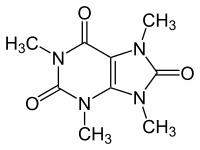
Theacrine
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
1,3,7,9-Tetramethyl-7,9-dihydro-1H-purine-2,6,8(3H)-trione | |
| Other names
1,3,7,9-Tetramethyluric acid; Temurin; Temorine; Tetramethyluric acid; Tetramethyl uric acid; TeaCrine (trade name)
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.017.268 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C9H12N4O3 | |
| Molar mass | 224.220 g·mol−1 |
| Melting point | 226 °C (439 °F; 499 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Theacrine, also known as 1,3,7,9-tetramethyluric acid, is a
Pharmacology
Pharmacodynamics
The exact mechanism of action of theacrine is uncertain, as no
Administration of selective dopamine D1 and D2 antagonists demonstrate that, similarly to caffeine,[4] theacrine's actions are in part mediated by dopamine receptors.[2] This should not be taken as evidence that theacrine directly interacts with and/or augments dopamine receptors distinct from caffeine, as some marketers have misleadingly claimed.
Pharmacokinetics
Theacrine has half-life of 30 to 33 hours.[5]
Safety
Theacrine has demonstrated clinical safety and non-habituating effects in healthy humans over eight weeks of daily use at up to 300 mg/day.[6] Moreover, there was no evidence of the tachyphylaxis typical of neuroactive agents like caffeine and other stimulants.[6]
In animal studies, theacrine has an LD50 of 810 mg/kg,[3][6] compared to 265 mg/kg for caffeine.[7]

