Pemetrexed
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Alimta, Pemfexy, Ciambra, others |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | NA |
| Protein binding | 81% |
| Metabolism | Negligible |
| Elimination half-life | 3.5 hours |
| Excretion | Kidney |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Pemetrexed, sold under the brand name Alimta among others, is a
It is available as a
Medical use
In February 2004, the U.S.
Carboplatin
Pemetrexed is also recommended in combination with carboplatin and pembrolizumab for the first-line treatment of advanced non-small cell lung cancer.[12][13] However, the relative efficacy or toxicity of pemetrexed-cisplatin versus pemetrexed-carboplatin has not been established beyond what is generally thought about cisplatin or carboplatin doublet drug therapy.[14]
Supplementation
Patients are recommended to take
Side effects
Pemetrexed, whether used alone or in combination with cisplatin, has these side effects:[2]
- Low blood cell counts, as measured by a complete blood count. This is a dose-limiting toxicity.
- Nausea and vomiting. Pemetrexed's emetogenic effects are managed with prophylactic antiemetics.
- Diarrhea
- Oral Oral ulcers can be mitigated by proper oral hygiene, including rinsing of the mouth with salt water following consumption of food or drink.[17]
- Loss of appetite
- Skin rash. Physician-prescribed glucocorticoids administered on the day prior, day of, and day after infusion typically avoid skin rashes.
- Constipation
Mechanism of action
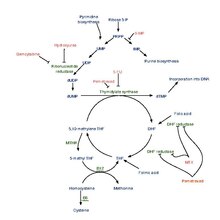
Pemetrexed is chemically similar to
Society and culture
Economics
In the United States, as of 2015[update], each vial of the medication costs between US$2,623 and US$3,100.[20]
Brand names
In February 2020, Pemfexy was approved for use in the United States.[21]
Research
A Phase III study showed benefits of maintenance use of pemetrexed for non-squamous NSCLC.[22] Activity has been shown in malignant peritoneal mesothelioma.[23]
References
- ^ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 30 March 2024.
- ^ a b c d e f "Alimta- pemetrexed disodium injection, powder, lyophilized, for solution". DailyMed. 25 March 2020. Archived from the original on 20 October 2020. Retrieved 20 October 2020.
- ^ "Pemfexy- pemetrexed injection". DailyMed. 20 January 2022. Archived from the original on 21 May 2022. Retrieved 20 May 2022.
- ^ "Alimta EPAR". European Medicines Agency. 17 September 2018. Archived from the original on 18 January 2021. Retrieved 14 October 2022.
- ^ "Pemetrexed Baxter: Pending EC decision". European Medicines Agency. 14 October 2022. Archived from the original on 14 October 2022. Retrieved 14 October 2022.
- ^ "2022 First Generic Drug Approvals". U.S. Food and Drug Administration (FDA). 3 March 2023. Archived from the original on 30 June 2023. Retrieved 30 June 2023.
- ^ "Competitive Generic Therapy Approvals". U.S. Food and Drug Administration (FDA). 29 June 2023. Archived from the original on 29 June 2023. Retrieved 29 June 2023.
- PMID 12917819.
- ^ National Cancer Institute: FDA Approval for Pemetrexed Disodium Archived 6 April 2015 at the Wayback Machine
- S2CID 28209589.
- PMID 19577816.
- ^ Ettinger DS, et al. "Non-small Cell Lung Cancer V.1.2007" (PDF). NCCN Clinical Practice Guidelines in Oncology. National Comprehensive Cancer Network (NCCN). Archived from the original (PDF) on 23 March 2007.
- PMID 29658856.
- PMID 17551137.
- S2CID 11444611.
- ^ "Scientists Help Breast Cancer Survivor Lift the Fog of "Chemo Brain"" (PDF). Dialogue. James P Wilmot Cancer Center, University of Rochester Medical Center: 2–3. Fall 2007. Retrieved 1 December 2008.[permanent dead link]
- ^ "Oral Complications of Chemotherapy and Head/Neck Radiation". Cancer Topics – Coping with Cancer. National Cancer Institute. Archived from the original on 6 December 2008. Retrieved 1 December 2008.
- from the original on 28 July 2009. Retrieved 2 December 2008.
- ISBN 978-0-444-52824-7.
- ^ Langreth R (29 June 2016). "Decoding Big Pharma's Secret Drug Pricing Practices". Bloomberg. Archived from the original on 13 July 2016. Retrieved 15 July 2016.
- ^ "Pemfexy: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 19 October 2020. Retrieved 13 February 2020.
- doi:10.1200/jco.2009.27.18_suppl.cra8000. Archived from the originalon 16 June 2009. Retrieved 22 July 2009.
- PMID 19042053.
External links
- "Pemetrexed disodium". NCI Drug Dictionary. National Cancer Institute.
- "Pemetrexed disodium". National Cancer Institute. 5 October 2006.
