Immune thrombocytopenic purpura
| Immune thrombocytopenic purpura | |
|---|---|
| Other names | Idiopathic thrombocytopenic purpura, idiopathic immune thrombocytopenia, primary immune thrombocytopenia, idiopathic thrombocytopenic purpura, primary immune thrombocytopenic purpura, autoimmune thrombocytopenic purpura |
 | |
| Petechiae, or small bruise-like markings, may occur in ITP | |
| Specialty | Hematology |
| Frequency | 50–100 per million (12-month risk) |
Immune thrombocytopenic purpura (ITP), also known as idiopathic thrombocytopenic purpura or immune thrombocytopenia, is a type of
ITP is considered an
Diagnosis of ITP involves identifying a low platelet count through a
For mild cases, careful observation may be sufficient. However, in instances of very low platelet counts or significant bleeding, treatment options may include
Signs and symptoms
Signs of ITP include the spontaneous formation of bruises (
In cases where platelet counts drop to extremely low levels (<5,000 per μl), serious and potentially fatal complications may arise. These complications include
-
Petechiae on the lower extremities
-
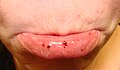
Oral petechiae/purpura - lower lip
-
Petechia on the tongue in a person with platelets of 3 due to ITP
-
Petechia of the lower leg in a person with platelets of 3 due to ITP
Pathogenesis
In approximately 60 percent of cases,
The coating of platelets with IgG renders them susceptible to
The stimulus for auto-antibody production in ITP is probably abnormal T cell activity.[8][9][10] Preliminary findings suggest that these T cells can be influenced by medications that target B cells, such as rituximab.[11]
Diagnosis

The diagnosis of ITP is a process of exclusion. First, it has to be determined that there are no blood abnormalities other than a low platelet count, and no physical signs other than bleeding. Then, secondary causes (5–10 percent of suspected ITP cases) should be excluded. Such secondary causes include leukemia, medications (e.g., quinine, heparin), lupus erythematosus, cirrhosis, HIV, hepatitis C, congenital causes, antiphospholipid syndrome, von Willebrand factor deficiency, onyalai and others.[4][12] All patients with presumed ITP should be tested for HIV and hepatitis C virus, as platelet counts may be corrected by treating the underlying disease. In approximately 2.7 to 5 percent of cases, autoimmune hemolytic anemia and ITP coexist, a condition referred to as Evans syndrome.[13][14]
Despite the destruction of platelets by splenic macrophages, the spleen is normally not enlarged. In fact, an enlarged spleen should lead to a search for other possible causes for the thrombocytopenia. Bleeding time is usually prolonged in ITP patients. However, the use of bleeding time in diagnosis is discouraged by the American Society of Hematology practice guidelines[15] and a normal bleeding time does not exclude a platelet disorder.[16]
Bone marrow examination may be performed on patients over the age of 60 and those who do not respond to treatment, or when the diagnosis is in doubt.[12] On examination of the marrow, an increase in the production of megakaryocytes may be observed and may help in establishing a diagnosis of ITP. An analysis for anti-platelet antibodies is a matter of clinician's preference, as there is disagreement on whether the 80 percent specificity of this test is sufficient to be clinically useful.[12]
Treatment
With rare exceptions, there is usually no need to treat based on platelet counts. Many older recommendations suggested a certain platelet count threshold (usually somewhere below 20.0/µl) as an indication for hospitalization or treatment. Current guidelines recommend treatment only in cases of significant bleeding. Treatment recommendations sometimes differ for adult and pediatric ITP.[17]
Steroids
Initial treatment usually consists of the administration of
Anti-D
Another option, suitable for
Steroid-sparing agents
There is increasing use of
Intravenous immunoglobulin
Thrombopoietin receptor agonists
Thrombopoietin receptor agonists are pharmaceutical agents that stimulate platelet production in the bone marrow. In this, they differ from the previously discussed agents that act by attempting to curtail platelet destruction.[28] Two such products are currently available:
- Food and Drug Administration (FDA) for long-term treatment of adult chronic ITP on August 22, 2008.[31]
- GlaxoSmithKline and also designated an orphan drug by the FDA, Promacta was approved by the FDA on November 20, 2008.[33]
Thrombopoietin receptor agonists exhibited the greatest success so far in treating patients with refractory ITP.[34]
Side effects of thrombopoietin receptor agonists include headache, joint or muscle pain, dizziness, nausea or vomiting, and an increased risk of blood clots.[28]
Surgery
Platelet transfusion
Platelet transfusion alone is normally not recommended except in an emergency and is usually unsuccessful in producing a long-term platelet count increase. This is because the underlying autoimmune mechanism that is destroying the patient's platelets will also destroy donor platelets, and so platelet transfusions are not considered a long-term treatment option.[41]
H. pylori eradication
In adults, particularly those living in areas with a high prevalence of
Other agents
- Dapsone (also called diphenylsulfone, DDS, or avlosulfon) is an anti-infective sulfone medication. Dapsone may also be helpful in treating lupus, rheumatoid arthritis, and as a second-line treatment for ITP. The mechanism by which dapsone assists in ITP is unclear but an increased platelet count is seen in 40–60 percent of recipients.[44][45]
- The off-label use of randomized controlled trials are inconclusive.[46]
Prognosis
In general patients with acute ITP will only rarely have life-threatening bleeding.[47] most of the patients ultimately have stable but lower platelet counts which is hemostatic for a person. Unlike in pediatric patients who can be cured, most of the adults will run a chronic course even after splenectomy.[48]
Epidemiology
A normal platelet count is considered to be in the range of 150,000–450,000 per microlitre (μl) of blood for most healthy individuals. Hence one may be considered thrombocytopenic below that range, although the threshold for a diagnosis of ITP is not tied to any specific number.[49]
The incidence of ITP is estimated at 50–100 new cases per million per year, with children accounting for half of that number. At least 70 percent of childhood cases will end up in remission within six months, even without treatment.[50][51][52] Moreover, a third of the remaining chronic cases will usually remit during follow-up observation, and another third will end up with only mild thrombocytopenia (defined as a platelet count above 50,000).[50] A number of immune related genes and polymorphisms have been identified as influencing predisposition to ITP, with FCGR3a-V158 allele and KIRDS2/DL2 increasing susceptibility and KIR2DS5 shown to be protective.[53][54]
ITP is usually chronic in adults
The
Pregnancy
The incidence of ITP in pregnancy is not well known. It may occur during any trimester of pregnancy. It is the most common cause of significant thrombocytopenia (platelets less than 100,000) in the second trimester, and it is a common cause of significant thrombocytopenia in the first and third trimesters.
Women with ITP often have a decrease in their platelet counts when they become pregnant, often requiring treatment.
Anti-platelet autoantibodies in a pregnant woman with ITP will attack the patient's own platelets and will also cross the placenta and react against fetal platelets leading to thrombocytopenia. Thrombopoietin levels are also increased during pregnancy (which is thought to be due to placental production of estradiol) and this estradiol leads to decreased megakaryocyte activity and therefore decreased platelet production.[58] Therefore, ITP is a significant cause of fetal and neonatal immune thrombocytopenia. Approximately 10% of newborns affected by ITP will have platelet counts <50,000/uL and 1% to 2% will have a risk of intracerebral hemorrhage, comparable to that of infants with neonatal alloimmune thrombocytopenia (NAIT).[59][60]
No lab test can reliably predict if neonatal thrombocytopenia will occur. The risk of neonatal thrombocytopenia is increased with:[61]
- Mothers with a history of splenectomy for ITP
- Mothers who had a previous infant affected with ITP
- Gestational (maternal) platelet count less than 100,000/uL
It is recommended that pregnant women with thrombocytopenia or a previous diagnosis of ITP be tested for serum antiplatelet antibodies. Indications for treatment of pregnant people with ITP include the presence of bleeding, platelet counts less than 20-30,000, planned procedures (such as an
History
After initial reports by the Portuguese physician
References
- ^ PMID 32965953.
- PMID 19005182.
- PMID 28416506.
- ^ PMID 15660520.
- ^ a b "Immune Thrombocytopenic Purpura". The Lecturio Medical Concept Library. Retrieved 27 July 2021.
- PMID 12522773.
- S2CID 9765360.
- PMID 1840468.
- S2CID 206867628.
- PMID 18420827.
- PMID 18463354.
- ^ PMID 15941913.
- S2CID 195879785.
- PMID 19638626.
- S2CID 53089071.
- PMID 9081003.
- PMID 21325604.
- ^ PMID 17122451.
- S2CID 23828538.
- S2CID 38433249.
- PMID 15032608.
- ISBN 9783527326693.
- ISBN 9781498704304.
- PMID 15141084.
- PMID 22609781.
- ISBN 9789811041426.
- S2CID 27193777.
- ^ a b "Idiopathic thrombocytopenic purpura: Treatments and Drugs". Mayo Clinic. Retrieved October 16, 2012.
- PMID 17050891.
- S2CID 22991485.
- ^ "AMGEN - Press Releases". Archived from the original on 2008-09-15. Retrieved 2008-08-23.
- PMID 18046028.
- GlaxoSmithKline. 2008-11-20. Archived from the originalon 2010-12-27. Retrieved 2008-11-25.
- ^ ISSN 2076-6327.
- PMID 29295846.
- PMID 15642672.
- PMID 15100149.
- PMID 23144195.
- S2CID 23025470.
- S2CID 4778713.
- ISSN 0006-4971.
- PMID 18945961.
- PMID 19846889.
- S2CID 23283382.
- PMID 24310318.
- ^ National Institute for Health and Care Excellence. Immune (idiopathic) thrombocytopenic purpura: rituximab. October 2014. [1]
- PMID 7733121– via elsevier.
- PMID 15100149.
- S2CID 14775350.
- ^ S2CID 43752571.
- PMID 17460024.
- PMID 17352309.
- S2CID 3327902.
- PMID 24571473.
- PMID 11919310.
- ^ "Amgen to Discuss Romiplostim BLA". drugs.com. March 12, 2008. Retrieved 2008-11-04.
- S2CID 45178771.
- ^ S2CID 260787282.
- ^ a b Roback et al. AABB Technical Manual, 16th Ed. Bethesda, AABB Press, 2008.
- S2CID 29019817.
- ^ a b Mais DD. ASCP Quick Compendium of Clinical Pathology. Chicago: ASCP Press, 2009.
- ^ S2CID 37150678.
- Who Named It?
- PMID 19074083.