Implantation (embryology)
| Implantation | |
|---|---|
Gastrula | |
| Identifiers | |
| MeSH | D010064 |
| Anatomical terminology] | |
Implantation, also known as nidation,
For implantation to take place the uterus must become receptive. Uterine receptivity involves much cross-talk between the embryo and the uterus, initiating changes to the endometrium. This stage gives a synchrony that opens a window of implantation that enables successful implantation of a viable embryo.[4] The endocannabinoid system plays a vital role in this synchrony in the uterus, influencing uterine receptivity, and embryo implantation.[5] The embryo expresses cannabinoid receptors early in its development that are responsive to anandamide (AEA) secreted in the uterus. AEA is produced at higher levels before implantation and is then down-regulated at the time of implantation. This signaling is of importance in the embryo-uterus crosstalk in regulating the timing of embryonic implantation and uterine receptivity. Adequate concentrations of AEA that are neither too high or too low, are needed for successful implantation.[5][6][7]
There is an extensive variation in the type of trophoblast cells, and structures of the placenta across the different species of mammals.[8] Of the five recognised stages of implantation including two pre-implantation stages that precede placentation, the first four are similar across the species. The five stages are migration and hatching, pre-contact, attachment, adhesion, and invasion.[8] The two pre-implantation stages are associated with the pre-implantation embryo.[9][10]
In humans, following the stage of hatching that takes place around four to five days after fertilization, the process of implantation begins. By the end of the first week, the blastocyst is superficially attached to the uterine endometrium. By the end of the second week, implantation has completed.[11]
Implantation stages
There are five recognized stages of implantation in mammals, including two pre-implantation stages that precede the formation of the placenta. They are: migration and hatching, pre-contact, attachment, adhesion, and invasion. The first four stages are similar across the species with the process of invasion being variable.[8][12] These three stages of apposition, attachment, and invasion are also alternatively termed contact (apposition), adhesion (attachment), and penetration (invasion),[10][9] and can only take place during a limited timeframe known as the window of implantation when the uterus is at its most receptive.
Migration and hatching
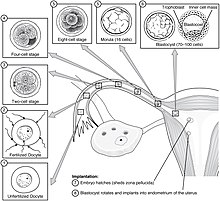
There are two stages of migration involved in implantation, the first is the migration of the zygote, and the second is the migration of the trophoblast.[13] Fertilization of the oocyte, takes place in the ampulla of the fallopian tube. Cilia on the lining of the tube move the zygote in its migration towards the uterus.
During this migration the zygote undergoes
The blastocyst is still enclosed in the egg-coat known as the zona pellucida, and for it to be able to implant into the uterine wall it must rid itself of this covering. This stage is known as zona hatching, and when there is sufficient dissolution the blastocyst is able to initiate the apposition stage of implantation. Lytic factors in the uterine cavity, as well as factors from the blastocyst itself are essential for the breakdown of the egg-coat. Mechanisms in the latter are indicated by the fact that the zona pellucida remains intact if an unfertilized egg is placed in the uterus under the same conditions.[16]
Among the known molecular regulators that promote hatching are predominantly
Assisted zona hatching may take place in assisted reproduction, where the zona pellucida may be artificially pierced to facilitate hatching.[18]
Apposition
Following zona hatching, the very first loose connection or contact between the blastocyst and the endometrium is called apposition. Apposition is usually made where there is a small crypt in the endometrium, and also where there has been enough breakdown of the zona pellucida to allow the blastocyst trophoblast to directly contact the underlying endometrium. Ultimately, the inner cell mass (also embryoblast), inside the trophoblast layer, is aligned closest to the decidua. If the inner cell mass is not aligned with the decidua at apposition, it has the ability to freely rotate within the trophoblast and achieve this alignment. Apposition is only a weak interaction of the trophectoderm with the uterine epithelium that is unstable to shear stress. Apposition is also reversible allowing repositioning of the blastocyst in the uterus.[14]
Adhesion
Adhesion is a much stronger attachment to the endometrium than the loose apposition.
The trophoblasts adhere by penetrating the endometrium, with protrusions of trophoblast cells.
This adhering activity is by microvilli that are on the trophoblast. The trophoblast have binding fiber connections, laminin, collagen type IV, and integrins that assist in this adhesion process.[19]
Mucin-16 is a transmembrane mucin expressed at the apical surface of uterine epithelia. This mucin prevents the blastocyst from implanting in an undesired located on the epithelium. Thus, MUC-16 inhibits cell-cell adhesion. Its removal during pinopode formation has been shown to facilitate trophoblast invasion in vitro.[20]
The identity of the molecules on the trophoblast and the endometrial epithelia that mediate the initial interaction between the two remain unidentified. However, a number of research groups have proposed that
Invasion
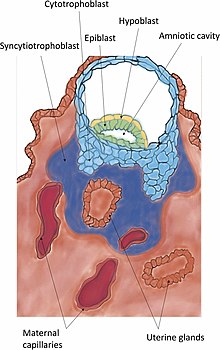
Invasion is the further establishment of the blastocyst into the endometrium. The protrusions of trophoblast cells that adhere into the endometrium continue to proliferate and penetrate into the endometrium using
When the syncytiotrophoblast reaches the basal membrane beneath the
Extravillous trophoblasts
Secretions
Pre-implantation blastocysts have been shown to be capable of secreting growth factors, hormones and trypsin-like proteases to participate in the hatching process.[33]
During invasion the blastocyst secretes factors for a multitude of purposes.
Immunosuppressive
The embryo differs from the cells of the mother, and would be rejected as a parasite by the
Other factors
Other factors secreted by the blastocyst are;[citation needed]
- Embryo-derived histamine-releasing factor
- Tissue plasminogen activatoras well as its inhibitors
- Estradiol
- β1-integrins
- Fibroblast growth factor
- Transforming growth factor alpha
- Inhibin
Uterus receptivity
To enable implantation, the uterus goes through changes in order to be able to receive the conceptus. Receptivity includes changes to endometrial cells in the formation of pinopodes that help to absorb uterine fluid; changes in the thickness of the endometrium and its blood supply development, and the formation of the decidua. Collectively these changes are known as plasma membrane transformation, and bring the blastocyst nearer to the endometrium and immobilize it. During this stage the blastocyst can still be eliminated by being flushed out of the uterus.[34][35]
Successful implantation is co-dependent on the viability of the embryo, and the receptivity of the uterus.[4] A critical involved factor is the developmental synchrony between the embryo and the uterus.[36] The synchrony gives a short period of receptivity known as the window of implantation, and involves much crosstalk between the blastocyst and the endometrium at this stage.[37][38][39]
The endocannabinoid system plays a vital role in this synchrony in the uterus, influencing uterine receptivity, and embryo implantation.[5] The embryo expresses cannabinoid receptors early in its development that are responsive to anandamide (AEA) secreted in the uterus. This signaling is of importance in the embryo-uterus crosstalk in regulating the timing of embryonic implantation and uterine receptivity. Adequate concentrations of AEA that are neither too high or too low, are needed for successful implantation.[5][40] IL-6 and FAAH are both crucial for uterine receptivity and together with AEA there is seen to be a link with adequate endometrial thickness that sustains pregnancy.[5]
During adhesion the cross-talk is conveyed by receptor-ligand-interactions, both integrin-matrix and proteoglycan ones. Proteoglycan receptors are found on the surface of the decidua, and their counterparts, the proteoglycans, are found around the trophoblast cells of the blastocyst. This ligand-receptor system is also present just at the implantation window.[30] The blastocyst signals to the endometrium to adapt further to its presence, for example by changes in the cytoskeleton of decidual cells. This, in turn, dislodges the decidual cells from their connection to the underlying basal lamina, which enables the blastocyst to perform the succeeding invasion.[30]
Window of implantation
The window of implantation is a limited timeframe for the successful attachment of the blastocyst.[41] In humans uterine receptivity is optimum on days 20-24 of the secretory phase of the menstrual cycle when luteinizing hormone levels are at their peak.[9][42] The crosstalk between the embryo and the endometrium takes place during this time.[9] The endothelial epithelial cells lining the uterus are the first cells to detect signals from the blastocyst, and they are transduced into downstream signalling pathways.[33] In humans the window of implantation is only available for 24–36 hours.[43]
The endometrial microbiome has been indicated as having an important role in successful implantation in controlling endometrial cell function, and the function of the local immunity system that prevents pathogen growth. This is associated with the secretion of protective substances.[44][45]
Pinopodes
Pinopodes are formed at the beginning of the window of implantation, and are found in many species.[46][41] They are mushroom-like protrusions from the apical cell membrane of uterine epithelial cells.[41] Pinopodes are formed by the swelling of these epithelial cells, and the fusing together of a number of microvilli, to reach a maximum size.[46] They appear between day 19 and day 21 of
Their development is enhanced by
Pinopodes bring uterine fluid and its macromolecules into the cells by the process of endocytosis. This decreases the volume of the uterus, taking the walls closer to the blastocyst floating in it. Thus, the period of active pinopodes might limit the implantation window.[30] Pinopodes continue to absorb fluid, removing most of it during the early stages of implantation.
Predecidualization
The endometrium increases thickness, becomes vascularized and its glands grow to be tortuous and boosted in their secretions. These changes reach their maximum about seven days after ovulation.
Furthermore, the surface of the endometrium produces a kind of rounded cells, which cover the whole area toward the uterine cavity. This happens about 9 to 10 days after ovulation.
The decidual cells originate from the stromal cells that are always present in the endometrium, and make up a new layer, the
Decidualization

Decidualization expands if pregnancy occurs, further developing the uterine glands, the zona compacta and the epithelium of decidual cells lining it. The decidual cells become filled with lipids and glycogen and take the polyhedral shape characteristic of decidual cells. Factors from the blastocyst also trigger the final formation of decidual cells into their proper form. In contrast, some decidual cells in the proximity of the blastocyst degenerate, providing nutrients for it.[30] An indication of embryonic influence is that decidualization occurs at a higher degree in conception cycles than in nonconception cycles.[30] Furthermore, similar changes are observed when giving stimuli mimicking the natural invasion of the embryo.[30]
The embryo releases serine proteases which causes the epithelial cell membrane to
Parts of decidua
The decidua can be organized into separate sections, although they have the same composition.
- Decidua basalis – This is the part of the decidua which is located basalolateral to the embryo after implantation.
- Decidua capsularis – Decidua capsularis grows over the embryo on the luminal side, enclosing it into the endometrium. It surrounds the embryo together with decidua basalis.
- Decidua parietalis – All other decidua on the uterine surface belongs to decidua parietalis.
Decidua throughout pregnancy
After implantation the decidua remains, at least through the first trimester.[30] However, its most prominent time is during the early stages of pregnancy, during implantation. Its function as a surrounding tissue is replaced by the definitive placenta. However, some elements of the decidualization remain throughout pregnancy.[30]
The compacta and spongiosa layers are still observable beneath the decidua in pregnancy. The glands of the spongiosa layer continue to secrete during the first trimester, when they degenerate. However, before that disappearance, some glands secrete unequally much. This phenomenon of hypersecretion is called the
Uterine glands

| Proteins, glycoproteins and peptides
secreted by the uterine glands[30] |
| Matrix-associated: |
| Fibronectin |
| Laminin |
Entactin
|
Type-IV collagen
|
| Heparan sulfate |
| Proteoglycan |
Integrins
|
| – |
| Others: |
| Mucins |
| Prolactin |
IGFBP -1
|
| Glycodelin |
| Endometrial protein 15 |
| Albumin |
| Beta-Lipoprotein |
| Relaxin |
Fibroblast growth factor 1
|
Fibroblast growth factor 2
|
| Pappalysin-1 |
| Stress response protein 27 (SRP-27) |
CA-125
|
Beta-endorphin
|
| Leu-enkephalin |
| Diamine oxidase |
Tissue plasminogen activator
|
| Renin |
| Progesterone-dependent carbonic anhydrase |
| Lactoferrin |
Not only the lining of the uterus transforms, but the secretion from its glands changes. This change is induced by increased levels of progesterone from the corpus luteum. The target of the secretions is the embryoblast, and has several functions on it.
Nourishment
The embryo spends approximately 72 hours in the uterine cavity before implanting. In that time, it cannot receive nourishment directly from the blood of the mother, and must rely on secreted nutrients into the uterine cavity, e.g. iron and fat-soluble vitamins.[30]
Growth and implantation
In addition to nourishment, the endometrium secretes several
Clinical significance
Implantation failure
Reproduction in humans is not very efficient. Only around 30% of natural conceptions result in successful pregnancies. Of the failed pregnancies around 85% are due to implantation failure.
Inadequate uterine receptivity may be caused by abnormal
In women with more than
Zinc deficiency
Zinc is crucial in pre-conception, (and successful pregnancy), and its deficiency can lead to incompetent blastocyst development. Once an egg is fertilized zinc is released in a zinc spark which promotes changes that include the hardening of the zona pellucida preventing polyspermy.[55]
Implantation bleeding
Bleeding and spotting are common during the luteal phase of the menstrual cycle, and early stages of pregnancy, but are unrelated to implantation. Implantation bleeding occurs between 7 and 14 days after fertilization,[56] and is a small amount of light vaginal bleeding or spotting that can occur in early pregnancy due to the blastocyst penetrating the lining of the uterus during implantation.[57][58][59] By day 13 the penetration site in the endometrium has usually been closed by a fibrin plug but increased blood flow into the syncytiotrophoblast spaces can sometimes cause bleeding at that site.[57] Implantation bleeding may be accompanied by symptoms such as cramping, nausea, breast tenderness, and headaches.[60] Implantation bleeding can be distinguished from period bleeding by color, clotting, strength and duration of flow.[61][62]
See also
- Cannabinoids
- Embryonic diapause
- Hofbauer cells
- Homeostatic capacity
- Human overpopulation
- Reichert's membrane
References
- ^ "Definition of implantation". www.merriam-webster.com. Retrieved 26 April 2023.
- PMID 29716588.
- ^ PMID 32296829.
- ^ PMID 34558134.
- ^ S2CID 220943305.
- S2CID 39733100.
- PMID 14749627.
- ^ PMID 29259417.
- ^ PMID 29359200.
- ^ PMID 25023681.
- ^ ISBN 978-0-323-61154-1.)
{{cite book}}: CS1 maint: location missing publisher (link - PMID 20729534.
- PMID 32183093.
- ^ PMID 35661923.
- ISBN 978-0-7817-9069-7.
- ^ Boron 2004, p. 1172.
- ^ S2CID 11540123.
- ^ IVF-infertility.com
- PMID 19453302.
- S2CID 44999196.
- ^ PMID 20826587.
- PMID 16720361.
- PMID 33596915.
- S2CID 15462671.
- PMID 23483768.
- PMID 25602074.
- ^ PMID 32202400.
- PMID 22022598.
- PMID 35393235.
- ^ OCLC 61527528.[page needed]
- PMID 27774579.
- PMID 19262088.
- ^ PMID 37060079.
- S2CID 24023502.
- S2CID 22769575.
- PMID 27480540.
- PMID 34440776.
- PMID 36362169.
- PMID 29162091.
- S2CID 39733100.
- ^ PMID 31738970.
- PMID 19361790.
- PMID 32521725.
- PMID 31213058.
- PMID 27717732.
- ^ PMID 34496883.
- S2CID 5116022.
- ^ PMID 35784461.
- PMID 24306146.
- ^ PMID 20729534.
- PMID 34363466.
- PMID 35118399.
- PMID 23912476.
- PMID 30117155.
- PMID 33598687.
- ^ "Implantation Bleeding vs A Period: How To Tell The Difference". ReUnite Rx. 2020-12-09. Retrieved 2021-07-29.
- ^ ISBN 978-0-7817-9069-7.
- ISBN 978-1-4419-9810-1.
- PMID 12923154.
- ^ Heger, L. Demosthenes E (ed.). "What do implantation cramps feel like? How to tell the difference between implantation and menstrual cramping". Insider. Retrieved 2021-07-29.
- S2CID 212812977.
- ^ "How Do I Know if It's Implantation Bleeding?". MedicineNet. Retrieved 2021-07-29.
Books
- OCLC 61527528.
