Bartoli indole synthesis
| Bartoli indole synthesis | |
|---|---|
| Named after | Giuseppe Bartoli[1] |
| Reaction type | Ring forming reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000494 |
The Bartoli indole synthesis (also called the Bartoli reaction) is the
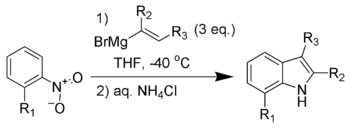
The reaction is often unsuccessful without substitution ortho to the nitro group, with bulkier ortho substituents usually resulting in higher yields for the reaction. The steric bulk of the ortho group assists in the [3,3]-sigmatropic rearrangement required for product formation. Three equivalents of the vinyl Grignard reagent are necessary for the reaction to achieve full conversion when performed on nitroarenes, and only two equivalents when performed on nitrosoarenes.
This method has become one of the shortest and most flexible routes to 7-substituted indoles.
Reaction mechanism
The reaction mechanism[8] of the Bartoli indole synthesis is illustrated below using o-nitrotoluene (1) and propenyl Grignard (2) to form 3,7-dimethylindole (13).

The mechanism begins by the addition of the Grignard reagent (2) onto the nitroarene (1) to form intermediate 3. Intermediate 3 spontaneously decomposes to form a
Therefore, three equivalents of the Grignard reagent are necessary, as one equivalent becomes carbonyl compound 6, one equivalent deprotonates 10 forming an alkene (11), and one equivalent gets incorporated into the indole ring.
The nitroso intermediate (4) has been isolated from the reaction. Additionally, reaction of the nitroso intermediate (4) with two equivalents of the Grignard reagent produces the expected indole.
The scope of the reaction includes substituted pyridines which can be used to make 4-azaindoles(left) and 6-azaindoles(right).[9]
Variations
Dobbs modification
Adrian Dobbs greatly enhanced the scope of the Bartoli indole synthesis by using an ortho-
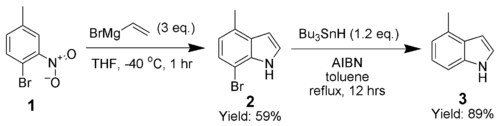
The synthesis of 4-methylindole (3) highlights the ability of this technique to produce highly substituted indoles.
See also
References
- PMID 32167641.
- .
- .
- S2CID 196737477.
- doi:10.2174/1385272053369204. Archived from the original(PDF) on 2007-09-28.
- .
- S2CID 196759261.
- .
- PMID 11925251.
- PMID 11429846.

