Osmium(V) chloride
Appearance
 | |
| Names | |
|---|---|
| Other names
pentachloroosmium, osmium pentachloride
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| |
SMILES
| |
| Properties | |
| Cl5Os | |
| Molar mass | 367.48 g·mol−1 |
| Appearance | black solid |
| Density | 4.09 |
| Melting point | 160 °C (320 °F; 433 K) |
| soluble | |
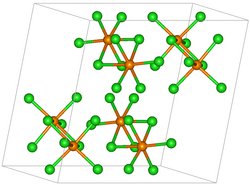
| Structure | |
| P21/c[1] | |
a = 9.17 Å, b = 11.5 Å, c = 11.97 Å α = 90°, β = 109°°, γ = 90°.[2]
| |
Lattice volume (V)
|
1194 Å3 |
Formula units (Z)
|
8 |
| Related compounds | |
Related compounds
|
Osmium pentafluoride; Rhenium pentachloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Osmium(V) chloride is an
Potential synthesis
Osmium(V) chloride can be obtained in small amounts by reacting osmium hexafluoride with excess boron trichloride:[5]
- 2OsF6 + 4BCl3 → 2OsCl5 + 4BF3 + Cl2
It can also be obtained by the action of sulfur dichloride on osmium tetroxide:[2]
- 2OsO4 + 8SCl2 + 5Cl2 → 2OsCl5 + 8SOCl2
Physical properties
Osmium(V) chloride forms a black dimeric solid, isomorphic with
POCl3 to form a red-brown solution, which can crystallize OsCl5·POCl3.[6]
References
- ^ "Crystallography Open Database". qiserver.ugr.es. Retrieved 31 March 2023.
- ^ ISSN 0020-1669. Retrieved 31 March 2023.
- ISBN 978-0-12-352651-9. Retrieved 31 March 2023.
- .
- ISBN 978-0-412-30120-9. Retrieved 31 March 2023.
- ISSN 1865-7117.
