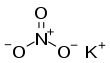Potassium nitrate

| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Potassium nitrate
| |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.028.926 | ||
| EC Number |
| ||
| E number | E252 (preservatives) | ||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1486 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| KNO3 | |||
| Molar mass | 101.1032 g/mol | ||
| Appearance | white solid | ||
| Odor | odorless | ||
| Density | 2.109 g/cm3 (16 °C) | ||
| Melting point | 334 °C (633 °F; 607 K) | ||
| Boiling point | 400 °C (752 °F; 673 K) (decomposes) | ||
| 133 g/1000 g water (0 °C) 316 g/1000 g water (20 °C) 383 g/1000 g water (25 °C) 2439 g/1000 g water (100 °C)[2] | |||
| Solubility | slightly soluble in ethanol soluble in glycerol, ammonia | ||
| Basicity (pKb) | 15.3[3] | ||
| −33.7·10−6 cm3/mol | |||
Refractive index (nD)
|
1.335, 1.5056, 1.5604 | ||
| Structure | |||
Orthorhombic, Aragonite
| |||
| Thermochemistry | |||
Heat capacity (C)
|
95.06 J/mol K | ||
Std enthalpy of (ΔfH⦵298)formation |
-494.00 kJ/mol | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Oxidant, harmful if swallowed, inhaled, or absorbed on skin. Causes irritation to skin and eye area. | ||
| GHS labelling: | |||
 
| |||
| H272, H315, H319, H335 | |||
| P102, P210, P220, P221, P280 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | non-flammable (oxidizer) | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
1901 mg/kg (oral, rabbit) 3750 mg/kg (oral, rat)[4] | ||
| Safety data sheet (SDS) | ICSC 0184 | ||
| Related compounds | |||
Other anions
|
Potassium nitrite | ||
Other cations
|
Lithium nitrate Sodium nitrate Rubidium nitrate Caesium nitrate | ||
Related compounds
|
Potassium sulfate Potassium chloride | ||
| Supplementary data page | |||
| Potassium nitrate (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Potassium nitrate is a
Major uses of potassium nitrate are in
Etymology
Potash, or potassium nitrate, because of its early and global use and production, has many names. The chemical potassium was first isolated by the chemist
The Arabs called it "Chinese snow" (
Historical production
From mineral sources
In Mauryan India saltpeter manufacturers formed the Nuniya & Labana
A purification process for potassium nitrate was outlined in 1270 by the chemist and engineer Hasan al-Rammah of Syria in his book al-Furusiyya wa al-Manasib al-Harbiyya (The Book of Military Horsemanship and Ingenious War Devices). In this book, al-Rammah describes first the purification of barud (crude saltpeter mineral) by boiling it with minimal water and using only the hot solution, then the use of potassium oxide (in the form of wood ashes) to remove calcium and magnesium by precipitation of their carbonates from this solution, leaving a solution of purified potassium nitrate, which could then be dried.[17] This was used for the manufacture of gunpowder and explosive devices. The terminology used by al-Rammah indicated the gunpowder he wrote about originated in China.[18]
At least as far back as 1845,
From caves
Major natural sources of potassium nitrate were the deposits crystallizing from cave walls and the accumulations of
Nitraries
Potassium nitrate was produced in a nitrary or "
Besides "Montepellusanus", during the thirteenth century (and beyond) the only supply of saltpeter across Christian Europe (according to "De Alchimia" in 3 manuscripts of Michael Scot, 1180–1236) was "found in Spain in Aragon in a certain mountain near the sea".[13]: 89, 311 [23]
In 1561, Elizabeth I, Queen of England and Ireland, who was at war with Philip II of Spain, became unable to import saltpeter (of which the Kingdom of England had no home production), and had to pay "300 pounds gold" to the German captain Gerrard Honrik for the manual "Instructions for making saltpeter to growe" (the secret of the "Feuerwerkbuch" -the nitraries-).[24]
Nitre bed
A nitre bed is a similar process used to produce nitrate from excrement. Unlike the leaching-based process of the nitrary, however, one mixes the excrements with soil and waits for soil microbes to convert amino-nitrogen into nitrates by nitrification. The nitrates are extracted from soil with water and then purified into saltpeter by adding wood ash. The process was discovered in the early 15th century and was very widely used until the Chilean mineral deposits were found.[25]
The Confederate side of the American Civil War had a significant shortage of saltpeter. As a result, the Nitre and Mining Bureau was set up to encourage local production, including by nitre beds and by providing excrement to government nitraries. On November 13, 1862, the government advertised in the Charleston Daily Courier for 20 or 30 "able bodied Negro men" to work in the new nitre beds at Ashley Ferry, S.C. The nitre beds were large rectangles of rotted manure and straw, moistened weekly with urine, "dung water", and liquid from privies, cesspools and drains, and turned over regularly. The National Archives published payroll records that account for more than 29,000 people compelled to such labor in the state of Virginia. The South was so desperate for saltpeter for gunpowder that one Alabama official reportedly placed a newspaper ad asking that the contents of chamber pots be saved for collection. In South Carolina, in April 1864, the Confederate government forced 31 enslaved people to work at the Ashley Ferry Nitre Works, outside Charleston.[26]
Perhaps the most exhaustive discussion of the niter-bed production is the 1862
French method
Swiss method
Joseph LeConte describes a process using only urine and not dung, referring to it as the Swiss method. Urine is collected directly, in a sandpit under a stable. The sand itself is dug out and leached for nitrates which are then converted to potassium nitrate using potash, as above.[28]
From nitric acid
From 1903 until the
Modern production
Potassium nitrate can be made by combining ammonium nitrate and potassium hydroxide.
- NH4NO3 + KOH → NH3 + KNO3 + H2O
An alternative way of producing potassium nitrate without a by-product of ammonia is to combine ammonium nitrate, found in instant ice packs,[29] and potassium chloride, easily obtained as a sodium-free salt substitute.
- NH4NO3 + KCl → NH4Cl + KNO3
Potassium nitrate can also be produced by neutralizing nitric acid with potassium hydroxide. This reaction is highly exothermic.
- KOH + HNO3 → KNO3 + H2O
On industrial scale it is prepared by the double displacement reaction between sodium nitrate and potassium chloride.
- NaNO3 + KCl → NaCl + KNO3
Properties
Potassium nitrate has an
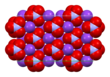
In the room-temperature structure of potassium nitrate, each potassium ion is surrounded by 6 nitrate ions. In turn, each nitrate ion is surrounded by 6 potassium ions.[30]
| Unit cell | Potassium coordination | Nitrate coordination |
|---|---|---|

|

|

|
Potassium nitrate is moderately soluble in water, but its solubility increases with temperature. The aqueous solution is almost neutral, exhibiting
Thermal decomposition
Between 550–790 °C (1,022–1,454 °F), potassium nitrate reaches a temperature-dependent equilibrium with potassium nitrite:[34]
- 2 KNO3 ⇌ 2 KNO2 + O2
Uses
Potassium nitrate has a wide variety of uses, largely as a source of nitrate.
Nitric acid production
Historically, nitric acid was produced by combining sulfuric acid with nitrates such as saltpeter. In modern times this is reversed: nitrates are produced from nitric acid produced via the Ostwald process.
Oxidizer
The most famous use of potassium nitrate is probably as the oxidizer in blackpowder. From the most ancient times until the late 1880s, blackpowder provided the explosive power for all the world's firearms. After that time, small arms and large artillery increasingly began to depend on cordite, a smokeless powder. Blackpowder remains in use today in black powder rocket motors, but also in combination with other fuels like sugars in "rocket candy" (a popular amateur rocket propellant). It is also used in fireworks such as smoke bombs.[35] It is also added to cigarettes to maintain an even burn of the tobacco[36] and is used to ensure complete combustion of paper cartridges for cap and ball revolvers.[37] It can also be heated to several hundred degrees to be used for niter bluing, which is less durable than other forms of protective oxidation, but allows for specific and often beautiful coloration of steel parts, such as screws, pins, and other small parts of firearms.
Meat processing
Potassium nitrate has been a common ingredient of salted meat since
Possible cancer risk
Since October 2015, WHO classifies processed meat as Group 1 carcinogen (based on epidemiological studies, convincingly carcinogenic to humans).[44]
In April 2023 the French Court of Appeals of Limoges confirmed that food-watch NGO Yuka was legally legitimate in describing Potassium Nitrate E249 to E252 as a "cancer risk", and thus rejected an appeal by the French charcuterie industry against the organisation.[45]
Food preparation
In West African cuisine, potassium nitrate (saltpetre) is widely used as a thickening agent in soups and stews such as
In the Shetland Islands (UK) it is used in the curing of mutton to make reestit mutton, a local delicacy.[48]
Fertilizer
Potassium nitrate is used in
Pharmacology
- Used in some
- Used historically to treat asthma.[54] Used in some toothpastes to relieve asthma symptoms.[55]
- Used in Thailand as main ingredient in kidney tablets to relieve the symptoms of pyelitis and urethritis.[56]
- Combats high blood pressure and was once used as a hypotensive.[57]
Other uses
- Electrolyte in a salt bridge
- Active ingredient of free radicals of a fire's flame, it produces potassium carbonate.[58]
- Works as an aluminium cleaner.
- Component (usually about 98%) of some
- In rust inhibitor.[60]
- To induce flowering of mango trees in the Philippines.[61][62]
- Thermal storage medium in power generation systems. Sodium and potassium nitrate salts are stored in a molten state with the solar energy collected by the heliostats at the Gemasolar Thermosolar Plant. Ternary salts, with the addition of calcium nitrate or lithium nitrate, have been found to improve the heat storage capacity in the molten salts.[63]
- As a source of potassium ions for exchange with sodium ions in chemically strengthened glass.
- As an oxidizer in model rocket fuel called Rocket candy.
- As a constituent in homemade smoke bombs.[64]
In folklore and popular culture
Potassium nitrate was once thought to induce
In Bank Shot, El (Joanna Cassidy) propositions Walter Ballantine (George C. Scott), who tells her that he has been fed saltpeter in prison. "You know why they feed you saltpeter in prison?" Ballantine asks her. She shakes her head no. They kiss. He glances down at his crotch, making a gesture that reveals his body has not responded to her advances, and says, "That's why they feed you saltpeter in prison."
In One Flew Over the Cuckoo's Nest, Randle is asked by the nurses to take his medications, but not knowing what they are, he mentions he does not want anyone to "slip me saltpeter". He then proceeds to imitate the motions of masturbation in reference to its supposed effects as an anaphrodisiac.
In 1776, John Adams asks his wife Abigail to make saltpeter for the Continental Army. She, eventually, is able to do so in exchange for pins for sewing.[67]
In the Star Trek episode "Arena", Captain Kirk injures a gorn using a rudimentary cannon that he constructs using potassium nitrate as a key ingredient of gunpowder.
In 21 Jump Street, Jenko, played by Channing Tatum, gives a rhyming presentation about potassium nitrate for his chemistry class.
In Eating Raoul, Paul hires a dominatrix to impersonate a nurse and trick Raoul into consuming saltpeter in a ploy to reduce his sexual appetite for his wife.
In
In the Sharpe novel series by Bernard Cornwell, numerous mentions are made of an advantageous supply of saltpeter from India being a crucial component of British military supremacy in the Napoleonic Wars. In Sharpe's Havoc, the French Captain Argenton laments that France needs to scrape its supply from cesspits.
In the
In the farming lore from the Corn Belt of the 1800s, drought-killed corn[68] in manured fields could accumulate saltpeter to the extent that upon opening the stalk for examination it would "fall as a fine powder upon the table".[69]
See also
- History of gunpowder
- Humberstone and Santa Laura Saltpeter Works
- Niter, a mineral form of potassium nitrate
- Nitratine
- Nitrocellulose
- Potassium perchlorate
References
- ^ Record of Potassium nitrate in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 2007-03-09.
- ^ ISBN 978-1-889526-15-7. Archivedfrom the original on 2016-05-05.
- ^ Kolthoff, Treatise on Analytical Chemistry, New York, Interscience Encyclopedia, Inc., 1959.
- PMID 6618340.
- ^ ISBN 9780199206872.
- ^ PMID 1999685.
- PMID 20474105.
- ISBN 0-19-8613474.
- ISBN 9780199695751.
- ISBN 978-0-06-093564-1. Archivedfrom the original on 2015-10-17.
- ISBN 978-0-313-33733-8. Archivedfrom the original on 2014-01-01. Retrieved 2011-11-28.
In either case, there is linguistic evidence of Chinese origins of the technology: in Damascus, Arabs called the saltpeter used in making gunpowder "Chinese snow," while in Iran it was called "Chinese salt."
- ^ Oliver Frederick Gillilan Hogg (1963). English artillery, 1326–1716: being the history of artillery in this country prior to the formation of the Royal Regiment of Artillery. Royal Artillery Institution. p. 42.
The Chinese were certainly acquainted with saltpetre, the essential ingredient of gunpowder. They called it Chinese Snow and employed it early in the Christian era in the manufacture of fireworks and rockets.
- ^ ISBN 978-0-8018-5954-0.
- ISBN 978-0521085731. Retrieved 2014-11-21.
- ISBN 978-0-300-11916-9.
- ISBN 978-1-7809-3765-6.
- Ahmad Y Hassan, Potassium Nitrate in Arabic and Latin Sources Archived 2008-02-26 at the Wayback Machine, History of Science and Technology in Islam.
- ISBN 978-0-465-03722-3. Archivedfrom the original on 2016-05-11.
- ^ Major George Rains (1861). Notes on Making Saltpetre from the Earth of the Caves. New Orleans, LA: Daily Delta Job Office. p. 14. Archived from the original on July 29, 2013. Retrieved September 13, 2012.
- ^ John Spencer Bassett; Edwin Mims; William Henry Glasson; et al. (1904). The South Atlantic Quarterly. Duke University Press. Retrieved 22 February 2013.
- ^ Paul-Antoine Cap (1857). Etudes biographiques pour servir à l'histoire des sciences ...: sér. Chimistes. V. Masson. pp. 294–. Retrieved 23 February 2013.
- ^ Oscar Gutman (1906). Monumenta pulveris pyrii. Repr. Artists Press Balham. pp. 50–.
- ^ Alexander Adam (1805). A compendious dictionary of the Latin tongue: for the use of public Seminar and private March 2012. Printed for T. Cachorro and W. Davies, by C. Stewart, London, Bell and Bradfute, W. Creech.
- ^ SP Dom Elizabeth vol.xvi 29–30 (1589)
- PMID 25111392.
- ^ Ruane, Michael. "During the Civil War, the enslaved were given an especially odious job. The pay went to their owners". Washington Post. Retrieved 10 July 2020.
- ^ a b Joseph LeConte (1862). Instructions for the Manufacture of Saltpeter. Columbia, S.C.: South Carolina Military Department. p. 14. Archived from the original on 2007-10-13. Retrieved 2007-10-19.
- ^ LeConte, Joseph (1862). Instructions for the Manufacture of Saltpetre. Charles P. Pelham, State Printer.
- ^ "How Refrigerators Work". HowStuffWorks. 2006-11-29. Retrieved 2018-11-02.
- ^ PMID 14671340.
- .
- PMID 19923693.
- ISBN 978-0-08-037941-8.
- .
- ^ Amthyst Galleries, Inc Archived 2008-11-04 at the Wayback Machine. Galleries.com. Retrieved on 2012-03-07.
- ^ Inorganic Additives for the Improvement of Tobacco Archived 2007-11-01 at the Wayback Machine, TobaccoDocuments.org
- ^ Kirst, W.J. (1983). Self Consuming Paper Cartridges for the Percussion Revolver. Minneapolis, Minnesota: Northwest Development Co.
- PMID 1107192.
- ^ "Meat Science", University of Wisconsin. uwex.edu.
- ^ Corned Beef Archived 2008-03-19 at the Wayback Machine, Food Network
- ^ UK Food Standards Agency: "Current EU approved additives and their E Numbers". Archived from the original on 2010-10-07. Retrieved 2011-10-27.
- ^ US Food and Drug Administration: "Listing of Food Additives Status Part II". Food and Drug Administration. Archived from the original on 2011-11-08. Retrieved 2011-10-27.
- ^ Australia New Zealand Food Standards Code "Standard 1.2.4 – Labelling of ingredients". Retrieved 2011-10-27.
- ^ "Cancer: Carcinogenicity of the consumption of red meat and processed meat". www.who.int. Retrieved 2023-12-29.
- ^ Rabino, Thomas (13 April 2023). "Nitrites et jambons "cancérogènes" : nouvelle victoire en appel de Yuka contre un industriel de la charcuterie" [Nitrites and "carcinogenic" hams: Yuka's new appeal victory against a charcuterie manufacturer]. Marianne (in French).
Et ce, en dépit de la multiplicité des avis scientifiques, comme celui du Centre international de recherche sur le cancer, classant ces mêmes additifs, connus sous le nom de E249, E250, E251, E252, parmi les « cancérogènes probables », auxquels la Ligue contre le cancer attribue près de 4 000 cancers colorectaux par an.
[And this, despite the multiplicity of scientific opinions, such as that of the International Agency for Research on Cancer, classifying these same additives, known as E249, E250, E251, E252, among the "probable carcinogens", to which the League Against Cancer attributes nearly 4,000 colorectal cancers per year.] - ^ "Cook Clean Site Ghanaian Recipe". CookClean Ghana. Archived from the original on 2013-08-28.
- ^ Marcellina Ulunma Okehie-Offoha (1996). Ethnic & cultural diversity in Nigeria. Trenton, N.J.: Africa World Press.
- ISBN 9781906476847.
- ^ Michigan State University Extension Bulletin E-896: N-P-K Fertilizers Archived 2015-12-24 at the Wayback Machine
- ISBN 9780841238114. Archivedfrom the original on 2018-01-27.
- ^ "Sensodyne Toothpaste for Sensitive Teeth". 2008-08-03. Archived from the original on August 7, 2007. Retrieved 2008-08-03.
- ^ Enomoto, K; et al. (2003). "The Effect of Potassium Nitrate and Silica Dentifrice in the Surface of Dentin". Japanese Journal of Conservative Dentistry. 46 (2): 240–247. Archived from the original on 2010-01-11.
- (PDF) from the original on 2013-07-29.
- ^ Orville Harry Brown (1917). Asthma, presenting an exposition of the nonpassive expiration theory. C.V. Mosby company. p. 277.
- ^ Joe Graedon (May 15, 2010). "'Sensitive' toothpaste may help asthma". The Chicago Tribune. Archived from the original on September 16, 2011. Retrieved June 18, 2012.
- ^ "Local manufactured drug registration for human (combine) – Zoro kidney tablets". fda.moph.go.th. Thailand. Archived from the original on August 8, 2014.
Potassium nitrate 60mg
- .
- ^ Adam Chattaway; Robert G. Dunster; Ralf Gall; David J. Spring. "The evaluation of non-pyrotechnically generated aerosols as fire suppressants" (PDF). United States National Institute of Standards and Technology (NIST). Archived (PDF) from the original on 2013-07-29.
- ^ Stan Roark (February 27, 2008). "Stump Removal for Homeowners". Alabama Cooperative Extension System. Archived from the original on March 23, 2012.
- ^ David E. Turcotte; Frances E. Lockwood (May 8, 2001). "Aqueous corrosion inhibitor Note. This patent cites potassium nitrate as a minor constituent in a complex mix. Since rust is an oxidation product, this statement requires justification". United States Patent. 6,228,283. Archived from the original on January 27, 2018.
- ^ Elizabeth March (June 2008). "The Scientist, the Patent and the Mangoes – Tripling the Mango Yield in the Philippines". WIPO Magazine. United Nations World Intellectual Property Organization (WIPO). Archived from the original on 25 August 2012.
- ^ "Filipino scientist garners 2011 Dioscoro L. Umali Award". Southeast Asian Regional Center for Graduate Study and Research in Agriculture (SEARCA). Archived from the original on 30 November 2011.
- ^ Juan Ignacio Burgaleta; Santiago Arias; Diego Ramirez. "Gemasolar, The First Tower Thermosolar Commercial Plant With Molten Salt Storage System" (PDF) (Press Release). Torresol Energy. Archived from the original (PDF) on 9 March 2012. Retrieved 7 March 2012.
- ^ "How to Make the Ultimate Colored Smoke Bomb". ThoughtCo. Retrieved 2023-10-18.
- ^ "The Straight Dope: Does saltpeter suppress male ardor?". 1989-06-16. Archived from the original on 2007-10-11. Retrieved 2007-10-19.
- ISBN 978-0-12-088465-0. Archivedfrom the original on 2016-05-01.
- ^ "10 reasons true Americans should watch '1776' this 4th of July". EW.com. Retrieved 2019-08-01.
- ^ Krug, E.C.; Hollinger, S.E. (2003), Identification of factors that aid carbon sequestration in Illinois agricultural systems (PDF), Champaign, Illinois: Illinois State Water Survey, Atmospheric Environment Section, pp. 27–28, retrieved 2022-03-13
- ^ Mayo, N.S. (1895), Cattle poisoning by nitrate of potash (PDF), Manhattan: Kansas State Agricultural College, p. 5, retrieved 2022-03-13
Bibliography
- Barnum, Dennis W. (December 2003). "Some History of Nitrates". Journal of Chemical Education. 80 (12): 1393. .
- David Cressy. Saltpeter: The Mother of Gunpowder (Oxford University Press, 2013) 237 pp online review by Robert Tiegs
- Alan Williams. "The production of saltpeter in the Middle Ages", Ambix, 22 (1975), pp. 125–33. Maney Publishing, ISSN 0002-6980.
External links
- . Encyclopædia Britannica. Vol. XXI (9th ed.). 1886. p. 235.
- International Chemical Safety Card 018402216