User:Dwong527/sandbox
| Phosphoglycerate kinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Phosphoglycerate kinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| |||||||||
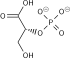
Phosphoglycerate kinase (
In humans, two isozymes of PGK have been so far identified, PGK1 and PGK2. The isoenzymes have 87-88% identical amino acid sequence identity and though they are structurally and functionally similar, they have different localizations: PGK2, encoded by an autosomal gene, is unique to meiotic and postmeiotic spermatogenic cells, while PGK1, encoded on the X-chromosome, is ubiquitously expressed in all cells.[2]
Biological function

PGK is present in all living organisms as one of the two ATP-generating enzymes in glycolysis. In the gluconeogenic pathway, PGK catalyzes the reverse reaction. Under biochemical
In the
PGK has been reported to exhibit thiol
The human isozyme PGK2, which is only expressed during spermatogenesis, was shown to be essential for sperm function in mice.[4]
Structure
Overview
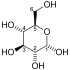
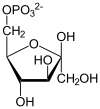
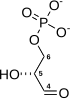
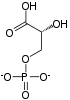
PGK is found in all living organisms and its sequence has been highly conserved throughout evolution. The enzyme exists as a 415-residue monomer containing two nearly equal-sized domains that correspond to the N- and C-termini of the protein.[5] 3-phosphoglycerate (3-PG) binds to the N-terminal, while the nucleotide substrates, MgATP or MgADP, bind to the C-terminal domain of the enzyme. This extended two-domain structure is associated with large-scale 'hinge-bending' conformational changes, similar to those found in hexokinase.[6] The two proteins of the domain are separated by a cleft and linked by two alpha-helices.[2] At the core of each domain is a 6-stranded parallel beta-sheet surrounded by alpha helices. The two lobes are capable of folding independently, consistent with the presence of intermediates on the folding pathway with a single domain folded.[7][8] Though the binding of either substrate triggers a conformational change, only the concerted binding of both substrates allows domain closure then transfer of the phosphoryl group.[2]
The enzyme has a tendency to exist in the open conformation with short periods of closure and catalysis, which allow for rapid diffusion of substrate and products through the binding sites; the open conformation of PGK is more conformationally stable due to the exposure of a hydrophobic region of the protein upon domain closure.[7]
Role of magnesium
Magnesium ions are normally complexed to the phosphate groups the nucleotide substrates of PGK. It is known that in the absence of magnesium, no enzyme activity occurs. [9] The The bivalent metal assists the enzyme ligands in shielding the bound phosphate group's negative charges, allowing the nucleophilic attack to occur; this charge-stabilization is a typical characteristic of phospho-transfer reaction.[10] It is theorized that the ion may also encourage domain closure when PGK has bound both substrates.[9]
Mechanism

Without either substrate bound, PGK exists in an "open" conformation. After both the triose and nucleotide substrates are bound to the N- and C-terminal domains, respectively, an extensive hinge-bending motion occurs, bringing the domains and their bound substrates into close proximity and leading to a "closed" conformation. The Lys219 on the enzyme guides the phosphate group to the substrate.
In the glycolytic pathyway, 1,3-BPG is the phosphate donor and has a high phosphoryl-transfer potential. The PGK-catalyzed transfer of the phosphoryl group from 1,3-BPG to ADP to yield ATP is powered by the energy from the carbon-oxidation reaction of the previous glycolytic step (converting glyceraldehyde 3-phosphate to 3-phosphoglycerate).
Regulation
The enzyme is activated by low concentrations of various multivalent anions, such as pyrophosphate, sulfate, phosphate, and citrate. High concentrations of MgATP and 3-PG activates PGK, while Mg2+ at high concentrations non-competitively inhibits the enzyme. [13] PGK activity is inhibited by salicylates, which appear to mimic the enzyme's nucleotide substrate.[14]
Macromolecular crowding has been shown to increase PGK activity in both computer simluations and in vitro environments simulating a cell interior; as a result of crowding, the enzyme becomes more enyzmatically active and more compact.[5]
Disease relevance
Phosphoglycerate kinase (PGK) deficiency is an X-linked recessive trait associated with
PGK1 overexpression has been associated with
Human isozymes
|
| ||||||||||||||||||||||||||||||
References
- ^ PMID 6765200.)
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link - ^ PMID 22348148.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 11130727.
- PMID 19759366.)
{{cite journal}}: Check date values in:|date=and|year=/|date=mismatch (help); Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|month=ignored (help - ^ PMID 20921368.
- PMID 10593256.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 2124145.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 21349853.)
{{cite journal}}: CS1 maint: date and year (link) CS1 maint: multiple names: authors list (link - ^ PMID 23231058.
- PMID 20397725. Retrieved 6 March 2013.)
{{cite journal}}: Check date values in:|date=and|year=/|date=mismatch (help); Unknown parameter|month=ignored (help - PMID 450128.
- ^ "Phosphoglycerate kinase". Mechanism, Annotation and Classification In Enzymes (MACiE). Retrieved 4 March 2013.
- PMID 6030358.)
{{cite journal}}: CS1 maint: date and year (link - PMID 343818.
- PMID 6689547.
- ^ PMID 17222195.)
{{cite journal}}: CS1 maint: date and year (link - PMID 21223252.
- PMID 19688824.)
{{cite journal}}: Check date values in:|date=and|year=/|date=mismatch (help); Unknown parameter|month=ignored (help - PMID 11130727.
External links
- . GPnotebook https://www.gpnotebook.co.uk/simplepage.cfm?ID=-858783685.
{{cite web}}: Missing or empty|title=(help) - Phosphoglycerate+kinase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Illustration at arizona.edu