number 20, pages 1-8 (article)
- S2CID 21368454.
- ^ "Musth in elephants". Deepa Ananth, Zoo's print journal, 15(5), pp. 259-262 (article Archived 2018-06-04 at the Wayback Machine)
- S2CID 45857570.
- ^ "Why study pig odor?".
- S2CID 29937875.
- ^ C.Michael Hogan (2008) Western poison-oak: Toxicodendron diversilobum, GlobalTwitcher, ed. Nicklas Stromberg "Archived copy". Archived from the original on 2009-07-21. Retrieved 2009-07-21.
{{cite web}}: CS1 maint: archived copy as title (link) - ^ Biogeochemistry: An Analysis of Global Change. 2nd Edition. William H. Schlesinger, Academic Press, 1997, 108, 135, 152–158, 180–183, 191–194
- Timber Press, Portland, Oregon. 2003
- S2CID 36409146.
- S2CID 19551247.
- S2CID 5655010.
- doi:10.4454/jpp.v91i3.549 (inactive 31 January 2024). Retrieved 2011-01-22.)
{{cite journal}}: CS1 maint: DOI inactive as of January 2024 (link) CS1 maint: multiple names: authors list (link - PMID 21821423.
- PMID 17962750.
- PMID 15752644.
- ^ "Sakuranetin". home.ncifcrf.gov. Archived from the original on 2018-12-03. Retrieved 2018-12-02.
- ^ Sakuranetin, a flavonone phytoalexin from ultraviolet-irradiated rice leaves, Kodama O., Miyakawa J., Akatsuka T. and Kiyosawa S., Phytochemistry, 1992, volume 31, number 11, pp. 3807–3809, INIST 4682303
- PMID 16943219.
- ^ "Biosynthesis and regulation of 3-deoxyanthocyanidin phytoalexins induced during Sorghum-Colletotrichum interaction: Heterologous expression in maize". Chopra Surinder, Gaffoor Iffa, Ibraheem Farag, Poster at the American Society of Plant Biologists (abstract Archived 2011-07-25 at the Wayback Machine)
- .
- .
- .
- ^ Danielone, a phytoalexin from papaya fruit. Echeverri F., Torres F., Quinones W., Cardona G., Archbold R., Roldan J., Brito I., Luis J. G., and Lahlou U. E.-H., Phytochemistry, 1997, vol. 44, no. 2, pp. 255–256, INIST 2558881
- .
- PMID 15755957.
- .
- S2CID 30091640.
- ^ "Involvement of acetosyringone in plant-pathogen recognition". Baker C. Jacyn, Mock Norton M., Whitaker Bruce D., Roberts Daniel P., Rice Clifford P., Deahl Kenneth L. and Aver'Yanov Andrey A., Biochemical and Biophysical Research Communications, 2005, volume 328, number 1, pp. 130–136, INIST 16656426
- PMID 10948245.
- S2CID 32005770.
- S2CID 85367601.
- .
- ^ Asami, Danny K. "Comparison of the Total Phenolic and Ascorbic Acid Content of Freeze-Dried and Air-Dried Marionberry, Strawberry, and Corn Grown Using Conventional, Organic, and Sustainable Agricultural Practices". Journal of Agricultural and Food Chemistry (American Chemical Society), 51 (5), 1237–1241, 2003. 10.1021/jf020635c S0021-8561(02)00635-0. Retrieved 10-Apr-2006.
- S2CID 21463708.
- ISBN 978-0-8138-1217-5
- ^ "Cloning and substrate specificity of a human phenol UDP glucuronosyltransferase expressed in COS-7 cells". David Harding, Sylvie Fournel-Gigleux, Michael R. Jackson and Brian Burchell, Proc. Natl. Acad. Sci. USA, November 1988, Volume 85, pp. 8381–8385, (abstract)
- PMID 1339448.
Books
- Biochemistry of phenolic compounds, by J. B. Harborne, 1964, Academic Press (Google Books)
- Plant phenolics, by Pascal Ribéreau-Gayon, 1972, Oliver and Boyd Editions (Google Books, ISBN 9780050025123)
- The Biochemistry of plant phenolics, by C. F. van Sumere and P. J. Lea, Phytochemical Society of Europe, 1985, Clarendon Press (Google Books, ISBN 9780198541707)
- Biochemistry of Phenolic Compounds, by Wilfred Vermerris and Ralph Nicholson, 2006, Springer (Google book)
External links
Databases
- Phenol-Explorer (phenol-explorer.eu), a database dedicated to phenolics found in food by Augustin Scalbert, INRA Clermont-Ferrand, Unité de Nutrition Humaine (Human food unit)
- Phenols at ChEBI (Chemical Entities of Biological Interest)
- ChEMBLdb, a database of bioactive drug-like small molecules by the European Bioinformatics Institute
- Foodb, a database of compounds found in food
