Envelope glycoprotein GP120
| Envelope glycoprotein gp120 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | GP120 | ||||||||
SCOP2 | 1gc1 / SCOPe / SUPFAM | ||||||||
| |||||||||
Envelope glycoprotein GP120 (or gp120) is a
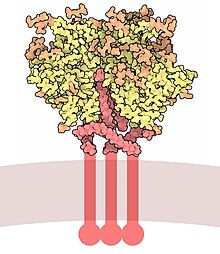
Gp120 is coded by the HIV
Variability
Since gp120 plays a vital role in the ability of HIV-1 to enter CD4+ cells, its evolution is of particular interest. Many neutralizing antibodies bind to sites located in variable regions of gp120, so mutations in these regions will be selected for strongly.[9] The diversity of env has been shown to increase by 1-2% per year in HIV-1 group M and the variable units are notable for rapid changes in amino acid sequence length. Increases in gp120 variability result in significantly elevated levels of viral replication, indicating an increase in viral fitness in individuals infected by diverse HIV-1 variants.[10] Further studies have shown that variability in potential N-linked glycosylation sites (PNGSs) also result in increased viral fitness. PNGSs allow for the binding of long-chain carbohydrates to the high variability regions of gp120, so the authors hypothesize that the number of PNGSs in env might affect the fitness of the virus by providing more or less sensitivity to neutralizing antibodies. The presence of large carbohydrate chains extending from gp120 might obscure possible antibody binding sites.[11]
The boundaries of the potential to add and eliminate PNGSs are naively explored by growing viral populations following each new infection.[12] While the transmitting host has developed a neutralizing antibody response to gp120, the newly infected host lacks immune recognition of the virus. Sequence data shows that initial viral variants in an immunologically naïve host have few glycosylation sites and shorter exposed variable loops. This may facilitate viral ability to bind host cell receptors.[13] As the host immune system develops antibodies against gp120, immune pressures seem to select for increased glycosylation, particularly on the exposed variable loops of gp120.[14] Consequently, insertions in env, which confer more PNGSs on gp120 may be more tolerated by the virus as higher glycan density promotes the viral ability to evade antibodies and thus promotes higher viral fitness.[15] In considering how much PNGS density could theoretically change, there may be an upper bound to PNGS number due to its inhibition of gp120 folding, but if the PNGS number decreases substantially, then the virus is too easily detected by neutralizing antibodies.[12] Therefore, a stabilizing selection balance between low and high glycan densities is likely established. A lower number of bulky glycans improves viral replication efficiency and higher number on the exposed loops aids host immune evasion via disguise.[citation needed]
The relationship between gp120 and neutralizing antibodies is an example of
Vaccine target
Since CD4 receptor binding is the most obvious step in HIV infection, gp120 was among the first targets of
Competition
The protein gp120 is necessary during the initial binding of HIV to its target cell. Consequently, anything which binds to gp120 or its targets can physically block gp120 from binding to a cell. Only one such agent,
HIV dementia
The HIV viral protein gp120 induces apoptosis of neuronal cells by inhibiting levels of furin and tissue plasminogen activator, enzymes responsible for converting pBDNF to mBDNF.[23] gp120 induces mitochondrial-death proteins like caspases which may influence the upregulation of the death receptor Fas leading to apoptosis of neuronal cells,[24] gp120 induces oxidative stress in the neuronal cells,[25] and it is also known to activate STAT1 and induce interleukins IL-6 and IL-8 secretion in neuronal cells.[26]
See also
References
- PMID 6204380.
- PMID 1518869.
- PMID 18040049.
- S2CID 4349809.
- PMID 23300605.
- ^ Kuiken, C., Leitner, T., Foley, B., et al. (2008). "HIV Sequence Compendium", Los Alamos National Laboratory.
- S2CID 4306605.
- PMID 19008954.
- S2CID 4384110.
- PMID 18973914.
- PMID 19424423.
- ^ PMID 15175256.
- PMID 18314154.
- PMID 16551265.
- ^ PMID 16339909.
- PMID 1384050.
- PMID 17301785.
- PMID 12477867.
- PMID 19995532.
- PMID 25331945.
- PMID 12134256.
- ^ aidsinfo.nih.gov/drugs/508/bms-663068/0/professional
- PMID 22787033.
- S2CID 25456692.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - S2CID 7362454.
- PMID 19103208.
Further reading
- Human Immunodeficiency Virus Glycoprotein 120
- Vashistha H, Husain M, Kumar D, Singhal PC (2009). "Tubular cell HIV-1 gp120 expression induces caspase 8 activation and apoptosis". Ren Fail. 31 (4): 303–12. S2CID 205593494.
External links
- https://web.archive.org/web/20060219135317/http://www.aidsmap.com/en/docs/4406022B-85D7-4A9B-B700-91336CBB6B18.asp
- http://www.mcld.co.uk/hiv/?q=gp120 Archived 2008-06-24 at the Wayback Machine
- http://www.ebi.ac.uk/interpro/IEntry?ac=IPR000777
- Vashistha, H.; Husain, M.; Kumar, D.; Singhal, P. C. (2009). "Tubular Cell HIV-1 gp120 Expression Induces Caspase 8 Activation and Apoptosis". Renal Failure. 31 (4): 303–312. S2CID 205593494.
