Bioinformatics


Bioinformatics (
Computational, statistical, and computer programming techniques have been used for computer simulation analyses of biological queries. They include reused specific analysis "pipelines", particularly in the field of genomics, such as by the identification of genes and single nucleotide polymorphisms (SNPs). These pipelines are used to better understand the genetic basis of disease, unique adaptations, desirable properties (esp. in agricultural species), or differences between populations. Bioinformatics also includes proteomics, which tries to understand the organizational principles within nucleic acid and protein sequences.[2]
Image and signal processing allow extraction of useful results from large amounts of raw data. In the field of genetics, it aids in sequencing and annotating genomes and their observed mutations. Bioinformatics includes text mining of biological literature and the development of biological and gene ontologies to organize and query biological data. It also plays a role in the analysis of gene and protein expression and regulation. Bioinformatics tools aid in comparing, analyzing and interpreting genetic and genomic data and more generally in the understanding of evolutionary aspects of molecular biology. At a more integrative level, it helps analyze and catalogue the biological pathways and networks that are an important part of systems biology. In structural biology, it aids in the simulation and modeling of DNA,[3] RNA,[3][4] proteins[5] as well as biomolecular interactions.[6][7][8][9]
History
The first definition of the term bioinformatics was coined by Paulien Hogeweg and Ben Hesper in 1970, to refer to the study of information processes in biotic systems.[10][11][12][13][14] This definition placed bioinformatics as a field parallel to biochemistry (the study of chemical processes in biological systems).[11]
Bioinformatics and computational biology involved the analysis of biological data, particularly DNA, RNA, and protein sequences. The field of bioinformatics experienced explosive growth starting in the mid-1990s, driven largely by the Human Genome Project and by rapid advances in DNA sequencing technology.
Analyzing biological data to produce meaningful information involves writing and running software programs that use
Sequences

There has been a tremendous advance in speed and cost reduction since the completion of the Human Genome Project, with some labs able to sequence over 100,000 billion bases each year, and a full genome can be sequenced for $1,000 or less.[15]
Computers became essential in molecular biology when
In the 1970s, new techniques for sequencing DNA were applied to bacteriophage MS2 and øX174, and the extended nucleotide sequences were then parsed with informational and statistical algorithms. These studies illustrated that well known features, such as the coding segments and the triplet code, are revealed in straightforward statistical analyses and were the proof of the concept that bioinformatics would be insightful.[22][23]

Goals
In order to study how normal cellular activities are altered in different disease states, raw biological data must be combined to form a comprehensive picture of these activities. Therefore[
Important sub-disciplines within bioinformatics and computational biology include:
- Development and implementation of computer programs to efficiently access, manage, and use various types of information.
- Development of new mathematical algorithms and statistical measures to assess relationships among members of large data sets. For example, there are methods to locate a gene within a sequence, to predict protein structure and/or function, and to cluster protein sequences into families of related sequences.
The primary goal of bioinformatics is to increase the understanding of biological processes. What sets it apart from other approaches is its focus on developing and applying computationally intensive techniques to achieve this goal. Examples include:
Bioinformatics entails the creation and advancement of databases, algorithms, computational and statistical techniques, and theory to solve formal and practical problems arising from the management and analysis of biological data.
Over the past few decades, rapid developments in genomic and other molecular research technologies and developments in
Common activities in bioinformatics include mapping and analyzing DNA and protein sequences, aligning DNA and protein sequences to compare them, and creating and viewing 3-D models of protein structures.
Sequence analysis
Since the bacteriophage
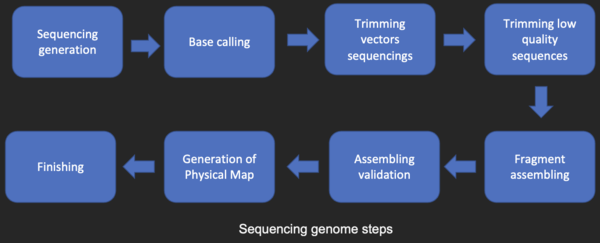
DNA sequencing
Before sequences can be analyzed, they are obtained from a data storage bank, such as GenBank. DNA sequencing is still a non-trivial problem as the raw data may be noisy or affected by weak signals. Algorithms have been developed for base calling for the various experimental approaches to DNA sequencing.
Sequence assembly
Most DNA sequencing techniques produce short fragments of sequence that need to be assembled to obtain complete gene or genome sequences. The
Genome annotation
In genomics, annotation refers to the process of marking the stop and start regions of genes and other biological features in a sequenced DNA sequence. Many genomes are too large to be annotated by hand. As the rate of sequencing exceeds the rate of genome annotation, genome annotation has become the new bottleneck in bioinformatics[when?].
Genome annotation can be classified into three levels: the nucleotide, protein, and process levels.
Gene finding is a chief aspect of nucleotide-level annotation. For complex genomes, a combination of ab initio gene prediction and sequence comparison with expressed sequence databases and other organisms can be successful. Nucleotide-level annotation also allows the integration of genome sequence with other genetic and physical maps of the genome.
The principal aim of protein-level annotation is to assign function to the protein products of the genome. Databases of protein sequences and functional domains and motifs are used for this type of annotation. About half of the predicted proteins in a new genome sequence tend to have no obvious function.
Understanding the function of genes and their products in the context of cellular and organismal physiology is the goal of process-level annotation. An obstacle of process-level annotation has been the inconsistency of terms used by different model systems. The Gene Ontology Consortium is helping to solve this problem.[28]
The first description of a comprehensive annotation system was published in 1995
Following the goals that the Human Genome Project left to achieve after its closure in 2003, the ENCODE project was developed by the National Human Genome Research Institute. This project is a collaborative data collection of the functional elements of the human genome that uses next-generation DNA-sequencing technologies and genomic tiling arrays, technologies able to automatically generate large amounts of data at a dramatically reduced per-base cost but with the same accuracy (base call error) and fidelity (assembly error).
Gene function prediction
While genome annotation is primarily based on sequence similarity (and thus homology), other properties of sequences can be used to predict the function of genes. In fact, most gene function prediction methods focus on protein sequences as they are more informative and more feature-rich. For instance, the distribution of hydrophobic amino acids predicts transmembrane segments in proteins. However, protein function prediction can also use external information such as gene (or protein) expression data, protein structure, or protein-protein interactions.[29]
Computational evolutionary biology
- trace the evolution of a large number of organisms by measuring changes in their DNA, rather than through physical taxonomy or physiological observations alone,
- compare entire genomes, which permits the study of more complex evolutionary events, such as gene duplication, horizontal gene transfer, and the prediction of factors important in bacterial speciation,
- build complex computational population genetics models to predict the outcome of the system over time[30]
- track and share information on an increasingly large number of species and organisms
Future work endeavours to reconstruct the now more complex
Comparative genomics
The core of comparative genome analysis is the establishment of the correspondence between
Many of these studies are based on the detection of sequence homology to assign sequences to protein families.[32]
Pan genomics
Pan genomics is a concept introduced in 2005 by Tettelin and Medini. Pan genome is the complete gene repertoire of a particular monophyletic taxonomic group. Although initially applied to closely related strains of a species, it can be applied to a larger context like genus, phylum, etc. It is divided in two parts: the Core genome, a set of genes common to all the genomes under study (often housekeeping genes vital for survival), and the Dispensable/Flexible genome: a set of genes not present in all but one or some genomes under study. A bioinformatics tool BPGA can be used to characterize the Pan Genome of bacterial species.[33]
Genetics of disease
As of 2013, the existence of efficient high-throughput next-generation sequencing technology allows for the identification of cause many different human disorders. Simple Mendelian inheritance has been observed for over 3,000 disorders that have been identified at the Online Mendelian Inheritance in Man database, but complex diseases are more difficult. Association studies have found many individual genetic regions that individually are weakly associated with complex diseases (such as infertility,[34] breast cancer[35] and Alzheimer's disease[36]), rather than a single cause.[37][38] There are currently many challenges to using genes for diagnosis and treatment, such as how we don't know which genes are important, or how stable the choices an algorithm provides. [39]
Genome-wide association studies have successfully identified thousands of common genetic variants for complex diseases and traits; however, these common variants only explain a small fraction of heritability.
Analysis of mutations in cancer
In
Two important principles can be used to identify cancer by mutations in the exome. First, cancer is a disease of accumulated somatic mutations in genes. Second, cancer contains driver mutations which need to be distinguished from passengers.[47]
Further improvements in bioinformatics could allow for classifying types of cancer by analysis of cancer driven mutations in the genome. Furthermore, tracking of patients while the disease progresses may be possible in the future with the sequence of cancer samples. Another type of data that requires novel informatics development is the analysis of lesions found to be recurrent among many tumors.[48]
Gene and protein expression
Analysis of gene expression
The

Analysis of protein expression
Protein microarrays and high throughput (HT) mass spectrometry (MS) can provide a snapshot of the proteins present in a biological sample. The former approach faces similar problems as with microarrays targeted at mRNA, the latter involves the problem of matching large amounts of mass data against predicted masses from protein sequence databases, and the complicated statistical analysis of samples when multiple incomplete peptides from each protein are detected. Cellular protein localization in a tissue context can be achieved through affinity proteomics displayed as spatial data based on immunohistochemistry and tissue microarrays.[50]
Analysis of regulation
Gene regulation is a complex process where a signal, such as an extracellular signal such as a hormone, eventually leads to an increase or decrease in the activity of one or more proteins. Bioinformatics techniques have been applied to explore various steps in this process.
For example, gene expression can be regulated by nearby elements in the genome. Promoter analysis involves the identification and study of sequence motifs in the DNA surrounding the protein-coding region of a gene. These motifs influence the extent to which that region is transcribed into mRNA. Enhancer elements far away from the promoter can also regulate gene expression, through three-dimensional looping interactions. These interactions can be determined by bioinformatic analysis of chromosome conformation capture experiments.
Expression data can be used to infer gene regulation: one might compare
Analysis of cellular organization
Several approaches have been developed to analyze the location of organelles, genes, proteins, and other components within cells. A
Microscopy and image analysis
Microscopic pictures allow for the location of organelles as well as molecules, which may be the source of abnormalities in diseases.
Protein localization
Finding the location of proteins allows us to predict what they do. This is called protein function prediction. For instance, if a protein is found in the nucleus it may be involved in gene regulation or splicing. By contrast, if a protein is found in mitochondria, it may be involved in respiration or other metabolic processes. There are well developed protein subcellular localization prediction resources available, including protein subcellular location databases, and prediction tools.[51][52]
Nuclear organization of chromatin
Data from high-throughput
Structural bioinformatics

Finding the structure of proteins is an important application of bioinformatics. The Critical Assessment of Protein Structure Prediction (CASP) is an open competition where worldwide research groups submit protein models for evaluating unknown protein models.[54][55]
Amino acid sequence
The linear
Homology
In the genomic branch of bioinformatics, homology is used to predict the function of a gene: if the sequence of gene A, whose function is known, is homologous to the sequence of gene B, whose function is unknown, one could infer that B may share A's function. In structural bioinformatics, homology is used to determine which parts of a protein are important in structure formation and interaction with other proteins. Homology modeling is used to predict the structure of an unknown protein from existing homologous proteins.
One example of this is hemoglobin in humans and the hemoglobin in legumes (leghemoglobin), which are distant relatives from the same protein superfamily. Both serve the same purpose of transporting oxygen in the organism. Although both of these proteins have completely different amino acid sequences, their protein structures are virtually identical, which reflects their near identical purposes and shared ancestor.[56]
Other techniques for predicting protein structure include protein threading and de novo (from scratch) physics-based modeling.
Another aspect of structural bioinformatics include the use of protein structures for
A 2021
Network and systems biology
Network analysis seeks to understand the relationships within biological networks such as metabolic or protein–protein interaction networks. Although biological networks can be constructed from a single type of molecule or entity (such as genes), network biology often attempts to integrate many different data types, such as proteins, small molecules, gene expression data, and others, which are all connected physically, functionally, or both.
Systems biology involves the use of computer simulations of cellular subsystems (such as the networks of metabolites and enzymes that comprise metabolism, signal transduction pathways and gene regulatory networks) to both analyze and visualize the complex connections of these cellular processes. Artificial life or virtual evolution attempts to understand evolutionary processes via the computer simulation of simple (artificial) life forms.
Molecular interaction networks

Tens of thousands of three-dimensional protein structures have been determined by
Other interactions encountered in the field include Protein–ligand (including drug) and protein–peptide. Molecular dynamic simulation of movement of atoms about rotatable bonds is the fundamental principle behind computational algorithms, termed docking algorithms, for studying molecular interactions.
Biodiversity informatics
Biodiversity informatics deals with the collection and analysis of
Others
Literature analysis
The enormous number of published literature makes it virtually impossible for individuals to read every paper, resulting in disjointed sub-fields of research. Literature analysis aims to employ computational and statistical linguistics to mine this growing library of text resources. For example:
- Abbreviation recognition – identify the long-form and abbreviation of biological terms
- Named-entity recognition – recognizing biological terms such as gene names
- Protein–protein interaction – identify which proteins interact with which proteins from text
The area of research draws from statistics and computational linguistics.
High-throughput image analysis
Computational technologies are used to automate the processing, quantification and analysis of large amounts of high-information-content
- high-throughput and high-fidelity quantification and sub-cellular localization (high-content screening, cytohistopathology, Bioimage informatics)
- morphometrics
- clinical image analysis and visualization
- determining the real-time air-flow patterns in breathing lungs of living animals
- quantifying occlusion size in real-time imagery from the development of and recovery during arterial injury
- making behavioral observations from extended video recordings of laboratory animals
- infrared measurements for metabolic activity determination
- inferring clone overlaps in DNA mapping, e.g. the Sulston score
High-throughput single cell data analysis
Computational techniques are used to analyse high-throughput, low-measurement single cell data, such as that obtained from flow cytometry. These methods typically involve finding populations of cells that are relevant to a particular disease state or experimental condition.
Ontologies and data integration
Biological ontologies are directed acyclic graphs of controlled vocabularies. They create categories for biological concepts and descriptions so they can be easily analyzed with computers. When categorised in this way, it is possible to gain added value from holistic and integrated analysis.[citation needed]
The
Databases
Databases are essential for bioinformatics research and applications. Databases exist for many different information types, including DNA and protein sequences, molecular structures, phenotypes and biodiversity. Databases can contain both empirical data (obtained directly from experiments) and predicted data (obtained from analysis of existing data). They may be specific to a particular organism, pathway or molecule of interest. Alternatively, they can incorporate data compiled from multiple other databases. Databases can have different formats, access mechanisms, and be public or private.
Some of the most commonly used databases are listed below:
- Used in biological sequence analysis: Genbank, UniProt
- Used in structure analysis: Protein Data Bank (PDB)
- Used in finding Protein Families and Motif Finding: InterPro, Pfam
- Used for Next Generation Sequencing: Sequence Read Archive
- Used in Network Analysis: Metabolic Pathway Databases (KEGG, BioCyc), Interaction Analysis Databases, Functional Networks
- Used in design of synthetic genetic circuits: GenoCAD[citation needed]
Software and tools
Software tools for bioinformatics include simple command-line tools, more complex graphical programs, and standalone web-services. They are made by bioinformatics companies or by public institutions.
Open-source bioinformatics software
Many
Open-source bioinformatics software includes Bioconductor, BioPerl, Biopython, BioJava, BioJS, BioRuby, Bioclipse, EMBOSS, .NET Bio, Orange with its bioinformatics add-on, Apache Taverna, UGENE and GenoCAD.
The non-profit Open Bioinformatics Foundation[60] and the annual Bioinformatics Open Source Conference promote open-source bioinformatics software.[61]
Web services in bioinformatics
SOAP- and REST-based interfaces have been developed to allow client computers to use algorithms, data and computing resources from servers in other parts of the world. The main advantage are that end users do not have to deal with software and database maintenance overheads.
Basic bioinformatics services are classified by the
Bioinformatics workflow management systems
A
- provide an easy-to-use environment for individual application scientists themselves to create their own workflows,
- provide interactive tools for the scientists enabling them to execute their workflows and view their results in real-time,
- simplify the process of sharing and reusing workflows between the scientists, and
- enable scientists to track the provenance of the workflow execution results and the workflow creation steps.
Some of the platforms giving this service: Galaxy, Kepler, Taverna, UGENE, Anduril, HIVE.
BioCompute and BioCompute Objects
In 2014, the US Food and Drug Administration sponsored a conference held at the National Institutes of Health Bethesda Campus to discuss reproducibility in bioinformatics.[63] Over the next three years, a consortium of stakeholders met regularly to discuss what would become BioCompute paradigm.[64] These stakeholders included representatives from government, industry, and academic entities. Session leaders represented numerous branches of the FDA and NIH Institutes and Centers, non-profit entities including the Human Variome Project and the European Federation for Medical Informatics, and research institutions including Stanford, the New York Genome Center, and the George Washington University.
It was decided that the BioCompute paradigm would be in the form of digital 'lab notebooks' which allow for the reproducibility, replication, review, and reuse, of bioinformatics protocols. This was proposed to enable greater continuity within a research group over the course of normal personnel flux while furthering the exchange of ideas between groups. The US FDA funded this work so that information on pipelines would be more transparent and accessible to their regulatory staff.[65]
In 2016, the group reconvened at the NIH in Bethesda and discussed the potential for a BioCompute Object, an instance of the BioCompute paradigm. This work was copied as both a "standard trial use" document and a preprint paper uploaded to bioRxiv. The BioCompute object allows for the JSON-ized record to be shared among employees, collaborators, and regulators.[66][67]
Education platforms
Bioinformatics is not only taught as in-person masters degree at many universities. The computational nature of bioinformatics lends it to computer-aided and online learning.[68][69] Software platforms designed to teach bioinformatics concepts and methods include Rosalind and online courses offered through the Swiss Institute of Bioinformatics Training Portal. The Canadian Bioinformatics Workshops provides videos and slides from training workshops on their website under a Creative Commons license. The 4273π project or 4273pi project[70] also offers open source educational materials for free. The course runs on low cost Raspberry Pi computers and has been used to teach adults and school pupils.[71][72] 4273 is actively developed by a consortium of academics and research staff who have run research level bioinformatics using Raspberry Pi computers and the 4273π operating system.[73][74]
MOOC platforms also provide online certifications in bioinformatics and related disciplines, including Coursera's Bioinformatics Specialization (UC San Diego) and Genomic Data Science Specialization (Johns Hopkins) as well as EdX's Data Analysis for Life Sciences XSeries (Harvard).
Conferences
There are several large conferences that are concerned with bioinformatics. Some of the most notable examples are Intelligent Systems for Molecular Biology (ISMB), European Conference on Computational Biology (ECCB), and Research in Computational Molecular Biology (RECOMB).
See also
- Biodiversity informatics
- Bioinformatics companies
- Computational biology
- Computational biomodeling
- Computational genomics
- Cyberbiosecurity
- Functional genomics
- Health informatics
- International Society for Computational Biology
- Jumping library
- List of bioinformatics institutions
- List of open-source bioinformatics software
- List of bioinformatics journals
- Metabolomics
- Nucleic acid sequence
- Phylogenetics
- Proteomics
- Gene Disease Database
References
- ISBN 978-1-119-69796-1.
- ^ Lesk AM (26 July 2013). "Bioinformatics". Encyclopaedia Britannica. Archived from the original on 14 April 2021. Retrieved 17 April 2017.
- ^ PMID 22538125.
- PMID 27125734.
- PMID 27333362.
- ISBN 978-1-4987-2497-5.
- PMID 25414269.
- PMID 25443955.
- PMID 29733895.
- PMID 14630646.
- ^ PMID 21483479.
- ^ Hesper B, Hogeweg P (1970). "BIO-INFORMATICA: een werkconcept" [BIO-INFORMATICS: a working concept]. Het Kameleon (in Dutch). 1 (6): 28–29.
- arXiv:2111.11832v1 [q-bio.OT].
- S2CID 61206099.
- ^ Colby B (2022). "Whole Genome Sequencing Cost". Sequencing.com. Archived from the original on 15 March 2022. Retrieved 8 April 2022.
- PMID 14886310.
- PMID 13032078.
- ISBN 978-0-471-32788-2.
- LCCN 65-29342.
- S2CID 23208558.
- PMID 10592229.
- S2CID 85199492.
- PMID 6456380.
- ISBN 978-0-511-16815-4– via Internet Archive.
- S2CID 4206886.
- PMID 18073190.
- ^ PMID 7542800.
- S2CID 12044602.
- PMID 21353529.
- PMID 20808525.
- ^ Brown TA (2002). "Mutation, Repair and Recombination". Genomes (2nd ed.). Manchester (UK): Oxford.
- PMID 12357458.
- PMID 27071527.
- S2CID 206007180.
- PMID 24521025.
- PMID 23954969.
- PMID 23824853.
- PMID 19474294.
- S2CID 21622726.
- PMID 19812666.
- PMID 35256806.
- PMID 33568819.
- PMID 32839606.
- S2CID 243873361.
- S2CID 253246835.
- S2CID 255084231.
- PMID 23300415.
- ISBN 978-0-12-396967-5.
- PMID 16845064.
- ^ "The Human Protein Atlas". www.proteinatlas.org. Archived from the original on 4 March 2020. Retrieved 2 October 2017.
- ^ "The human cell". www.proteinatlas.org. Archived from the original on 2 October 2017. Retrieved 2 October 2017.
- S2CID 10744558.
- PMID 26328929.
- PMID 31589781.
- ^ "Home - CASP14". predictioncenter.org. Archived from the original on 30 January 2023. Retrieved 12 June 2023.
- PMID 17560601.
- PMID 34265844.
- ^ "AlphaFold Protein Structure Database". alphafold.ebi.ac.uk. Archived from the original on 24 July 2021. Retrieved 10 October 2022.
- PMID 18509523.
- ^ a b "Open Bioinformatics Foundation: About us". Official website. Open Bioinformatics Foundation. Archived from the original on 12 May 2011. Retrieved 10 May 2011.
- ^ "Open Bioinformatics Foundation: BOSC". Official website. Open Bioinformatics Foundation. Archived from the original on 18 July 2011. Retrieved 10 May 2011.
- ISBN 978-0-08-091203-5.
- ^ Office of the Commissioner. "Advancing Regulatory Science – Sept. 24–25, 2014 Public Workshop: Next Generation Sequencing Standards". www.fda.gov. Archived from the original on 14 November 2017. Retrieved 30 November 2017.
- PMID 27974626.
- ^ Office of the Commissioner. "Advancing Regulatory Science – Community-based development of HTS standards for validating data and computation and encouraging interoperability". www.fda.gov. Archived from the original on 26 January 2018. Retrieved 30 November 2017.
- PMID 30596645.
- ^ BioCompute Object (BCO) project is a collaborative and community-driven framework to standardize HTS computational data. 1. BCO Specification Document: user manual for understanding and creating B., biocompute-objects, 3 September 2017, archived from the original on 27 June 2018, retrieved 30 November 2017
- PMID 12888845.
- PMID 34231941.
- PMID 23937194.
- S2CID 256396656.
- PMID 27006868.
- PMID 26462790.
- PMID 26467441.
Further reading
- Sehgal et al. : Structural, phylogenetic and docking studies of D-amino acid oxidase activator(DAOA ), a candidate schizophrenia gene. Theoretical Biology and Medical Modelling 2013 10 :3.
- Achuthsankar S Nair Computational Biology & Bioinformatics – A gentle Overview Archived 16 December 2008 at the Wayback Machine, Communications of Computer Society of India, January 2007
- ISBN 1-58488-406-1(Chapman & Hall/Crc Computer and Information Science Series)
- Baldi, P and Brunak, S, Bioinformatics: The Machine Learning Approach, 2nd edition. MIT Press, 2001. ISBN 0-262-02506-X
- Barnes, M.R. and Gray, I.C., eds., Bioinformatics for Geneticists, first edition. Wiley, 2003. ISBN 0-470-84394-2
- Baxevanis, A.D. and Ouellette, B.F.F., eds., Bioinformatics: A Practical Guide to the Analysis of Genes and Proteins, third edition. Wiley, 2005. ISBN 0-471-47878-4
- Baxevanis, A.D., Petsko, G.A., Stein, L.D., and Stormo, G.D., eds., ISBN 0-471-25093-7
- Cristianini, N. and Hahn, M. Introduction to Computational Genomics Archived 4 January 2009 at the ISBN 0-521-67191-4)
- Durbin, R., S. Eddy, A. Krogh and G. Mitchison, Biological sequence analysis. Cambridge University Press, 1998. ISBN 0-521-62971-3
- Gilbert D (September 2004). "Bioinformatics software resources". Briefings in Bioinformatics. 5 (3): 300–4. PMID 15383216.
- Keedwell, E., Intelligent Bioinformatics: The Application of Artificial Intelligence Techniques to Bioinformatics Problems. Wiley, 2005. ISBN 0-470-02175-6
- Kohane, et al. Microarrays for an Integrative Genomics. The MIT Press, 2002. ISBN 0-262-11271-X
- Lund, O. et al. Immunological Bioinformatics. The MIT Press, 2005. ISBN 0-262-12280-4
- ISBN 0-521-85700-7
- Pevzner, Pavel A. Computational Molecular Biology: An Algorithmic Approach The MIT Press, 2000. ISBN 0-262-16197-4
- Soinov, L. Bioinformatics and Pattern Recognition Come Together Archived 10 May 2013 at the Wayback Machine Journal of Pattern Recognition Research (JPRR Archived 8 September 2008 at the Wayback Machine), Vol 1 (1) 2006 p. 37–41
- Stevens, Hallam, Life Out of Sequence: A Data-Driven History of Bioinformatics, Chicago: The University of Chicago Press, 2013, ISBN 9780226080208
- Tisdall, James. "Beginning Perl for Bioinformatics" O'Reilly, 2001. ISBN 0-596-00080-4
- Catalyzing Inquiry at the Interface of Computing and Biology (2005) CSTB report Archived 28 January 2007 at the Wayback Machine
- Calculating the Secrets of Life: Contributions of the Mathematical Sciences and computing to Molecular Biology (1995) Archived 6 July 2008 at the Wayback Machine
- Foundations of Computational and Systems Biology MIT Course
- Computational Biology: Genomes, Networks, Evolution Free MIT Course Archived 8 April 2013 at the Wayback Machine
External links
 The dictionary definition of bioinformatics at Wiktionary
The dictionary definition of bioinformatics at Wiktionary Learning materials related to Bioinformatics at Wikiversity
Learning materials related to Bioinformatics at Wikiversity Media related to Bioinformatics at Wikimedia Commons
Media related to Bioinformatics at Wikimedia Commons- Bioinformatics Resource Portal (SIB)

