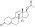
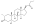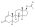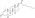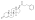Trenbolone acetate
 | |
| Clinical data | |
|---|---|
| Trade names | Finajet, Finaplix, others |
| Other names | RU-1697; Trenbolone 17β-acetate; 19-Nor-δ9,11-testosterone 17β-acetate; Estra-4,9,11-trien-17β-ol-3-one 17β-acetate |
| Routes of administration | Intramuscular injection |
| Drug class | Androgen; Anabolic steroid; Androgen ester; Progestogen |
| Pharmacokinetic data | |
| Elimination half-life | Intramuscular: 3 days[1] |
| Identifiers | |
| |
JSmol) | |
| |
| |
Trenbolone acetate, sold under brand names such as Finajet and Finaplix among others, is an
Trenbolone acetate was discovered in 1963 and was introduced for veterinary use in the early 1970s.[5][10][11] In addition to its veterinary use, trenbolone acetate is used to improve physique and performance, and is purchased from black market suppliers.[5] The drug is a controlled substance in many countries and so non-veterinary use is generally illicit.[5]
Uses
Veterinary uses

In the livestock industry, trenbolone acetate is more often called Finaplix. It was intentionally developed to promote androgen and gain muscle mass in cattle. Due to its properties, this allows livestock to grow as much muscle as possible before they are transported to a slaughterhouse.

Non-medical uses
Bodybuilding
Trenbolone acetate was never approved for use in humans and therefore guidelines for human consumption do not exist.
Medical uses
Trenbolone acetate was never approved for use in humans and hence has no medical uses.
Trenbolone was produced for human use as a human grade product, but only once. The product came in 1.5ml ampoules consisting of 76.2 mg of Trenbolone hexahydrobenzylcarbonate and made by Negma Pharmaceuticals of France. [citation needed]
Side effects
Trenbolone acetate, like any other AAS, has many
Androgenic
Trenbolone acetate has androgenic activity.
Hypogonadism
Trenbolone acetate contributes greatly to muscle mass and feed efficiency; however, administration of the AAS suppresses natural testosterone production; i.e., it has the potential to cause hypogonadism.[5][14][19] This is a common effect of all AAS; the only difference is the variation in how much they suppress in comparison to others.
Cardiovascular
Administration of any AAS can lead to cardiovascular issues.[20] Trenbolone acetate can have a negative and strong impact on cholesterol through suppressing both high-density lipoprotein (HDL) cholesterol and increasing low-density lipoprotein (LDL) cholesterol.[21] When compared to oral AAS, trenbolone acetate has a stronger negative effect on cholesterol levels. This negative effect is much more severe with the use of injectable AAS, particularly trenbolone acetate.[22]
"Tren cough"
The exact mechanisms underlying the cause of the tren cough are not known; however, trenbolone acetate's androgenic effect activates a variety of lipid-like active compounds which are called
Estrogenic and progestogenic
No form of trenbolone, including trenbolone acetate, is
Pharmacology
Pharmacodynamics

Trenbolone acetate is a
Similar to many other AAS, trenbolone acetate has the capability to produce
Trenbolone acetate also has the ability to increase red blood cell count. With a larger amount of
Pharmacokinetics
The
Chemistry
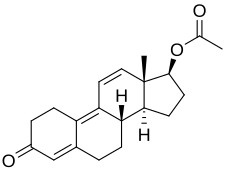
Trenbolone acetate, or trenbolone 17β-acetate, is a
| Anabolic steroid | Structure | Ester | Relative mol. weight |
Relative AAS contentb |
Durationc | ||||
|---|---|---|---|---|---|---|---|---|---|
| Position | Moiety | Type | Lengtha | ||||||
| Boldenone undecylenate | C17β | Undecylenic acid | Straight-chain fatty acid | 11 | 1.58 | 0.63 | Long | ||
| Drostanolone propionate | C17β | Propanoic acid |
Straight-chain fatty acid | 3 | 1.18 | 0.84 | Short | ||
| Metenolone acetate | C17β | Ethanoic acid |
Straight-chain fatty acid | 2 | 1.14 | 0.88 | Short | ||
| Metenolone enanthate | C17β | Heptanoic acid |
Straight-chain fatty acid | 7 | 1.37 | 0.73 | Long | ||
| Nandrolone decanoate | C17β | Decanoic acid |
Straight-chain fatty acid | 10 | 1.56 | 0.64 | Long | ||
| Nandrolone phenylpropionate | C17β | Phenylpropanoic acid | Aromatic fatty acid | – (~6–7) | 1.48 | 0.67 | Long | ||
| Trenbolone acetate | C17β | Ethanoic acid |
Straight-chain fatty acid | 2 | 1.16 | 0.87 | Short | ||
| Trenbolone enanthated | C17β | Heptanoic acid |
Straight-chain fatty acid | 7 | 1.41 | 0.71 | Long | ||
| Footnotes: a = Length of oil solution . d = Never marketed. Sources: See individual articles.
| |||||||||
Structure–activity relationships
Trenbolone acetate is a modified form of nandrolone.[16] The structure of trenbolone acetate is a 19-nor classification, which represents a structural change of the testosterone hormone. Trenbolone acetate lacks a carbon atom at the 19 position and carries a double bond at carbons 9 and 11. The position of these carbons slows its metabolism, which greatly increases its binding affinity to the AR, and inhibits it from undergoing aromatization into the corresponding estrogenic metabolite. Trenbolone acetate contains trenbolone modified with the addition of a carboxylic acid ester (acetic acid) at the 17β-hydroxyl group.[10] This facilitates the slow release of the AAS from the area of injection.
History
Trenbolone acetate was first synthesized in 1963 and approved by the livestock industry as a growth promoter for beef cattle in the early 1970s.[5][10][11] During this period of its first administration, trenbolone acetate was sold under the names Finajet and Finaject. The original manufacturer of trenbolone acetate discontinued during the late 1980s and administered the synthesis of subcutaneous pellets called Finaplix. These pellets aimed to increase muscle mass and lean tissue of cattle prior to slaughter to increase the profitability of livestock when measured in total pounds of meat sold.[10]
The drug appears to have been an early development project of
Trenbolone acetate became popular among bodybuilders and athletes during the early 1980s. During this period, the AAS was transported illegally from Europe in large quantities. Although trenbolone acetate was very popular for a short amount of time, the large amounts of supplies were discontinued in 1987.
Society and culture
Generic names
Trenbolone acetate is the
Brand names
Trenbolone acetate is or has been sold alone for veterinary use under the brand names Component TH, Component TS, Finaject, Finajet, Finaplix-H, and Finaplix-S.[3][4][5][36][37] It is or has also been sold in combination with estradiol or estradiol benzoate for veterinary use under the brand names Revalor and Synovex.[3][4][5][36][37]
Distribution and regulation
Trenbolone acetate, specifically referred to as Finaplix in the livestock industry, is available to purchase in veterinary drug markets.
Doping in sports
Regardless of their legality, AAS are still banned by most sporting leagues in the country, who routinely conduct drug tests to find the users of any AAS. There are known cases of doping in sports with trenbolone acetate by professional athletes.
References
- ^ ISBN 978-1-60547-277-5.
- ^ S2CID 205253265.
- ^ ISBN 978-94-011-4439-1.
- ^ a b c d "Trenbolone". Archived from the original on 2020-07-07. Retrieved 2017-11-11.
- ^ ISBN 978-0-9828280-1-4.
- S2CID 31765638.
- ^ PMID 18500378.
- ^ ISSN 0804-4643.
- ^ PMID 7382482.
- ^ S2CID 4706788.
- ^ PMID 16818248.
- PMID 24278538.
- PMID 11701661.
- ^ PMID 1874853.
- ^ "Trenbolone Acetate - A comprehensive list of side effects". trenacetate.biz. 21 June 2020. Retrieved 2021-02-22.
- ^ PMID 19447393.
- ^ S2CID 22776408.
- ^ S2CID 8804818.
- ^ PMID 2170557.
- PMID 15084526.
- PMID 26584015.
- PMID 26495342.
- PMID 12007900.
- S2CID 21643058.
- S2CID 6040673.
- ^ PMID 19880691.
- PMID 12441365.
- ISBN 978-0-9828280-1-4.
- ISBN 978-1-139-49430-4.
- PMID 782871.
- PMID 20138077.
- ^ PMID 18403176.
- ^ PMID 6193805.
- PMID 20398732.
- ^ ISSN 0003-3561.
- ^ ISBN 978-1-4757-2085-3.
- ^ ISBN 978-3-88763-075-1.
- ^ "Anabolic Steroids".
- ^ "Trenbolone Acetate 100 Norma".
Further reading
- Meyer HH (January 2001). "Biochemistry and physiology of anabolic hormones used for improvement of meat production". APMIS. 109 (1): S336–S344. S2CID 23149070.
- Yarrow JF, McCoy SC, Borst SE (June 2010). "Tissue selectivity and potential clinical applications of trenbolone (17beta-hydroxyestra-4,9,11-trien-3-one): A potent anabolic steroid with reduced androgenic and estrogenic activity". Steroids. 75 (6): 377–389. S2CID 205253265.