User:Abhinav2009/phosphofructokinase 1
copying within Wikipedia " guideline for an overview of the issues involved. |
| 6-phosphofructokinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Phosphofructokinase | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||||
| Symbol | PFK | ||||||||||
SCOP2 | 5pfk / SCOPe / SUPFAM | ||||||||||
| |||||||||||
Phosphofructokinase-1 (PFK-1) is one of the most important regulatory
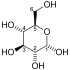
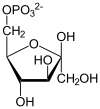
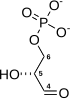
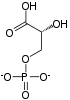
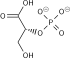
| β-D-fructose 6-phosphate | {{{forward_enzyme}}} | β-D-fructose 1,6-bisphosphate | |

|

| ||
| {{{minor_forward_substrate(s)}}} | {{{minor_forward_product(s)}}} | ||
| [[image:Biochem_reaction_arrow_{{{reaction_direction_(forward/reversible/reverse)}}}_NNYY_horiz_med.svg|75px]] | |||
| Pi | H2O | ||
| Fructose bisphosphatase | |||
Structure
Mammalian PFK1 is a 340kd
PFK1 is an allosteric enzyme and has a structure similar to that of hemoglobin insofar as it is a dimer of a dimer.[3] One half of each dimer contains the ATP binding site whereas the other half the substrate (fructose-6-phosphate or (F6P)) binding site as well as a separate allosteric binding site.[4]
Mechanism
PFK1 is an allosteric enzyme whose activity can be described using the
PFK1 belongs to the family of
Allosteric activators such as AMP and ADP bind to the allosteric site as to facilitate the formation of the R state by inducing structural changes in the enzyme. Similarly, inhibitors such as ATP and PEP bind to the same allosteric site and facilitate the formation of the T state, thereby inhibiting enzyme activity.
Regulation
PFK1 is the most important control site in the mammalian glycolytic pathway. This step is subject to extensive regulation since it is not only highly
PFK1 is
PFK1 is also inhibited by low pH levels which augment the inhibitory effect of ATP. The pH falls when muscle is functioning anaerobically and producing excessive quantities of lactic acid. This inhibitory effect serves to protect the muscle from damage that would result from the accumulation of too much acid.[1]
Finally, PFK1 is allosterically inhibited by both PEP and citrate. Phosphoenolpyruvic acid is a product further downstream the glycolytic pathway and citrate is an early intermediate in the citric acid cycle. Citrate buildup is a sign of the citric acid cycle reaching saturation and thus glycolysis slows down as there is no longer any need to commit more glucose to degradation.
PFK1 is
PFK is inhibited by
The precise regulation of PFK1 prevents
Genes
There are three phosphofructokinase genes in humans:
Disease
A genetic mutation in the
See also
- PFK2(converts fructose 6-phosphate to fructose 2,6-bisphosphate through on site, or the opposite, on another site)
- PFP (reversibly interconverts fructose 6-phosphate and fructose 1,6-bisphosphate using inorganic pyrophosphate rather than ATP)
- fructose bisphosphatase (hydrolyses fructose 1,6-bisphosphate to fructose 6-phosphate)
References
- ^ ISBN 978-0-7167-8724-2.)
{{cite book}}: CS1 maint: multiple names: authors list (link - PMID 2970843.)
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link - )
- )
- PMID 18763746.)
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link - PMID 2953977.
- PMID 11949936.)
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link
External links
- Phosphofructokinase-1 at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Proteopedia.org Phosphofructokinase