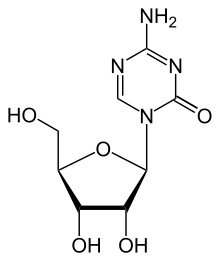
Azacitidine
 | |
| Clinical data | |
|---|---|
| Trade names | Vidaza, Azadine, Onureg |
| Other names | 5-Azacytidine, Azacytidine, Ladakamycin, 4-Amino-1-β-D-ribofuranosyl-s-triazin-2(1H)-one, U-18496, CC-486 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607068 |
| License data |
|
| Pregnancy category |
|
Subcutaneous, intravenous, by mouth | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 4 hr.[8] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Azacitidine, sold under the brand name Vidaza among others, is a
The most common adverse reactions in children with juvenile myelomonocytic leukemia include pyrexia, rash, upper respiratory tract infection, and anemia.[9]
Medical uses
Azacitidine is
Azacitidine is also indicated for the treatment of myeloid leukemia[5][6][14] and juvenile myelomonocytic leukemia.[4][9] The combination of azacitidine and venetoclax is also approved for AML.[15]
Mechanism of action
Azacitidine is a chemical analogue of the nucleoside
Inhibition of methylation
After azanucleosides such as azacitidine have been metabolized to 5-aza-2′-deoxycytidine-triphosphate (aka, decitabine-triphosphate), they can be incorporated into DNA and azacytosine can be substituted for cytosine. Azacytosine-guanine dinucleotides are recognized as substrate by the DNA methyltransferases, which catalyze the methylation reaction by a nucleophilic attack. This results in a covalent bond between the carbon-6 atom of the cytosine ring and the enzyme. The bond is normally resolved by beta-elimination through the carbon-5 atom, but this latter reaction does not occur with azacytosine because its carbon-5 is substituted by nitrogen, leaving the enzyme covalently bound to DNA and blocking its DNA methyltransferase function. In addition, the covalent protein adduction also compromises the functionality of DNA and triggers DNA damage signaling, resulting in the degradation of trapped DNA methyltransferases. As a consequence, methylation marks become lost during DNA replication.[19][20]
Toxicity
Azacitidine causes anemia (low red blood cell counts), neutropenia (low white blood cell counts), and thrombocytopenia (low platelet counts), and patients should have frequent monitoring of their complete blood counts, at least prior to each dosing cycle. The dose may have to be adjusted based on nadir counts and hematologic response.[4]
It can also be hepatotoxic in patients with severe liver impairment, and patients with extensive liver tumors due to metastatic disease have developed progressive hepatic coma and death during azacitidine treatment, especially when their albumin levels are less than 30 g/L. It is contraindicated in patients with advanced malignant hepatic tumors.[4]
Kidney toxicity, ranging from elevated serum creatinine to kidney failure and death, have been reported in patients treated with intravenous azacitidine in combination with other chemotherapeutic agents for conditions other than myelodysplastic syndrome. Renal tubular acidosis developed in five patients with chronic myelogenous leukemia (an unapproved use) treated with azacitidine and etoposide, and patients with renal impairment may be at increased risk for renal toxicity. Azacitidine and its metabolites are primarily excreted by the kidneys, so patients with chronic kidney disease should be closely monitored for other side effects, since their levels of azacitidine may progressively increase.[4]
Based on animal studies and its mechanism of action, azacitidine can cause severe fetal damage. Sexually active women of reproductive potential should use contraception during while receiving azacitidine and for one week after the last dose, and sexually active men with female partners of reproductive potential should use contraception during treatment and for three months following the last dose.[4]
A study undertaken to evaluate the immediate and long-term effects of a single-day exposure to Azacytidine (5-AzaC) on
Azacitidine can also cause nausea, vomiting, fevers, diarrhea, redness at its injection sites, constipation, bruising, petechiae, rigors, weakness, abnormally low potassium levels in the bloodstream, and many other side effects, some of which can be severe or even fatal.[4]
History
The efficacy of azacitidine to treat juvenile myelomonocytic leukemia was evaluated in AZA-JMML-001 (NCT02447666), an international, multicenter, open-label study to evaluate the pharmacokinetics, pharmacodynamics, safety, and activity of azacitidine prior to hematopoietic stem cell transplantation in 18 pediatric patients with juvenile myelomonocytic leukemia.[9]
Research
Azacitidine can be used
Azacitidine induces tumor regression on isocitrate dehydrogenase-1 mutant glioma xenografts in mice.[23]
In research, 5-azacitidine is commonly used for promoting cardiomyocyte differentiation of adult stem cells. However, it has been suggested that this drug has a compromised efficacy as a cardiac differentiation factor because it promotes the transdifferentiation of cardiac cells to skeletal myocytes.[24]
Azacitidine also has
References
- ^ "Azacitidine (Vidaza) Use During Pregnancy". Drugs.com. 5 May 2020. Retrieved 12 August 2020.
- ^ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 30 March 2024.
- ^ "Health product highlights 2021: Annexes of products approved in 2021". Health Canada. 3 August 2022. Retrieved 25 March 2024.
- ^ a b c d e f g h i j "Vidaza- azacitidine injection, powder, lyophilized, for solution". DailyMed. 2 March 2020. Retrieved 27 September 2020.
- ^ a b c "Onureg- azacitidine tablet, film coated". DailyMed. 20 May 2021. Retrieved 24 May 2022.
- ^ a b c "Onureg EPAR". European Medicines Agency. 20 April 2021. Retrieved 6 September 2021.
- ^ "Onureg Product information". Union Register of medicinal products. Retrieved 3 March 2023.
- ISBN 978-0-8036-1912-8.
- ^ a b c d "FDA approves azacitidine". U.S. Food and Drug Administration. 20 May 2022. Retrieved 24 May 2022.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- PMID 4142650.
- ^ "Drug Approval Package: Vidaza (Azacitidine) NDA #050794". U.S. Food and Drug Administration (FDA). 14 July 2004. Retrieved 26 September 2020.
- ^ "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations". fda.gov. U.S. Food and Drug Administration (FDA). Retrieved 20 May 2016.
- S2CID 11375964.
- PMID 36230820.
- S2CID 221121486.
- ISBN 978-3-642-01222-8.
- PMID 19726509.
- PMID 22214262.
- S2CID 14125490.
- PMID 24382388.
- PMID 27594097.
- ISBN 0-85199-910-7.
- PMID 24077805.
- PMID 25090621.
- PMID 29170916.
- PMID 29698664.
External links
- Clinical trial number NCT02447666 for "Study With Azacitidine in Pediatric Subjects With Newly Diagnosed Advanced Myelodysplastic Syndrome (MDS) and Juvenile Myelomonocytic Leukemia (JMML)" at ClinicalTrials.gov
