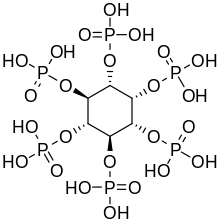
Phytic acid
| |
 | |

| |
| Names | |
|---|---|
| IUPAC name
(1R,2S,3r,4R,5S,6s)-cyclohexane-1,2,3,4,5,6-hexayl hexakis[dihydrogen (phosphate)]
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.001.369 |
| E number | E391 (antioxidants, ...) |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H18O24P6 | |
| Molar mass | 660.029 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phytic acid is a six-fold
The (myo) phytate anion is a colorless species that has significant nutritional role as the principal storage form of
The lower inositol polyphosphates are inositol esters with less than six phosphates, such as inositol penta- (IP5), tetra- (IP4), and triphosphate (IP3). These occur in nature as catabolites of phytic acid.
Significance in agriculture
Phytic acid was discovered in 1903.[2]
Generally, phosphorus and inositol in phytate form are not bioavailable to non-ruminant animals because these animals lack the enzyme phytase required to hydrolyze the inositol-phosphate linkages. Ruminants are able to digest phytate because of the phytase produced by rumen microorganisms.[3]
In most commercial
Also, viable low-phytic acid mutant lines have been developed in several crop species in which the seeds have drastically reduced levels of phytic acid and concomitant increases in inorganic phosphorus.[8] However, germination problems have reportedly hindered the use of these cultivars thus far. This may be due to phytic acid's critical role in both phosphorus and metal ion storage.[9] Phytate variants also have the potential to be used in soil remediation, to immobilize uranium, nickel, and other inorganic contaminants.[10]
Biological effects
Plants
Although indigestible for many animals as they occur in seeds and grains, phytic acid and its metabolites have several important roles for the seedling plant.
Most notably, phytic acid functions as a phosphorus store, as an energy store, as a source of cations and as a source of myo-inositol (a cell wall precursor). Phytic acid is the principal storage form of phosphorus in plant seeds.[11]
In vitro
In animal cells, myo-inositol polyphosphates are ubiquitous, and phytic acid (myo-inositol hexakisphosphate) is the most abundant, with its concentration ranging from 10 to 100 μM in mammalian cells, depending on cell type and developmental stage.[12][13]
Phytic acid is not obtained from the animal diet, but must be synthesized inside the cell from phosphate and inositol (which in turn is produced from glucose, usually in the kidneys). The interaction of intracellular phytic acid with specific intracellular proteins has been investigated in vitro, and these interactions have been found to result in the inhibition or potentiation of the activities of those proteins.[14][15]
Inositol hexaphosphate facilitates the formation of the six-helix bundle and assembly of the immature HIV-1 Gag lattice. IP6 makes ionic contacts with two rings of lysine residues at the centre of the Gag hexamer. Proteolytic cleavage then unmasks an alternative binding site, where IP6 interaction promotes the assembly of the mature capsid lattice. These studies identify IP6 as a naturally occurring small molecule that promotes both assembly and maturation of HIV-1.[16]
Dentistry
IP6 has potential use in endodontics, adhesive, preventive, and regenerative dentistry, and in improving the characteristics and performance of dental materials.[17][18][19]
Food science
Phytic acid, mostly as phytate in the form of phytin, is found within the hulls and kernels of seeds,[20] including nuts, grains, and pulses.[1]
In-home food preparation techniques may break down the phytic acid in all of these foods. Simply cooking the food will reduce the phytic acid to some degree. More effective methods are soaking in an acid medium, sprouting, and lactic acid fermentation such as in sourdough and pickling.[21]
No detectable phytate (less than 0.02% of wet weight) was observed in vegetables such as scallion and cabbage leaves or in fruits such as apples, oranges, bananas, or pears.[22]
As a food additive, phytic acid is used as the preservative E391.[23][24]
Dry food sources of phytic acid[25][22][26][27][28][29][30][31] Food Proportion by weight (g/100 g) Min. Max. Hulled Hemp Seed[20] 4.5 4.5 Pumpkin seed 4.3 4.3 Linseed2.15 2.78 Sesame seedsflour5.36 5.36 Chia seeds0.96 1.16 Almonds1.35 3.22 Brazil nuts 1.97 6.34 Coconut 0.36 0.36 Hazelnut 0.65 0.65 Peanut 0.95 1.76 Walnut 0.98 0.98 Maize (corn) 0.75 2.22 Oat 0.42 1.16 Oat meal 0.89 2.40 Brown rice 0.84 0.99 Polished rice 0.14 0.60 Wheat 0.39 1.35 Wheat flour 0.25 1.37 Wheat germ0.08 1.14 Whole wheat bread0.43 1.05 Beans, pinto 2.38 2.38 Buckwheat 1.00 1.00 Chickpeas0.56 0.56 Lentils0.44 0.50 Soybeans1.00 2.22 Tofu 1.46 2.90 Soybeverage1.24 1.24 Soy protein concentrate 1.24 2.17 New potato 0.18 0.34 Spinach 0.22 NR Avocado fruit 0.51 0.51 Chestnuts[32] 0.47 Sunflower seeds1.60
Fresh food sources of phytic acid[27] Food Proportion by weight (%) Min. Max. Taro 0.143 0.195 Cassava 0.114 0.152
Dietary mineral absorption
Phytic acid has a strong affinity to the dietary
Because phytic acid also can affect the absorption of
See also
- Antinutrient
- Essential nutrient
- Oxalic acid
References
- ^ PMID 19774556.
- ^ Mullaney EJ, Ullah, Abul H.J. "Phytases: attributes, catalytic mechanisms, and applications" (PDF). United States Department of Agriculture–Agricultural Research Service. Archived from the original (PDF) on 2012-11-07. Retrieved May 18, 2012.
- ^ a b Klopfenstein TJ, Angel R, Cromwell G, Erickson GE, Fox DG, Parsons C, Satter LD, Sutton AL, Baker DH (July 2002). "Animal Diet Modification to Decrease the Potential for Nitrogen and Phosphorus Pollution". Council for Agricultural Science and Technology. 21.
- .
- .
- S2CID 154321894.
- .
- S2CID 33700393.
- PMID 23890387, retrieved 2020-04-24
- PMID 12549554.
- PMID 6299067.
- PMID 3426614.
- PMID 7543266.
- S2CID 112839.
- PMID 7814377.
- PMID 30069050.
- .
- .
- PMID 34765567.
- ^ S2CID 233822572.
- ^ "Phytates in cereals and legumes". fao.org.
- ^ .
- ^ Functional Food - Improve Health through Adequate Food edited by María Chávarri Hueda, pg. 86
- ^ "Wise Eating, Made Easy".
- ^ Dephytinisation with Intrinsic Wheat Phytase and Iron Fortification Significantly Increase Iron Absorption from Fonio (Digitaria exilis) Meals in West African Women (2013)
- ISBN 978-1-56676-867-2.[page needed]
- ^ PMID 12517094.
- PMID 3341259.
- PMID 6321704.
- ISBN 978-0-85709-892-4.
- ^ Pereira Da Silva B. Concentration of nutrients and bioactive compounds in chia (Salvia Hispanica L.), protein quality and iron bioavailability in wistar rats (Ph.D. thesis). Federal University of Viçosa.
- ^ Scuhlz M. "Paleo Diet Guide: With Recipes in 30 Minutes or Less: Diabetes Heart Disease: Paleo Diet Friendly: Dairy Gluten Nut Soy Free Cookbook". PWPH Publications – via Google Books.
- PMID 25694676.
- doi:10.1002/jsfa.2471. Archived from the originalon 2020-02-23. Retrieved 2018-12-29.
- PMID 12949395.
- ISBN 978-0-309-02117-3.
- PMID 12716674.
- PMID 31979164.
