Drosophila melanogaster
| Drosophila melanogaster | |
|---|---|

| |
| Fruit fly feeding off a banana | |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Diptera |
| Family: | Drosophilidae |
| Genus: | Drosophila |
| Subgenus: | Sophophora |
| Species group: | melanogaster |
| Species subgroup: | melanogaster |
| Species complex: | melanogaster |
| Species: | D. melanogaster
|
| Binomial name | |
| Drosophila melanogaster | |


Drosophila melanogaster is a species of fly (an insect of the order Diptera) in the family Drosophilidae. The species is often referred to as the fruit fly or lesser fruit fly, or less commonly the "vinegar fly", "pomace fly",[a][4] or "banana fly".[5] In the wild, D. melanogaster are attracted to rotting fruit and fermenting beverages, and are often found in orchards, kitchens and pubs.
Starting with Charles W. Woodworth's 1901 proposal of the use of this species as a model organism,[6][7] D. melanogaster continues to be widely used for biological research in genetics, physiology, microbial pathogenesis, and life history evolution. As of 2017, six Nobel Prizes have been awarded to drosophilists for their work using the insect.[8][9]
Drosophila melanogaster is typically used in research owing to its rapid life cycle, relatively simple genetics with only four pairs of chromosomes, and large number of offspring per generation.[10] It was originally an African species, with all non-African lineages having a common origin.[11] Its geographic range includes all continents, including islands.[12] D. melanogaster is a common pest in homes, restaurants, and other places where food is served.[13]
Flies belonging to the family Tephritidae are also called "fruit flies". This can cause confusion, especially in the Mediterranean, Australia, and South Africa, where the Mediterranean fruit fly Ceratitis capitata is an economic pest.
Etymology
The term "Drosophila", meaning "dew-loving", is a modern scientific Latin adaptation from Greek words δρόσος, drósos, "dew", and φιλία, philía, "lover". The term "melanogaster" meaning "black-belly", comes from Ancient Greek μέλας, mélas, “black”, and γᾰστήρ, gastḗr, "belly".
Physical appearance

Drosophila melanogaster can be distinguished from related species by the following combination of features: gena ~1/10 diameter of eye at greatest vertical height; wing hyaline and with costal index 2.4; male protarsus with a single row of ~12
Drosophila melanogaster flies can sense air currents with the hairs on their backs. Their eyes are sensitive to slight differences in light intensity and will instinctively fly away when a shadow or other movement is detected.[18]
Lifecycle and reproduction

Under optimal growth conditions at 25 °C (77 °F), the D. melanogaster lifespan is about 50 days from egg to death.
Drosophila melanogaster, commonly known as the fruit fly, has been a significant model organism in embryonic development research. Many of its genes that regulate embryonic development and their mechanisms of action have been crucial in understanding the fundamental principles of embryonic development regulation in many multicellular organisms, including humans. Here are some important genes regulating embryonic development in Drosophila melanogaster and their modes of action:
Maternal genes: These genes are encoded in the female fruit fly and are present in the early stages of embryo development. They determine the embryo's main features and early development. For example, the gene called Bicoid regulates the formation of the embryo's anterior end, and its absence leads to an embryo lacking a head.
Zygotic genes: These genes are activated in later stages of embryo development when the fruit fly embryo begins to produce its own genetic products. For example, the hunchback gene regulates the formation of segments in the embryo.
Homeotic genes: This gene family regulates segmentation and axial patterning in development. They act as regulatory factors that determine cell fate in embryonic development. For example, the gene called Antennapedia regulates the formation of anterior limbs in the embryo.
Morphogens: These are molecules that form gradients in embryonic development and regulate cell fate depending on their position in the gradient. For example, the Hedgehog morphogen regulates the differentiation of segments and segment identity in the fruit fly embryo.
These genes and their modes of action form a complex regulatory network that guides the embryonic development of Drosophila melanogaster. They influence cell differentiation, segment formation, and axial patterning in the embryo, ultimately leading to the development of a fully formed adult fruit fly.
Males perform a sequence of five behavioral patterns to court females. First, males orient themselves while playing a courtship song by horizontally extending and vibrating their wings. Soon after, the male positions himself at the rear of the female's abdomen in a low posture to tap and lick the female genitalia. Finally, the male curls his abdomen and attempts copulation. Females can reject males by moving away, kicking, and extruding their ovipositor.
D. melanogaster is often used for
Meiosis
Meiotic recombination in D. melanogaster appears to be employed in repairing damage in germ-line DNA as indicated by the findings that meiotic recombination is induced by the DNA damaging agents ultraviolet light [36] and mitomycin C[37].
Females

Females become receptive to courting males about 8–12 hours after emergence.[38] Specific neuron groups in females have been found to affect copulation behavior and mate choice. One such group in the abdominal nerve cord allows the female fly to pause her body movements to copulate.[31] Activation of these neurons induces the female to cease movement and orient herself towards the male to allow for mounting. If the group is inactivated, the female remains in motion and does not copulate. Various chemical signals such as male pheromones often are able to activate the group.[31]
Also, females exhibit mate choice copying. When virgin females are shown other females copulating with a certain type of male, they tend to copulate more with this type of male afterwards than naïve females (which have not observed the copulation of others). This behavior is sensitive to environmental conditions, and females copulate less in bad weather conditions.[39]
Males
This section needs additional citations for verification. (October 2015) |
D. melanogaster males exhibit a strong reproductive learning curve. That is, with sexual experience, these flies tend to modify their future mating behavior in multiple ways. These changes include increased selectivity for courting only intraspecifically, as well as decreased courtship times.
Sexually naïve D. melanogaster males are known to spend significant time courting interspecifically, such as with D. simulans flies. Naïve D. melanogaster will also attempt to court females that are not yet sexually mature, and other males. D. melanogaster males show little to no preference for D. melanogaster females over females of other species or even other male flies. However, after D. simulans or other flies incapable of copulation have rejected the males' advances, D. melanogaster males are much less likely to spend time courting nonspecifically in the future. This apparent learned behavior modification seems to be evolutionarily significant, as it allows the males to avoid investing energy into futile sexual encounters.[40]
In addition, males with previous sexual experience modify their courtship dance when attempting to mate with new females—the experienced males spend less time courting, so have lower mating latencies, meaning that they are able to reproduce more quickly. This decreased mating latency leads to a greater mating efficiency for experienced males over naïve males.[41] This modification also appears to have obvious evolutionary advantages, as increased mating efficiency is extremely important in the eyes of natural selection.
Polygamy
Both male and female D. melanogaster flies act polygamously (having multiple sexual partners at the same time).[42] In both males and females, polygamy results in a decrease in evening activity compared to virgin flies, more so in males than females.[42] Evening activity consists of those in which the flies participate other than mating and finding partners, such as finding food.[43] The reproductive success of males and females varies, because a female only needs to mate once to reach maximum fertility.[43] Mating with multiple partners provides no advantage over mating with one partner, so females exhibit no difference in evening activity between polygamous and monogamous individuals.[43] For males, however, mating with multiple partners increases their reproductive success by increasing the genetic diversity of their offspring.[43] This benefit of genetic diversity is an evolutionary advantage because it increases the chance that some of the offspring will have traits that increase their fitness in their environment.
The difference in evening activity between polygamous and monogamous male flies can be explained with courtship. For polygamous flies, their reproductive success increases by having offspring with multiple partners, and therefore they spend more time and energy on courting multiple females.[43] On the other hand, monogamous flies only court one female, and expend less energy doing so.[43] While it requires more energy for male flies to court multiple females, the overall reproductive benefits it produces has kept polygamy as the preferred sexual choice.[43]
The mechanism that affects courtship behavior in Drosophila is controlled by the oscillator neurons DN1s and LNDs.
Model organism in genetics
D. melanogaster remains one of the most studied
History of use in genetic analysis
D. melanogaster was among the first
Thomas Hunt Morgan began using fruit flies in experimental studies of heredity at Columbia University in 1910 in a laboratory known as the Fly Room. The Fly Room was cramped with eight desks, each occupied by students and their experiments. They started off experiments using milk bottles to rear the fruit flies and handheld lenses for observing their traits. The lenses were later replaced by microscopes, which enhanced their observations. Morgan and his students eventually elucidated many basic principles of heredity, including sex-linked inheritance, epistasis, multiple alleles, and gene mapping.[45]
D. melanogaster had historically been used in laboratories to study genetics and patterns of inheritance. However, D. melanogaster also has importance in environmental mutagenesis research, allowing researchers to study the effects of specific environmental mutagens.[46]
Reasons for use in laboratories

There are many reasons the fruit fly is a popular choice as a model organism:
- Its care and culture require little equipment, space, and expense even when using large cultures.
- It can be safely and readily anesthetized (usually with ether, carbon dioxide gas, by cooling, or with products such as FlyNap).
- Its morphology is easy to identify once anesthetized.
- It has a short generation time (about 10 days at room temperature), so several generations can be studied within a few weeks.
- It has a high fecundity (females lay up to 100 eggs per day, and perhaps 2000 in a lifetime).[10]
- Males and females are readily distinguished, and virgin females can be easily identified by their light-colored, translucent abdomen, facilitating genetic crossing.
- The mature larva has giant chromosomes in the salivary glands called polytene chromosomes, "puffs", which indicate regions of transcription, hence gene activity. The under-replication of rDNA occurs resulting in only 20% of DNA compared to the brain. Compare to the 47%, less rDNA in Sarcophaga barbata ovaries.
- It has only four pairs of chromosomes – three autosomes, and one pair of sex chromosomes.
- Males do not show meiotic recombination, facilitating genetic studies.
- Recessive lethal "lethal allelesin a heterozygous state without recombination due to multiple inversions in the balancer.
- The development of this organism—from fertilized egg to mature adult—is well understood.
- Genetic transformation techniques have been available since 1987. One approach of inserting foreign genes into the Drosophila genome involves P elements. The transposable P elements, also known as transposons, are segments of bacterial DNA that are transferred into the fly genome. Transgenic flies have already contributed to many scientific advances, e.g., modeling such human diseases as Parkinson's, neoplasia, obesity, and diabetes.
- Its complete genome was sequenced and first published in 2000.[47]
- Sexual mosaics can be readily produced, providing an additional tool for studying the development and behavior of these flies.[48]
Genetic markers

Genetic markers are commonly used in Drosophila research, for example within balancer chromosomes or P-element inserts, and most phenotypes are easily identifiable either with the naked eye or under a microscope. In the list of a few common markers below, the allele symbol is followed by the name of the gene affected and a description of its phenotype. (Note: Recessive alleles are in lower case, while dominant alleles are capitalised.)
- Cy1: Curly; the wings curve away from the body, flight may be somewhat impaired
- e1: Ebony; black body and wings (heterozygotes are also visibly darker than wild type)
- Sb1: Stubble; bristles are shorter and thicker than wild type
- w1: White; eyes lack pigmentation and appear white
- bw: Brown; eye color determined by various pigments combined.
- y1: Yellow; body pigmentation and wings appear yellow, the fly analog of albinism
Classic genetic mutations
Drosophila genes are traditionally named after the phenotype they cause when mutated. For example, the absence of a particular gene in Drosophila will result in a mutant embryo that does not develop a heart. Scientists have thus called this gene tinman, named after the Oz character of the same name.[50] Likewise changes in the Shavenbaby gene cause the loss of dorsal cuticular hairs in Drosophila sechellia larvae.[51] This system of nomenclature results in a wider range of gene names than in other organisms.
- b: black- The black mutation was discovered in 1910 by Thomas Hunt Morgan.[52] The black mutation results in a darker colored body, wings, veins, and segments of the fruit fly's leg.[53] This occurs due to the fly's inability to create beta-alanine, a beta amino acid.[52] The phenotypic expression of this mutation varies based on the genotype of the individual; for example, whether the specimen is homozygotic or heterozygotic results in a darker or less dark appearance.[53] This genetic mutation is x-linked recessive.[54]
- bw: brown- The brown eye mutation results from inability to produce or synthesize pteridine (red) pigments, due to a point mutation on chromosome II.[55] When the mutation is homozygous, the pteridine pigments are unable to be synthesized because in the beginning of the pteridine pathway, a defective enzyme is being coded by homozygous recessive genes.[56][unreliable source?] In all, mutations in the pteridine pathway produces a darker eye color, hence the resulting color of the biochemical defect in the pteridine pathway being brown.
- m: miniature- One of the first records of the miniature mutation of wings was also made by Thomas Hunt Morgan in 1911. He described the wings as having a similar shape as the wild-type phenotype. However, their miniature designation refers to the lengths of their wings, which do not stretch beyond their body and, thus, are notably shorter than the wild-type length. He also noted its inheritance is connected to the sex of the fly and could be paired with the inheritance of other sex-determined traits such as white eyes.[57] The wings may also demonstrate other characteristics deviant from the wild-type wing, such as a duller and cloudier color.[58] Miniature wings are 1.5x shorter than wild-type but are believed to have the same number of cells. This is due to the lack of complete flattening by these cells, making the overall structure of the wing seem shorter in comparison. The pathway of wing expansion is regulated by a signal-receptor pathway, where the neurohormone bursicon interacts with its complementary G protein-coupled receptor; this receptor drives one of the G-protein subunits to signal further enzyme activity and results in development in the wing, such as apoptosis and growth.[59]
- se: sepia- The eye color of the sepia mutant is
- v: vermilion- The vermilion mutants cannot produce the brown ommochromes leaving the red drosopterins so that the eyes are vermilion colored (a radiant red) compared to a wild-type D. melanogaster. The vermilion mutation is sex-linked and recessive. The gene that is defect lies on the X chromosome.[64] The brown ommochromes are synthesised from kynurenine, which is made from tryptophane. Vermilion flies cannot convert tryptophane into kynurenine and thus cannot make ommochromes, either.[64] Vermilion mutants live longer than wild-type flies. This longer life span may be associated with the reduced amount of tryptophan converted to kynurenine in vermilion flies.[65]

- vg: vestigial- A spontaneous mutation, discovered in 1919 by Thomas Morgan and Calvin Bridges. Vestigial wings are those not fully developed and that have lost function. Since the discovery of the vestigial gene in Drosophila melanogaster, there have been many discoveries of the vestigial gene in other vertebrates and their functions within the vertebrates.[66] The vestigial gene is considered to be one of the most important genes for wing formation, but when it becomes over expressed the issue of ectopic wings begin to form.[67] The vestigial gene acts to regulate the expression of the wing imaginal discs in the embryo and acts with other genes to regulate the development of the wings. A mutated vestigial allele removes an essential sequence of the DNA required for correct development of the wings.[68]
- w: white- Drosophila melanogaster wild type typically expresses a brick red eye color. The white eye mutation in fruit flies is caused due to the absence of two pigments associated with red and brown eye colors; peridines (red) and ommochromes (brown).[61] In January 1910, Thomas Hunt Morgan first discovered the white gene and denoted it as w. The discovery of the white-eye mutation by Morgan brought about the beginnings of genetic experimentation and analysis of Drosophila melanogaster. Hunt eventually discovered that the gene followed a similar pattern of inheritance related to the meiotic segregation of the X chromosome. He discovered that the gene was located on the X chromosome with this information. This led to the discovery of sex-linked genes and also to the discovery of other mutations in Drosophila melanogaster.[69] The white-eye mutation leads to several disadvantages in flies, such as a reduced climbing ability, shortened life span, and lowered resistance to stress when compared to wild type flies.[70] Drosophila melanogaster has a series of mating behaviors that enable them to copulate within a given environment and therefore contribute to their fitness. After Morgan's discovery of the white-eye mutation being sex-linked, a study led by Sturtevant (1915) concluded that white-eyed males were less successful than wild-type males in terms of mating with females.[71] It was found that the greater the density in eye pigmentation, the greater the success in mating for the males of Drosophila melanogaster.[71]
- y: yellow- The yellow gene is a

Genome
 D. melanogaster chromosomes to scale with megabase-pair references oriented as in the National Center for Biotechnology Information database, centimorgan distances are approximate and estimated from the locations of selected mapped loci. | |
| NCBI genome ID | 47 |
|---|---|
| Ploidy | diploid |
| Number of chromosomes | 8 |
| Year of completion | 2015 |
The
There are three transferrin orthologs, all of which are dramatically divergent from those known in chordate models.[78]
Similarity to humans
A March 2000 study by National Human Genome Research Institute comparing the fruit fly and human genome estimated that about 60% of genes are conserved between the two species.[79] About 75% of known human disease genes have a recognizable match in the genome of fruit flies,[80] and 50% of fly protein sequences have mammalian homologs [citation needed]. An online database called Homophila is available to search for human disease gene homologues in flies and vice versa.[81]
Drosophila is being used as a genetic model for several human diseases including the neurodegenerative disorders
Development
The life cycle of this insect has four stages: fertilized egg, larva, pupa, and adult.[12]
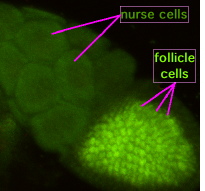
During oogenesis, cytoplasmic bridges called "ring canals" connect the forming oocyte to nurse cells. Nutrients and developmental control molecules move from the nurse cells into the oocyte. In the figure to the left, the forming oocyte can be seen to be covered by follicular support cells.
After fertilization of the oocyte, the early embryo (or syncytial embryo) undergoes rapid DNA replication and 13 nuclear divisions until about 5000 to 6000 nuclei accumulate in the unseparated cytoplasm of the embryo. By the end of the eighth division, most nuclei have migrated to the surface, surrounding the yolk sac (leaving behind only a few nuclei, which will become the yolk nuclei). After the 10th division, the pole cells form at the posterior end of the embryo, segregating the germ line from the syncytium. Finally, after the 13th division, cell membranes slowly invaginate, dividing the syncytium into individual somatic cells. Once this process is completed, gastrulation starts.[86]
Nuclear division in the early Drosophila embryo happens so quickly, no proper checkpoints exist, so mistakes may be made in division of the DNA. To get around this problem, the nuclei that have made a mistake detach from their centrosomes and fall into the centre of the embryo (yolk sac), which will not form part of the fly.
The gene network (transcriptional and protein interactions) governing the early development of the fruit fly embryo is one of the best understood gene networks to date, especially the patterning along the anteroposterior (AP) and dorsoventral (DV) axes (See under morphogenesis).[86]
The embryo undergoes well-characterized morphogenetic movements during gastrulation and early development, including germ-band extension, formation of several furrows, ventral invagination of the mesoderm, and posterior and anterior invagination of endoderm (gut), as well as extensive body segmentation until finally hatching from the surrounding cuticle into a first-instar larva.
During larval development, tissues known as imaginal discs grow inside the larva. Imaginal discs develop to form most structures of the adult body, such as the head, legs, wings, thorax, and genitalia. Cells of the imaginal disks are set aside during embryogenesis and continue to grow and divide during the larval stages—unlike most other cells of the larva, which have differentiated to perform specialized functions and grow without further cell division. At metamorphosis, the larva forms a pupa, inside which the larval tissues are reabsorbed and the imaginal tissues undergo extensive morphogenetic movements to form adult structures.
Developmental plasticity
Temperature is one of the most pervasive factors influencing arthropod development. In Drosophila melanogaster temperature-induced developmental plasticity can be beneficial and/or detrimental.[93][94] Most often lower developmental temperatures reduce growth rates which influence many other physiological factors.[95] For example, development at 25 °C increases walking speed, thermal performance breadth, and territorial success, while development at 18 °C increases body mass, wing size, all of which are tied to fitness.[90][93] Moreover, developing at certain low temperatures produces proportionally large wings which improve flight and reproductive performance at similarly low temperatures (See acclimation).[96]
While certain effects of developmental temperature, like body size, are irreversible in ectotherms, others can be reversible.[88][97] When Drosophila melanogaster develop at cold temperatures they will have greater cold tolerance, but if cold-reared flies are maintained at warmer temperatures their cold tolerance decreases and heat tolerance increases over time.[97][98] Because insects typically only mate in a specific range of temperatures, their cold/heat tolerance is an important trait in maximizing reproductive output.[99]
While the traits described above are expected to manifest similarly across sexes, developmental temperature can also produce sex-specific effects in D. melanogaster adults.
- Females-
- Males- Stressful developmental temperatures will cause sterility in D. melanogaster males; although the upper temperature limit can be increased by maintaining strains at high temperatures (See acclimation).[94] Male sterility can be reversible if adults are returned to an optimal temperature after developing at stressful temperatures.[103] Male flies are smaller and more successful at defending food/oviposition sites when reared at 25 °C versus 18 °C; thus smaller males will have increased mating success and reproductive output.[90]
Sex determination
Drosophila flies have both X and Y chromosomes, as well as
| X Chromosomes | Autosomes | Ratio of X:A | Sex |
|---|---|---|---|
| XXXX | AAAA | 1 | Normal Female |
| XXX | AAA | 1 | Normal Female |
| XXY | AA | 1 | Normal Female |
| XXYY | AA | 1 | Normal Female |
| XX | AA | 1 | Normal Female |
| XY | AA | 0.50 | Normal Male |
| X | AA | 0.50 | Normal Male (sterile) |
| XXX | AA | 1.50 | Metafemale |
| XXXX | AAA | 1.33 | Metafemale |
| XX | AAA | 0.66 | Intersex
|
| X | AAA | 0.33 | Metamale |
Three major genes are involved in determination of Drosophila sex. These are sex-lethal, sisterless, and deadpan. Deadpan is an autosomal gene which inhibits sex-lethal, while sisterless is carried on the X chromosome and inhibits the action of deadpan. An AAX cell has twice as much deadpan as sisterless, so sex-lethal will be inhibited, creating a male. However, an AAXX cell will produce enough sisterless to inhibit the action of deadpan, allowing the sex-lethal gene to be transcribed to create a female.
Later, control by deadpan and sisterless disappears and what becomes important is the form of the sex-lethal gene. A secondary promoter causes transcription in both males and females. Analysis of the
Presence or absence of functional sex-lethal proteins now go on to affect the transcription of another protein known as doublesex. In the absence of sex-lethal, doublesex will have the fourth exon removed and be translated up to and including exon 6 (DSX-M[ale]), while in its presence the fourth exon which encodes a stop codon will produce a truncated version of the protein (DSX-F[emale]). DSX-F causes transcription of Yolk proteins 1 and 2 in somatic cells, which will be pumped into the oocyte on its production.
Immunity
The D. melanogaster immune system can be divided into two responses: humoral and cell-mediated. The former is a systemic response mediated in large part through the
The immune response to infection can involve up to 2,423 genes, or 13.7% of the genome. Although the fly's transcriptional response to microbial challenge is highly specific to individual pathogens, Drosophila differentially expresses a core group of 252 genes upon infection with most bacteria. This core group of genes is associated with gene ontology categories such as antimicrobial response, stress response, secretion, neuron-like, reproduction, and metabolism among others.[107][108] Drosophila also possesses several immune mechanisms to both shape the microbiota and prevent excessive immune responses upon detection of microbial stimuli. For instance, secreted PGRPs with amidase activity scavenge and degrade immunostimulatory DAP-type PGN in order to block Imd activation.[109]
Unlike mammals, Drosophila have
JAK-STAT signalling
Multiple elements of the Drosophila JAK-STAT signalling pathway bear direct homology to human JAK-STAT pathway genes. JAK-STAT signalling is induced upon various organismal stresses such as heat stress, dehydration, or infection. JAK-STAT induction leads to the production of a number of stress response proteins including Thioester-containing proteins (TEPs),[112] Turandots,[113] and the putative antimicrobial peptide Listericin.[114] The mechanisms through which many of these proteins act is still under investigation. For instance, the TEPs appear to promote phagocytosis of Gram-positive bacteria and the induction of the toll pathway. As a consequence, flies lacking TEPs are susceptible to infection by toll pathway challenges.[112]
The cellular response to infection
Circulating hemocytes are key regulators of infection. This has been demonstrated both through genetic tools to generate flies lacking hemocytes, or through injecting microglass beads or lipid droplets that saturate hemocyte ability to phagocytose a secondary infection.
Behavioral genetics and neuroscience
In 1971,
Since then, Benzer and others have used behavioral screens to isolate genes involved in vision, olfaction, audition, learning/memory, courtship, pain, and other processes, such as longevity.
Following the pioneering work of Alfred Henry Sturtevant[121] and others, Benzer and colleagues[48] used sexual mosaics to develop a novel fate mapping technique. This technique made it possible to assign a particular characteristic to a specific anatomical location. For example, this technique showed that male courtship behavior is controlled by the brain.[48] Mosaic fate mapping also provided the first indication of the existence of pheromones in this species.[122] Males distinguish between conspecific males and females and direct persistent courtship preferentially toward females thanks to a female-specific sex pheromone which is mostly produced by the female's tergites.
The first learning and memory mutants (dunce, rutabaga, etc.) were isolated by William "Chip" Quinn while in Benzer's lab, and were eventually shown to encode components of an intracellular signaling pathway involving cyclic AMP, protein kinase A, and a transcription factor known as CREB. These molecules were shown to be also involved in synaptic plasticity in Aplysia and mammals.[123]
The Nobel Prize in Physiology or Medicine for 2017 was awarded to Jeffrey C. Hall, Michael Rosbash, Michael W. Young for their works using fruit flies in understanding the "molecular mechanisms controlling the circadian rhythm".[124]
Male flies sing to the females during courtship using their wings to generate sound, and some of the genetics of sexual behavior have been characterized. In particular, the
Flies use a modified version of Bloom filters to detect novelty of odors, with additional features including similarity of novel odor to that of previously experienced examples, and time elapsed since previous experience of the same odor.[128]
Aggression
As with most insects, aggressive behaviors between male flies commonly occur in the presence of courting a female and when competing for resources. Such behaviors often involve raising wings and legs towards the opponent and attacking with the whole body.[129] Thus, it often causes wing damage, which reduces their fitness by removing their ability to fly and mate.[130]
Acoustic communication
In order for aggression to occur, male flies produce sounds to communicate their intent. A 2017 study found that songs promoting aggression contain pulses occurring at longer intervals.[131] RNA sequencing from fly mutants displaying over-aggressive behaviors found more than 50 auditory-related genes (important for transient receptor potentials, Ca2+ signaling, and mechanoreceptor potentials) to be upregulated in the AB neurons located in Johnston's organ.[131] In addition, aggression levels were reduced when these genes were knocked out via RNA interference.[131] This signifies the major role of hearing as a sensory modality in communicating aggression.
Pheromone signaling
Other than hearing, another sensory modality that regulates aggression is pheromone signaling, which operates through either the olfactory system or the gustatory system depending on the pheromone.[132] An example is cVA, an anti-aphrodisiac pheromone used by males to mark females after copulation and to deter other males from mating.[133] This male-specific pheromone causes an increase in male-male aggression when detected by another male's gustatory system.[132] However, upon inserting a mutation that makes the flies irresponsive to cVA, no aggressive behaviors were seen.[134] This shows how there are multiple modalities for promoting aggression in flies.
Competition for food
Specifically, when competing for food, aggression occurs based on amount of food available and is independent of any social interactions between males.[135] Specifically, sucrose was found to stimulate gustatory receptor neurons, which was necessary to stimulate aggression.[135] However, once the amount of food becomes greater than a certain amount, the competition between males lowers.[135] This is possibly due to an over-abundance of food resources. On a larger scale, food was found to determine the boundaries of a territory since flies were observed to be more aggressive at the food's physical perimeter.
Effect of sleep deprivation
However, like most behaviors requiring arousal and wakefulness, aggression was found to be impaired via sleep deprivation. Specifically, this occurs through the impairment of Octopamine and dopamine signaling, which are important pathways for regulating arousal in insects.[136][137] Due to reduced aggression, sleep-deprived male flies were found to be disadvantaged at mating compared to normal flies.[137] However, when octopamine agonists were administered upon these sleep-deprived flies, aggression levels were seen to be increased and sexual fitness was subsequently restored.[137] Therefore, this finding implicates the importance of sleep in aggression between male flies.
Vision

The
Each photoreceptor cell consists of two main sections, the cell body and the
Opsins and spectral sensitivity
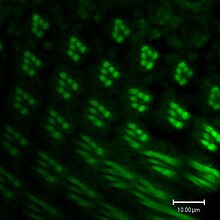
The genome of Drosophila encodes seven opsins,[140] five of those are expressed in the omatidia of the eye. The photoreceptor cells R1-R6 express the opsin Rh1,[141] which absorbs maximally blue light (around 480 nm),[142][143][144] however the R1-R6 cells cover a broader range of the spectrum than an opsin would allow due to a sensitising pigment[145][146] that adds two sensitivity maxima in the UV-range (355 and 370 nm).[144] The R7 cells come in two types with yellow and pale rhabdomeres (R7y and R7p).[147][148] The pale R7p cells express the opsin Rh3,[149][150] which maximally absorbs UV-light (345 nm).[151] The R7p cells are strictly paired with the R8p cells that express Rh5,[150] which maximally absorbs violet light (437 nm).[144] The other, the yellow R7y cells express a blue-absorbing screening pigment[147] and the opsin Rh4,[152] which maximally absorbs UV-light (375 nm).[151] The R7y cells are strictly paired with R8y cells that express Rh6,[153] which maximally absorbs UV-light (508 nm).[144] In a subset of omatidia both R7 and R8 cells express the opsin Rh3.[150]
However, these absorption maxima of the opsins where measured in white eyed flies without screening pigments (Rh3-Rh6),[151][144] or from the isolated opsin directly (Rh1).[142] Those pigments reduce the light that reaches the opsins depending on the wavelength. Thus in fully pigmented flies, the effective absorption maxima of opsins differs and thus also the sensitivity of their photoreceptor cells. With screening pigment, the opsin Rh3 is short wave shifted from 345 nm[b] to 330 nm and Rh4 from 375 nm to 355 nm. Whether screening pigment is present does not make a practical difference for the opsin Rh5 (435 nm and 437 nm), while the opsin R6 is long wave shifted by 92 nm from 508 nm to 600 nm.[139]
Additionally of the opsins of the eye, Drosophila has two more opsins: The ocelli express the opsin Rh2,[154][155] which maximally absorbs violet light (~420 nm).[155] And the opsin Rh7, which maximally absorbs UV-light (350 nm) with an unusually long wavelength tail up to 500 nm. The long tail disappears if a lysine at position 90 is replaced by glutamic acid. This mutant then absorbs maximally violet light (450 nm).[156] The opsin Rh7 entrains with cryptochrome the circadian rhythm of Drosophila to the day-night-cycle in the central pacemaker neurons.[157]
Each Drosophila opsin binds the carotenoid chromophore 11-cis-3-hydroxyretinal via a lysine.[158][159] This lysine is conserved in almost all opsins, only a few opsins have lost it during evolution.[160] Opsins without it are not light sensitive.[161][162][163] In particular, the Drosophila opsins Rh1, Rh4, and Rh7 function not only as photoreceptors, but also as chemoreceptors for aristolochic acid. These opsins still have the lysine like other opsins. However, if it is replaced by an arginine in Rh1, then Rh1 loses light sensitivity but still responds to aristolochic acid. Thus, the lysine is not needed for Rh1 to function as chemoreceptor.[162]
-
Spectral sensitivities of Drosophila melanogaster opsins in white eyed flies. The sensitivities of Rh3–R6 are modelled with opsin templates and sensitivity estimates from Salcedo et al. (1999).[144] The opsin Rh1 (redrawn from Salcedo et al.[144]) has a characteristic shape as it is coupled to a UV-sensitising pigment.
-
Normalized mean spectral sensitivity curves of Drosophila melanogaster opsins Rh1, Rh3, Rh4, Rh5, and Rh6 measured in their native photoreceptor cells in red eye flies with screening pigment. Each spectral curve is the average from six flies.
Phototransduction
As in
PLCβ hydrolyzes
Calcium binds to proteins such as calmodulin (CaM) and an eye-specific protein kinase C (PKC) known as InaC. These proteins interact with other proteins and have been shown to be necessary for shut off of the light response. In addition, proteins called arrestins bind metarhodopsin and prevent it from activating more Gq. A sodium-calcium exchanger known as CalX pumps the calcium out of the cell. It uses the inward sodium gradient to export calcium at a stoichiometry of 3 Na+/ 1 Ca++.[166]
TRP, InaC, and PLC form a signaling complex by binding a scaffolding protein called InaD. InaD contains five binding domains called PDZ domain proteins, which specifically bind the C termini of target proteins. Disruption of the complex by mutations in either the PDZ domains or the target proteins reduces the efficiency of signaling. For example, disruption of the interaction between InaC, the protein kinase C, and InaD results in a delay in inactivation of the light response.
Unlike vertebrate metarhodopsin, invertebrate metarhodopsin can be converted back into rhodopsin by absorbing a photon of orange light (580 nm).
About two-thirds of the Drosophila brain is dedicated to visual processing.[167] Although the spatial resolution of their vision is significantly worse than that of humans, their temporal resolution is around 10 times better.
Grooming
Drosophila are known to exhibit grooming behaviors that are executed in a predictable manner. Drosophila consistently begin a grooming sequence by using their front legs to clean the eyes, then the head and antennae. Using their hind legs, Drosophila proceed to groom their abdomen, and finally the wings and thorax. Throughout this sequence, Drosophila periodically rub their legs together to get rid of excess dust and debris that accumulates during the grooming process.[168]
Grooming behaviors have been shown to be executed in a suppression hierarchy. This means that grooming behaviors that occur at the beginning of the sequence prevent those that come later in the sequence from occurring simultaneously, as the grooming sequence consists of mutually exclusive behaviors.[169][170] This hierarchy does not prevent Drosophila from returning to grooming behaviors that have already been accessed in the grooming sequence.[169] The order of grooming behaviors in the suppression hierarchy is thought to be related to the priority of cleaning a specific body part. For example, the eyes and antennae are likely executed early on in the grooming sequence to prevent debris from interfering with the function of D. melanogaster's sensory organs.[169][170]
Walking

Like many other hexapod insects, Drosophila typically walk using a
Flight
Flies fly via straight sequences of movement interspersed by rapid turns called saccades.[178] During these turns, a fly is able to rotate 90° in less than 50 milliseconds.[178]
Characteristics of Drosophila flight may be dominated by the viscosity of the air, rather than the inertia of the fly body, but the opposite case with inertia as the dominant force may occur.[178] However, subsequent work showed that while the viscous effects on the insect body during flight may be negligible, the aerodynamic forces on the wings themselves actually cause fruit flies' turns to be damped viscously.[179]
Connectome
Drosophila is one of the few animals (C. elegans being another) where detailed neural circuits (a connectome) are available.
A high-level connectome, at the level of brain compartments and interconnecting tracts of neurons, exists for the full fly brain.[180] A version of this is available online.[181]
Detailed circuit-level connectomes exist for the lamina[182][183] and a medulla[184] column, both in the visual system of the fruit fly, and the alpha lobe of the mushroom body.[185]
In May 2017 a paper published in bioRxiv presented an electron microscopy image stack of the whole adult female brain at synaptic resolution. The volume is available for sparse tracing of selected circuits.[186][187] Since then, multiple datasets have been collected including a dense connectome of half the central brain of Drosophila in 2020,[188][189] and a dense connectome of the entire female adult nerve cord in 2021.[190] Generally, these datasets are acquired by sectioning the tissue (e.g. the brain) into thin sections (on order of ten or hundreds of nanometers). Each section is then imaged using an electron microscope and these images are stitched and aligned together to create a 3D image volume. The methods used in reconstruction and initial analysis of the such datasets followed.[191] Due to advancements in deep learning, automated methods for image segmentation have made large scale reconstruction providing dense reconstructions of all the neurites within the volume.[192] Furthermore, the resolution of electron microscopy illuminates ultrastructural variations between neurons as well as the location of individual synapses, thereby providing a wiring diagram of synaptic connectivity between all neurites within the given dataset.
In 2023, the complete map of a Drosophila larval brain at the synapse level, and an analysis of its architecture was published. The larval brain consists of 3016 neurons and 548,000 synaptic sites,[193] whereas the adult brain has about 150,000 neurons and 150 million synapses.
Misconceptions
Drosophila is sometimes referred to as a pest due to its tendency to live in human settlements where fermenting fruit is found. Flies may collect in homes, restaurants, stores, and other locations.[13] The name and behavior of this species of fly have led to the misconception that it is a biological security risk in Australia and elsewhere. While other "fruit fly" species do pose a risk, D. melanogaster is attracted to fruit that is already rotting, rather than causing fruit to rot.[194][195]
See also
- Animal testing on invertebrates
- Eating behavior in Insects (Measurement)
- Fruit flies in space
- Genetically modified insect
- Gynandromorphism
- JETLAG gene
- List of Drosophila databases
- Spätzle (gene)
- Time flies like an arrow; fruit flies like a banana
- Transgenesis
- Zebrafish – another widely used model organism in scientific research
Notes
- ^ "Vinegar fly" is preferred by a handful of recent publications as being a more accurate description than "fruit fly".[2][3][4]
- ^ Sharkey et al.[139] give the absorption maximum of Rh3 as 334 nm in their result section. However, in the introduction and the material and methods section they give it as 345 nm. For both values, they cite Feiler et al., who reported 345 nm only.[151] Therefore, this seems to be a mistake and they probably meant there 345 nm, too.
References
- ^ Meigen JW (1830). Systematische Beschreibung der bekannten europäischen zweiflügeligen Insekten. (Volume 6) (PDF) (in German). Schulz-Wundermann. Archived from the original (PDF) on February 9, 2012.
- ^ "Drosophila | insect genus". Encyclopedia Britannica. Retrieved October 30, 2021.
- ^ "Vinegar Flies". Penn State Extension. Retrieved October 30, 2021.
- ^ PMID 12242218.
- ^ Schilthuizen, Menno (April 28, 2015). "Semen's Chemical Cocktail Can Hijack a Mate's Brain". Discover. Retrieved September 11, 2023.
Even in the ejaculate of the lowly banana fly Drosophila melanogaster, researchers have identified no fewer than 133 different kinds of proteins.
- ^ T.H. Morgan's Nobel Prize biography mentioning C. W. Woodworth
- ISBN 978-0-9864105-3-6.
- ^ "Nobel Prizes". The Guardian. October 7, 2017.
- ^ "FruitFly-ResearchGate".
- ^ ISBN 978-1-884964-34-3. Retrieved July 1, 2009.
- PMID 15014160.
- ^ PMID 26041333.
- ^ a b "Vinegar Flies, Drosophila species, Family: Drosophilidae". Department of Entomology, College of Agricultural Sciences, Pennsylvania State University. 2017. Retrieved July 20, 2017.
- ^ PMID 9711556.
- ^ "FlyBase: A database of Drosophila genes and genomes". Genetics Society of America. 2009. Archived from the original on August 15, 2009. Retrieved August 11, 2009.
- S2CID 211570766.
- .
- ^ "Drosophila Melanogaster". Animal Diversity Web. 2000. Archived from the original on November 30, 2014. Retrieved August 11, 2009.
- PMID 23328955.
- ^ a b c d e f g Ashburner M, Thompson JN (1978). "The laboratory culture of Drosophila". In Ashburner M, Wright TRF (ed.). The genetics and biology of Drosophila. Vol. 2A. Academic Press. 1–81.
- ^ ISBN 978-0-87969-706-8.
- Indiana University: Basic Methods of Culturing Drosophila Archived 2006-09-01 at the Wayback Machine
- ^ JSTOR 1948580.
- S2CID 85129022.
- PMID 18314716.
- PMID 24194543.
- S2CID 85393769.
- PMID 20543131.
- ISBN 978-0-87893-250-4. Archived from the originalon February 7, 2007.
- ^ S2CID 4393369.
- ^ a b c d "Fruit fly research may reveal what happens in female brains during courtship, mating". Retrieved October 5, 2014.
- PMID 28439025.
- PMID 8756302.
- PMID 26378456.
- PMID 31518666.
- ^ Prudhommeau C, Proust J. UV irradiation of poplar cells of Drosophila melanogaster embryos. V. A study of the meiotic recombination in females with chromosomes of different structure. Mutat Res. 1974 Apr;23(1):63-6. PMID: 4209047
- ^ Schewe MJ, Suzuki DT, Erasmus U. The genetic effects of mitomycin C in Drosophila melanogaster. II. Induced meiotic recombination. Mutat Res. 1971 Jul;12(3):269-79. doi: 10.1016/0027-5107(71)90015-7. PMID: 5563942
- S2CID 83654824.
- .
- .
- PMID 24805129.
- ^ .
- ^ S2CID 7529509.
- ^ PMID 18103134.
- ^ ISBN 978-0-7167-8881-2.
- PMID 6790982.
- ^ PMID 10731132.
- ^ S2CID 4181921.
- . Retrieved June 28, 2021.
- PMID 8101173.
- PMID 24218640.
- ^ PMID 15878647.
- ^ a b "FlyBase Gene Report: Dmel\b". flybase.org. Retrieved March 26, 2019.
- S2CID 1210971.
- PMID 5961338.
- ^ "TEACHER REFERENCE PAGES-FLY EYE PIGMENTS LAB" (PDF). Archived from the original (PDF) on March 26, 2019. Retrieved March 26, 2019.
- PMID 17774436.
- ^ "FlyBase Gene Report: Dmel\m". flybase.org. Retrieved March 26, 2019.
- PMID 23745366.
- ^ PMID 16712527.
- ^ PMID 27558665.
- PMID 6438092.
- ^ "Inheritance Patterns in Drosophila Melanogaster". Retrieved March 26, 2019.
- ^ PMID 16589094.
- PMID 19941150.
- S2CID 16651247.
- PMID 28357056.
- PMID 1752439.
- PMID 20061564.
- PMID 29354028.
- ^ PMID 28794482.
- ^ "Gene:Dmel\y". Flybase.org. The FlyBase Consortium. Retrieved March 26, 2019.
- PMID 11934851.
- ^ PMID 3933004.
- ^ "NCBI (National Center for Biotechnology Information) Genome Database". Retrieved November 30, 2011.
- PMID 16751341.
- PMID 12433579.
- PMID 23552219.
- ^ "Background on Comparative Genomic Analysis". US National Human Genome Research Institute. December 2002.
- PMID 11381037.
- PMID 11752278.
- PMID 22974305.
- ISBN 978-0-12-802905-3.
- PMID 25421701.
- PMID 22350798.
- ^ PMID 12801722.
- PMID 15851679.
- ^ S2CID 9099834.
- S2CID 2991855.
- ^ S2CID 9124942.
- PMID 28927826.
- PMID 12770129.
- ^ PMID 28565273.
- ^ S2CID 23847613.
- PMID 12770407.
- PMID 18552301.
- ^ PMID 27353229.
- S2CID 1329035.
- PMID 23360477.
- ^ S2CID 6875815.
- PMID 31064855.
- PMID 27927623.
- S2CID 12465060.
- PMID 26710087.
- ISBN 978-0-87893-243-6.
- PMID 17201680.
- PMID 29394281.
- PMID 12032070.
- PMID 22118526.
- S2CID 119527642.
- PMID 32047838.
- ^ PMID 28874153.
- PMID 27871362.
- PMID 20348097.
- PMID 24409421.
- PMID 24631888.
- PMID 19890048.
- PMID 12930778.
- PMID 31735669.
- PMID 31358113.
- ^ Sturtevant AH (1929). "The claret mutant type of Drosophila simulans: a study of chromosome elimination and cell-lineage". Zeitschrift für Wissenschaftliche Zoologie. 135: 323–356.
- PMID 805823.
- ISBN 978-1-4398-2009-4.
- ^ "The 2017 Nobel Prize in Physiology or Medicine jointly to Jeffrey C. Hall, Michael Rosbash and Michael W. Young for their discoveries of molecular mechanisms controlling the circadian rhythm". Nobelprize.org. October 2, 2017. Retrieved October 5, 2017.
- PMID 23312520.
- PMID 23898199.
- ^ "Homosexuality Turned On and Off in Fruit Flies"
- PMID 30509984.
- PMID 22513455.
- PMID 29109180.
- ^ PMID 28115690.
- ^ PMID 24830032. Retrieved May 30, 2019.
- PMID 27484362.
- PMID 21516101.
- ^ PMID 25162609.
- PMID 22829591.
- ^ PMID 26216041.
- S2CID 4415605.
- ^ S2CID 215551298..
 Material was copied and adapted from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied and adapted from this source, which is available under a Creative Commons Attribution 4.0 International License - PMID 34270718.
- S2CID 45888026.
- ^ PMID 4213042.
- S2CID 5802525.
- ^ S2CID 17575850.
- S2CID 28890008.
- S2CID 12451748.
- ^ S2CID 40233531.
- S2CID 5846094.
- S2CID 1490332.
- ^ S2CID 18294965.
- ^ PMID 1403087.
- S2CID 17003459.
- S2CID 18368117.
- S2CID 4303934.
- ^ S2CID 4248264.
- S2CID 3276084.
- S2CID 4468254.
- S2CID 88980658.
- S2CID 24205801.
- PMID 35954284.
- S2CID 201420079.
- ^ PMID 32243853.
- S2CID 23060331.
- ^ S2CID 23861204.
- S2CID 140206989.
- PMID 15694324.
- S2CID 15785406.
- S2CID 53186674.
- ^ S2CID 53180462.
- ^ PMID 25139955.
- S2CID 4748395.
- S2CID 12965869.
- PMID 31250807.
- PMID 23038731.
- PMID 23326642.
- PMID 30274987.
- PMID 22575473.
- ^ S2CID 40952385. Archived from the original(PDF) on September 24, 2015.
- PMID 18055621.
- S2CID 17155338.
- ^ "FlyCircuit - A Database of Drosophila Brain Neurons". Retrieved August 30, 2013.
- S2CID 35301798.
- PMID 22119527.
- PMID 23925240.
- PMID 28718765.
- ^ "Entire Fruit Fly Brain Imaged with Electron Microscopy". The Scientist Magazine. Retrieved July 15, 2018.
- PMID 30033368.
- S2CID 213140797.
- ^ "Analysis tools for connectomics". HHMI.
- PMID 33400916.
- S2CID 215790785.
- bioRxiv 10.1101/2022.03.25.485816.
- PMID 36893230.
- ^ "Non pest species". Plant Health Australia. Retrieved September 19, 2017.
- ^ McEvey S (February 5, 2014). "Fruit Flies: A Case Of Mistaken Identity". Australian Museum. Retrieved September 19, 2017.
Further reading
- Kohler RE (1994). Lords of the Fly: Drosophila genetics and the experimental life. Chicago: University of Chicago Press. ISBN 978-0-226-45063-6.
- Gilbert SF (2000). Developmental Biology (6th ed.). Sunderland (MA): Sinauer Associates; 2000. ISBN 978-0-87893-243-6.
- Perrimon N, Bonini NM, Dhillon P (March 2016). "Fruit flies on the front line: the translational impact of Drosophila". Disease Models & Mechanisms. 9 (3): 229–31. PMID 26935101.
- Henderson M (April 8, 2010). "Row over fruit fly Drosophila melanogaster name bugs scientists". The Times. The Australian. Retrieved September 19, 2017.
External links
- "A quick and simple introduction to Drosophila melanogaster". Drosophila Virtual Library.
- "Drosophila Genomics Resource Center" – collects, maintains and distributes Drosophila DNA clones and cell lines.
- "Bloomington Drosophila Stock Center" – collects, maintains and distributes Drosophila melanogaster strains for research
- "FlyBase—A Database of Drosophila Genes & Genomes".
- "NCBI Map Viewer – Drosophila melanogaster".
- "Drosophila Virtual Library".
- "The Berkeley Drosophila Genome Project".
- "FlyMove". – video resources for Drosophila development
- "Drosophila Nomenclature—naming of genes". Archived from the original on October 8, 2011.
- View the Fruitfly genome on Ensembl
- View the dm6 genome assembly in the UCSC Genome Browser.
- Manchester Fly Facility – for the public Archived 2015-05-13 at the Wayback Machine from the University of Manchester
- The droso4schools website with school-relevant resources about Drosophila
- Part 1 of the "Small fly: BIG impact" educational videos explaining the history and importance of the model organism Drosophila.
- Part 2 of the "Small fly: BIG impact" educational videos explaining how research is carried out in Drosophila.
- "Inside the Fly Lab"—broadcast by WGBH and PBS, in the program series Curious, January 2008.
- "How a Fly Detects Poison" Archived 2013-01-13 at archive.today—WhyFiles.org article describes how the fruit fly tastes a larva-killing chemical in food.



![Spectral sensitivities of Drosophila melanogaster opsins in white eyed flies. The sensitivities of Rh3–R6 are modelled with opsin templates and sensitivity estimates from Salcedo et al. (1999).[144] The opsin Rh1 (redrawn from Salcedo et al.[144]) has a characteristic shape as it is coupled to a UV-sensitising pigment.](http://upload.wikimedia.org/wikipedia/commons/thumb/f/fc/Spectral_sensitivities_of_Drosophila_melanogaster_opsins_in_white_eyed_flies.png/750px-Spectral_sensitivities_of_Drosophila_melanogaster_opsins_in_white_eyed_flies.png)
