Tunicate
| Tunicates | |
|---|---|

| |
| Gold-mouth sea squirt (Polycarpa aurata) | |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Chordata |
| Clade: | Olfactores |
| Subphylum: | Tunicata Lamarck, 1816[4][5] |
| Classes and unplaced genera[4][7] | |
| |
| Synonyms | |
|
Urochordata Lankester, 1877 | |
A tunicate is a marine
Some tunicates live as solitary individuals, but others replicate by
Various species of
The earliest probable species of tunicate appears in the fossil record in the early
Taxonomy


About 3,000 species of tunicate exist in the world's oceans, living mostly in shallow water. The most numerous group is the
The Tunicata were established by Jean-Baptiste Lamarck in 1816. In 1881, Francis Maitland Balfour introduced another name for the same group, "Urochorda", to emphasize the affinity of the group to other chordates.[13] No doubt largely because of his influence, various authors supported the term, either as such, or as the slightly older "Urochordata", but this usage is invalid because "Tunicata" has precedence, and grounds for superseding the name never existed. Accordingly, the current (formally correct) trend is to abandon the name Urochorda or Urochordata in favour of the original Tunicata, and the name Tunicata is almost invariably used in modern scientific works. It is accepted as valid by the World Register of Marine Species[14] but not by the Integrated Taxonomic Information System.[15]
Various common names are used for different species. Sea tulips are tunicates with colourful bodies supported on slender stalks.[16] Sea squirts are so named because of their habit of contracting their bodies sharply and squirting out water when disturbed.[17] Sea liver and sea pork get their names from the resemblance of their dead colonies to pieces of meat.[18]
Classification
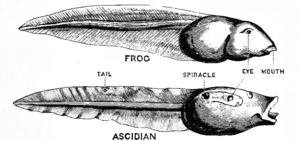
Tunicates are more closely related to craniates (including hagfish, lampreys, and jawed vertebrates) than to lancelets, echinoderms, hemichordates, Xenoturbella or other invertebrates.[19][20][21]
The clade consisting of tunicates and vertebrates is called Olfactores.[22]
The Tunicata contain roughly 3,051 described species,[12] traditionally divided into these classes:
- Ascidiacea (Aplousobranchia, Phlebobranchia, and Stolidobranchia)
- Thaliacea (Pyrosomida, Doliolida, and Salpida)
- Copelata)
Members of the
The following cladogram is based on the 2018 phylogenomic study of Delsuc and colleagues.[25]
| Tunicata |
| |||||||||||||||||||||||||||
Fossil record

Undisputed fossils of tunicates are rare. The best known and earliest unequivocally identified species is
Three enigmatic species were also found from the
Fossils of tunicates are rare because their bodies decay soon after death, but in some tunicate families, microscopic spicules are present, which may be preserved as microfossils. These spicules have occasionally been found in Jurassic and later rocks, but, as few palaeontologists are familiar with them, they may have been mistaken for sponge spicules.[32]
In Permian and Triassic there were also forms with a calcareous exoskeleton. At first they were mistaken for corals.[33][34]
Hybridization studies
A multi-taxon
Anatomy
Body form

Colonies of tunicates occur in a range of forms, and vary in the degree to which individual organisms, known as
Body structure
By far the largest class of tunicates is the
The
A third class, the
Physiology and internal anatomy


Like all other
Ascidian tunicates begin life as a lecithotrophic (non-feeding) mobile
Tunicates have a well-developed heart and circulatory system. The heart is a double U-shaped tube situated just below the gut. The blood vessels are simple connective tissue tubes, and their blood has several types of corpuscle. The blood may appear pale green, but this is not due to any respiratory pigments, and oxygen is transported dissolved in the plasma. Exact details of the circulatory system are unclear, but the gut, pharynx, gills, gonads, and nervous system seem to be arranged in series rather than in parallel, as happens in most other animals. Every few minutes, the heart stops beating and then restarts, pumping fluid in the reverse direction.[12]
Tunicate
Tunicates lack the kidney-like metanephridial organs typical of deuterostomes. Most have no excretory structures, but rely on the diffusion of ammonia across their tissues to rid themselves of nitrogenous waste, though some have a simple excretory system. The typical renal organ is a mass of large clear-walled vesicles that occupy the rectal loop, and the structure has no duct. Each vesicle is a remnant of a part of the primitive coelom, and its cells extract nitrogenous waste matter from circulating blood. They accumulate the wastes inside the vesicles as urate crystals, and do not have any obvious means of disposing of the material during their lifetimes.[42]
Adult tunicates have a hollow cerebral ganglion, equivalent to a brain, and a hollow structure known as a neural gland. Both originate from the embryonic neural tube and are located between the two siphons. Nerves arise from the two ends of the ganglion; those from the anterior end innervate the buccal siphon and those from the posterior end supply the rest of the body, the atrial siphon, organs, gut and the musculature of the body wall. There are no sense organs but there are sensory cells on the siphons, the buccal tentacles and in the atrium.[12]
Tunicates are unusual among animals in that they produce a large fraction of their tunic and some other structures in the form of cellulose. The production in animals of cellulose is so unusual that at first some researchers denied its presence outside of plants, but the tunicates were later found to possess a functional cellulose synthesizing enzyme, encoded by a gene horizontally transferred from a bacterium.[46] When, in 1845, Carl Schmidt first announced the presence in the test of some ascidians of a substance very similar to cellulose, he called it "tunicine", but it is now recognized as cellulose rather than any alternative substance.[47][48][49]
-
Oikopleura cophocerca in its "house". Arrows indicate water movement and (x) the lateral reticulated parts of the house.
-
Blue sea squirts from the genus Rhopalaea.
-
Fluorescent-colored sea squirts, Rhopalaea crassa.
Feeding

Nearly all adult tunicates are
Some ascidians that live on soft sediments are
Life cycle

Ascidians are almost all
When sufficiently developed, the larva of the sessile species finds a suitable rock and cements itself in place. The larval form is not capable of feeding, though it may have a rudimentary digestive system,[51] and is only a dispersal mechanism. Many physical changes occur to the tunicate's body during metamorphosis, one of the most significant being the reduction of the cerebral ganglion, which controls movement and is the equivalent of the vertebrate brain. From this comes the common saying that the sea squirt "eats its own brain".[52] However, the adult does possess a cerebral ganglion adapted to lack of self-locomotion.[53] In the Thaliacea, the larval stage is rudimentary or suppressed, and the adults are pelagic (swimming or drifting in the open sea).[42] Colonial forms also increase the size of the colony by budding off new individuals to share the same tunic.[54]
Pyrosome colonies grow by budding off new zooids near the posterior end of the colony. Sexual reproduction starts within a zooid with an internally fertilized egg. This develops directly into an oozooid without any intervening larval form. This buds precociously to form four blastozooids which become detached in a single unit when the oozoid disintegrates. The atrial siphon of the oozoid becomes the exhalent siphon for the new, four-zooid colony.[12]
Doliolids have a very complex life cycle that includes various zooids with different functions. The sexually reproducing members of the colony are known as gonozooids. Each one is a hermaphrodite with the eggs being fertilised by sperm from another individual. The gonozooid is viviparous, and at first, the developing embryo feeds on its yolk sac before being released into the sea as a free-swimming, tadpole-like larva. This undergoes metamorphosis in the water column into an oozooid. This is known as a "nurse" as it develops a tail of zooids produced by budding asexually. Some of these are known as trophozooids, have a nutritional function, and are arranged in lateral rows. Others are phorozooids, have a transport function, and are arranged in a single central row. Other zooids link to the phorozooids, which then detach themselves from the nurse. These zooids develop into gonozooids, and when these are mature, they separate from the phorozooids to live independently and start the cycle over again. Meanwhile, the phorozooids have served their purpose and disintegrate. The asexual phase in the lifecycle allows the doliolid to multiply very rapidly when conditions are favourable.[12]
Salps also have a complex lifecycle with an
Larvaceans only reproduce
During embryonic development, tunicates exhibit
Promotion of out-crossing
Ciona intestinalis (class Ascidiacea) is a hermaphrodite that releases sperm and eggs into the surrounding seawater almost simultaneously. It is self-sterile, and thus has been used for studies on the mechanism of self-incompatibility.[56] Self/non-self-recognition molecules play a key role in the process of interaction between sperm and the vitelline coat of the egg. It appears that self/non-self recognition in ascidians such as C. intestinalis is mechanistically similar to self-incompatibility systems in flowering plants.[56] Self-incompatibility promotes out-crossing, and thus provides the adaptive advantage at each generation of the masking of deleterious recessive mutations (i.e. genetic complementation)[57] and the avoidance of inbreeding depression.
Botryllus schlosseri (class Ascidiacea) is a colonial tunicate, a member of the only group of chordates that are able to reproduce both sexually and asexually. B. schlosseri is a sequential (protogynous) hermaphrodite, and in a colony, eggs are ovulated about two days before the peak of sperm emission.[58] Thus self-fertilization is avoided, and cross-fertilization is favored. Although avoided, self-fertilization is still possible in B. schlosseri. Self-fertilized eggs develop with a substantially higher frequency of anomalies during cleavage than cross-fertilized eggs (23% vs. 1.6%).[58] Also a significantly lower percentage of larvae derived from self-fertilized eggs metamorphose, and the growth of the colonies derived from their metamorphosis is significantly lower. These findings suggest that self-fertilization gives rise to inbreeding depression associated with developmental deficits that are likely caused by expression of deleterious recessive mutations.[57]
A model tunicate
Invasive species
Over the past few decades, tunicates (notably of the genera Didemnum and Styela) have been invading coastal waters in many countries. The carpet tunicate (Didemnum vexillum) has taken over a 6.5 sq mi (17 km2) area of the seabed on the Georges Bank off the northeast coast of North America, covering stones, molluscs, and other stationary objects in a dense mat.[60] D. vexillum, Styela clava and Ciona savignyi have appeared and are thriving in Puget Sound and Hood Canal in the Pacific Northwest.[61]
Invasive tunicates usually arrive as
Use by humans
Medical uses
Tunicates contain a host of potentially useful chemical compounds, including:
- Plitidepsin, a didemnin effective against various types of cancer; as of late January 2021 undergoing Phase III trials as a treatment for COVID-19[63]
- Trabectedin, an FDA approved anticancer drug.
Tunicates are able to correct their own cellular abnormalities over a series of generations, and a similar regenerative process may be possible for humans. The mechanisms underlying the phenomenon may lead to insights about the potential of cells and tissues to be reprogrammed and to regenerate compromised human organs.[64][65][66]
As food

Various
Other uses
The use of tunicates as a source of biofuel is being researched. The cellulose body wall can be broken down and converted into ethanol, and other parts of the animal are protein-rich and can be converted into fish feed. Culturing tunicates on a large scale may be possible and the economics of doing so are attractive. As tunicates have few predators, their removal from the sea may not have profound ecological impacts. Being sea-based, their production does not compete with food production as does the cultivation of land-based crops for biofuel projects.[68]
Some tunicates are used as model organisms. Ciona intestinalis and Ciona savignyi have been used for developmental studies. Both species' mitochondrial[69][70] and nuclear[71][72] genomes have been sequenced. The nuclear genome of the appendicularian Oikopleura dioica appears to be one of the smallest among metazoans[73] and this species has been used to study gene regulation and the evolution and development of chordates.[74]
See also
- Vetulicolia – stem-group chordates which are possibly the sister group of modern tunicates
- Donald I. Williamson – claimed hybridization
References
- S2CID 135091168.
- S2CID 128415270.
- S2CID 244957825.
- ^ a b Sanamyan, Karen (2013). "Tunicata". WoRMS. World Register of Marine Species. Retrieved 4 April 2013.
- S2CID 83266247.
- PMID 29703197.
- ^ S2CID 86421513.
- S2CID 3553695.
- ISBN 9780585252728– via Google Books.
- S2CID 20806327. Retrieved 13 May 2023.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - S2CID 213181394.
- ^ ISBN 978-81-315-0104-7.
- ^ Foster, M. (ed.); Sedgwick, Adam (ed.); The Works of Francis Maitland Balfour. Vol. III. Memorial edition. Pub: Macmillan and co. 1885. May be downloaded from [1]
- ^ Tunicata World Register of Marine Species. Retrieved 2011-11-12.
- ^ Tunicata Lamarck, 1816 Integrated Taxonomic Information System. Retrieved 2017-03-30.
- ^ "Sea squirts and sea tulips". Australian Museum. Retrieved 25 September 2013.
- ^ "Sea squirt". Dictionary.com. Retrieved 25 September 2013.
- ^ "Sea pork, Aplidium stellatum". Smithsonian at Fort Pierce. Retrieved 25 September 2013.
- ^ S2CID 4382758.
- S2CID 205771088.
- PMID 19922605.
- ^ Jefferies, R. P. S. (1991) in Biological Asymmetry and Handedness (eds Bock, G. R.; Marsh, J.) pp. 94–127 (Wiley, Chichester).
- doi:10.1139/z05-010.
- PMID 19656395.
- ^ PMID 29653534.
- S2CID 55491438.
- PMID 12835415.
- ^ Palmer, T. J.; Wilson, M. A. (1988). "Parasitism of Ordovician bryozoans and the origin of pseudoborings" (PDF). Palaeontology. 31: 939–949. Archived from the original (PDF) on 27 September 2013. Retrieved 7 April 2013.
- ^ Nagalwade, Vidya (7 July 2023). "A 500 million-year-old fossil reveals the amazing secrets of tunicate origins". Tech Explorist.
- ^ Vickers-Rich P. (2007). "Chapter 4. The Nama Fauna of Southern Africa". In: Fedonkin, M. A.; Gehling, J. G.; Grey, K.; Narbonne, G. M.; Vickers-Rich, P. "The Rise of Animals: Evolution and Diversification of the Kingdom Animalia", Johns Hopkins University Press. pp. 69–87
- ^ a b Fedonkin, M. A.; Vickers-Rich, P.; Swalla, B.; Trusler, P.; Hall, M. (2008). "A Neoproterozoic chordate with possible affinity to the ascidians: New fossil evidence from the Vendian of the White Sea, Russia and its evolutionary and ecological implications". HPF-07 Rise and fall of the Ediacaran (Vendian) biota. International Geological Congress - Oslo 2008.
- ^ "Introduction to the Urochordata". University of California Museum of Paleontology. Archived from the original on 21 April 2009. Retrieved 7 April 2013.
- ^ [https://www.cambridge.org/core/services/aop-cambridge-core/content/view/0FE5DCCCDFDD464B92DCA4AF68F36F2B/S0022336019001094a.pdf/rare_case_of_an_evolutionary_late_and_ephemeral_biomineralization_tunicates_with_composite_calcareous_skeletons.pdf A rare case of an evolutionary late and ephemeral biomineralization: tunicates with composite calcareous skeletons]
- S2CID 135456629– via CrossRef.
- .
- PMID 35995990.
- PMID 19100250.
- S2CID 241318821.
- PMID 26085517.
- PMID 33881514.
- PMID 25925633.
- ^ ISBN 978-0-03-030504-7– via archive.org.
- ISBN 978-1-000-36913-7.
- S2CID 116921.
- .
- PMID 14722352.
- PMID 22446551.
- PMID 28003446.
- PMID 16214891.
- ^ Response of Marine Ecosystems to Global Change: Ecological Impact of Appendicularians
- ^ a b Cavanihac, Jean-Marie (2000). "Tunicates extraordinaire". Microscope UK. Retrieved 7 December 2011.
- ISBN 978-0316-18065-8.
- doi:10.1139/z04-177.
- ^ Parmentier, Jan (1998). "Botryllus: A colonial ascidian". Microscope UK. Retrieved 7 April 2013.
- S2CID 5549210.
- ^ PMID 24878524.
- ^ )
- ^ S2CID 205772576.
- PMID 17126826.
- ^ "Have You Seen This Tunicate?". NOAA Fisheries Service. 19 November 2004. Archived from the original on 9 January 2009. Retrieved 7 December 2011.
- ^ a b Dornfeld, Ann (1 May 2008). "Invasive Tunicates of Washington State". NPR. Archived from the original on 14 July 2014. Retrieved 6 April 2013.
- ^ "Marine Nuisance Species". Woods Hole Science Center. Retrieved 7 December 2011.
- ^ Johnson, Mark. "International team of scientists identifies new treatment for COVID-19 that appears to be far more effective than drugs in use now". Journal Sentinel.
- OCLC 233972733.
- ^ "Sea Squirt, Heal Thyself: Scientists Make Major Breakthrough in Regenerative Medicine". Sciencedaily.com. 24 April 2007. Retrieved 7 December 2011.
- S2CID 37526690.
- ^ "Sea squirt". Korea-US Aquaculture. Archived from the original on 2 March 2013. Retrieved 6 April 2013.
- ^ "Biofuel made from marine filter feeders? Tunicates usable as source of biofuels". Cleantechnica. 26 March 2013. Retrieved 6 April 2013.
- PMID 17640763.
- S2CID 19474615.
- S2CID 15987281.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 17374142.
- PMID 11752568.
- PMID 17449201.








