Trenbolone
 | |
| Clinical data | |
|---|---|
| Other names | Trienolone; Trienbolone; RU-2341; Δ9,11-Nandrolone; 19-Nor-δ9,11-testosterone; Estra-4,9,11-trien-17β-ol-3-one |
| AHFS/Drugs.com | International Drug Names |
| License data |
|
| Pregnancy category |
|
esters) | |
| Drug class | Androgen; Anabolic steroid; Progestogen |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Intramuscular: 80-100%[citation needed] |
| Metabolism | Liver |
| Elimination half-life | 6–8 hours[citation needed] |
| Excretion | Urine |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Trenbolone is an

Uses
Veterinary
Trenbolone, as trenbolone acetate, improves muscle mass, feed efficiency, and mineral absorption in cattle.[6]
Side effects
Sometimes human users may experience an event called "tren cough" shortly after or during an injection, where the user experiences a violent and extreme coughing fit, which can last for minutes and in some cases even longer.
"Tren cough", despite its name, is not exclusive to trenbolone. It can occur when injecting any oil-steroid solutions, if the solution accidentally is injected intravenously. When the oil-steroid solution gets into the bloodstream, the steroid oil solution travels into the lungs, therefore causing a coughing fit. There exist several theories on why this phenomenon happens.[9]
It is possible that the androgenic effect from steroids, activates a variety of lipid-like active compounds which are called
Pharmacology
Pharmacodynamics
Trenbolone has both
Studies on metabolism are mixed, with some studies showing that it is metabolized by
The potency of Trenbolone is not known, although it's often falsely believed to be five times high as that of
Pharmacokinetics
To prolong its
Trenbolone and 17-epitrenbolone are both excreted in urine as conjugates that can be hydrolyzed with beta-glucuronidase.[24] This implies that trenbolone leaves the body as beta-glucuronides or sulfates.
Chemistry
Trenbolone, also known as 19-nor-δ9,11-testosterone or as estra-4,9,11-trien-17β-ol-3-one, is a
| Name: | Trenbolone | Trenbolone acetate | Trenbolone enanthate | Trenbolone hexahydrobenzylcarbonate
(cyclohexylmethylcarbonate) |
|---|---|---|---|---|
| Structural[25] | 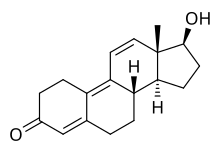
|

|

|

|
| Formula | C18H22O2 | C20H24O3 | C25H34O3 | C26H34O4 |
| Crystal system[25] | monocrystalic | monocrystalic | monocrystalic | |
| Elimination half life | 48–72 hours[citation needed] | short | long
11 days[25] |
8 days[25] |
History
Trenbolone was first synthesized in 1963.[27]
Society and culture
Generic names
Trenbolone is the
Legal status
Some
Doping in sports
There are known cases of doping in sports with trenbolone esters by professional athletes.
See also
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-15.
- ^ ISBN 978-1-4757-2085-3. Archivedfrom the original on 11 January 2023. Retrieved 11 November 2017.
- ^ ISBN 978-3-88763-075-1.
- ^ ISBN 978-94-011-4439-1. Archivedfrom the original on 11 January 2023. Retrieved 11 November 2017.
- ^ "Trenbolone". Archived from the original on 2020-07-07. Retrieved 2017-11-11.
- ^ ISBN 978-0-9828280-1-4.
- ^ Nichols W, Hutcheson J, Streeter M, Corrigan M, Nuttelman B. "Implant Strategies for Finishing Cattle using Revalor® (trenbolone acetate and estradiol), Finaplix® (trenbolone) and/or Ralgro® (zeranol)" (PDF). Merck Animal Health. Archived (PDF) from the original on 2012-10-13. Retrieved 2011-08-22.
- PMID 18500378.
- PMID 22059184.
- PMID 12007900.
- PMID 10792135.
- PMID 8673930.
- ^ Fahey TD (March 1998). "Anabolic Steroids: Mechanisms and Effects". Encyclopedia of sports medicine and science. Internet Society for Sport Science. Archived from the original on 2011-08-23. Retrieved 2011-08-23.
- PMID 12441365.
- S2CID 205253265.
- PMID 6361192.
- ISBN 978-0-9828280-1-4.
- ISBN 978-1-139-49430-4.
- PMID 782871.
- PMID 20138077.
- ISBN 978-1-139-49430-4.
- ^ ISBN 9788716164575.
- ISBN 978-1-4684-3824-6.
- PMID 8674183.
- ^ S2CID 216299984.
- ISBN 978-1-60547-277-5.
- PMID 8674183.
- ISBN 978-92-5-102933-6.
- ^ "Trenbolone hexahydrobenzylcarbonate use in bodybuilding". 20 November 2021. Archived from the original on 24 November 2021. Retrieved 24 November 2021.
- ^ "Controlled Substances Act". United States Food and Drug Administration. 11 June 2009. Archived from the original on 2 March 2017. Retrieved 17 June 2016.
- ^ "Controlled Drugs and Substances Act". laws-lois.justice.gc.ca. Archived from the original on 2012-09-15.
- ^ "Consideration of the Anabolic Steroids". London: Advisory Council on the Misuse of Drugs. September 2010. Archived from the original on 2011-09-22.
- ^ "Australian Institute of Criminology - Steroids". Archived from the original on 2012-03-23. Retrieved 2011-08-22.
Further reading
- Meyer HH (January 2001). "Biochemistry and physiology of anabolic hormones used for improvement of meat production". APMIS. 109 (1): 1–8. S2CID 23149070.
- Yarrow JF, McCoy SC, Borst SE (June 2010). "Tissue selectivity and potential clinical applications of trenbolone (17beta-hydroxyestra-4,9,11-trien-3-one): A potent anabolic steroid with reduced androgenic and estrogenic activity". Steroids. 75 (6): 377–389. S2CID 205253265.
