Source: Wikipedia, the free encyclopedia.
Histidine methyl ester

|
| Names
|
| Other names
Methyl L-histidinate, HME
|
| Identifiers
|
|
|
|
|
|
|
|
|
957974
|
| ChEBI
|
|
| ChEMBL
|
|
| ChemSpider
|
|
ECHA InfoCard
|
100.014.645 
|
| EC Number
|
|
|
|
|
| UNII
|
|
|
|
|
InChI=1S/C7H11N3O2/c1-12-7(11)6(8)2-5-3-9-4-10-5/h3-4,6H,2,8H2,1H3,(H,9,10)/t6-/m0/s1 Key: BXRMEWOQUXOLDH-LURJTMIESA-N
|
|
|
| Properties
|
|
|
C7H11N3O2
|
| Molar mass
|
169.184 g·mol−1
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
Chemical compound
Histidine methyl ester (HME) is an irreversible histidine decarboxylase inhibitor.[1][2][3] It is the methyl ester of histidine.
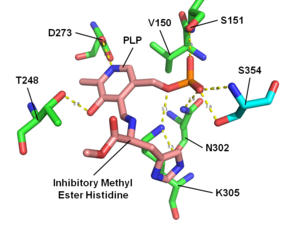 HME bound to histidine decarboxylase
HME bound to histidine decarboxylase
See also
References
|
|---|
| Non-specific | | AAADTooltip Aromatic L-amino acid decarboxylase | |
|---|
| MAOTooltip Monoamine oxidase | |
|---|
|
|---|
| | PAHTooltip Phenylalanine hydroxylase | |
|---|
| THTooltip Tyrosine hydroxylase | |
|---|
| Tooltip Dopamine beta-monooxygenase | |
|---|
| PNMTTooltip Phenylethanolamine N-methyltransferase |
- Substrates→Products:
Epinephrine (adrenaline)
|
|---|
| Tooltip Catechol-O-methyl transferase | |
|---|
|
|---|
Tryptamines
(serotonin, melatonin) | | TPHTooltip Tryptophan hydroxylase |
- Substrates→Products:
5-HTP
|
|---|
| Tooltip Serotonin N-acetyl transferase | |
|---|
| ASMTTooltip Acetylserotonin O-methyltransferase | |
|---|
|
|---|
| Histamine | | HDCTooltip Histidine decarboxylase | |
|---|
| HNMTTooltip Histamine N-methyltransferase | |
|---|
| DAOTooltip Diamine oxidase | |
|---|
|
|---|
|



