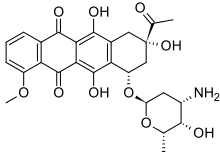
Daunorubicin
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Cerubidine, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682289 |
| Pregnancy category |
|
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Liver |
| Elimination half-life | 26.7 hours (metabolite) |
| Excretion | Bile duct and urinary |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Daunorubicin, also known as daunomycin, is a
Common side effects include hair loss, vomiting,
Daunorubicin was approved for medical use in the United States in 1979.[2] It is on the World Health Organization's List of Essential Medicines.[4] It was originally isolated from bacteria of the Streptomyces type.[5]
Medical uses
It slows or stops the growth of cancer cells in the body. Treatment is usually performed together with other chemotherapy drugs (such as cytarabine), and its administration depends on the type of tumor and the degree of response.[citation needed]
In addition to its major use in treating acute myeloid leukemia, daunorubicin is also used to treat neuroblastoma. Daunorubicin has been used with other chemotherapy agents to treat the blastic phase of chronic myelogenous leukemia.[citation needed]
Daunorubicin is also used as the starting material for semi-synthetic manufacturing of doxorubicin, epirubicin and idarubicin.[citation needed]
Mechanism of action
Similar to
History
In the 1950s, an
However, by 1967, it was recognized that daunorubicin could produce fatal cardiac toxicity.[14]
In 2015–16, a team at Ohio State University "showed that, by carefully manipulating strands of viral DNA, an origami structure with complex folds can be created in just 10 minutes. Incredibly, these structures are only 100 nanometers across – that’s 1,000 times smaller than the width of a human hair. Small volumes of daunorubicin can be wrapped up in these minuscule pods, which can then be released into a leukemia cell-filled environment."[15][unreliable medical source?]
Route of administration
Daunorubicin should only be administered in a rapid
References
- ^ "Daunorubicin (Cerubidine) Use During Pregnancy". Drugs.com. 19 September 2019. Retrieved 15 August 2020.
- ^ a b c d e f g h i "Daunorubicin hydrochloride". The American Society of Health-System Pharmacists. Archived from the original on 8 January 2017. Retrieved 8 December 2016.
- ISBN 9780857111562.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ISBN 9781118075630. Archivedfrom the original on 21 December 2016.
- PMID 8183243.
- from the original on 5 February 2009.
- PMID 6938965.
- PMID 23715267.
- PMID 25961671.
- PMID 1462166.
- PMID 5332105.
- ^ Camerino B, Palamidessi G (1960). "Derivati della parazina II. Sulfonamdopir" [Derivatives of parazine II. Sulfonamdopir]. Gazzetta Chimica Italiana [Italian Chemical Journal] (in Italian). 90: 1802–1815.
- S2CID 19272219.
- ^ "Researchers kill drug-resistant leukemia cells using DNA Trojan horse attack". IFL Science. 25 February 2016.
- PMID 1574039.
