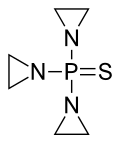
Thiotepa
| |||
| Clinical data | |||
|---|---|---|---|
| Trade names | Tepadina | ||
| Other names | N,N',N''(-triethylenethiophosphoramide, INN, Thiotepa, Tepadina, Tepylute | ||
| AHFS/Drugs.com | Monograph | ||
| MedlinePlus | a682821 | ||
| License data | |||
| Pregnancy category |
| ||
| Routes of administration | Intravenous, intracavitary, intravesical | ||
| ATC code | |||
| Legal status | |||
| Legal status | |||
| Pharmacokinetic data | |||
| Metabolism | Liver (CYP2B6, CYP3A) | ||
| Metabolites | Phase 1: Tetraethylenepentamine (tepa) Phase 2: Thiotepa-mercapturate | ||
| Elimination half-life | 1.5–4.1 hours | ||
| Excretion | Kidney 6 hours for thiotepa 8 hours for TEPA | ||
| Identifiers | |||
| |||
JSmol) | |||
| Melting point | 51.5 °C (124.7 °F) | ||
| Solubility in water | 0.19 g/mL (water, 25 °C) Freely soluble in alcohol, diethyl ether and chloroform mg/mL (20 °C) | ||
SMILES
| |||
| |||
| | |||
Thiotepa (N,N',N''(-triethylenethiophosphoramide,
Thiotepa is an
History
Thiotepa and its synthesis were patented in
In January 2007, the European Medicines Agency (EMA) designated thiotepa as an orphan drug. In April 2007, the United States FDA designated thiotepa as a conditioning treatment for use prior to hematopoietic stem cell transplantation.[12]
In June 2024, the FDA approved a ready-to-dilute liquid formulation of thiotepa to treat breast and ovarian cancer.[13]
Structure
Thiotepa consists of three
Reactivity
Thiotepa is a reactive compound that, under
In alkaline media, thiotepa undergoes degradation, though no detectable byproducts were identified. Like other aziridine-containing compounds,
Synthesis
Two separate syntheses of thiotepa have been described in literature. The most prevalent method involves the addition of an excess of aziridine to thiophosphoryl chloride in the presence of a base such as triethylamine (TEA) (or another molar equivalent of aziridine) and a suitable solvent (e.g., ether or benzene). The first molecule of aziridine reacts with thiophosphoryl chloride to produce dichloridophosphorothionate, which is sufficiently reactive due to the poor overlap of the nitrogen lone pair with the P=S bond, allowing it to react with another two molecules of aziridine [14]

Thiotepa has also been synthesized from phosphorus trichloride and six molar equivalents of aziridine. The trivalent triamide formed reacts with octasulfur (S₈) in benzene.[14]
Medical uses
Thiotepa is used in combination with other
Thiotepa is used in
In
Administration
Thiotepa is mainly administered
Clinical outcomes
In clinical trials the outcome of different types of treatment is compared to identify if a compound or regimen is favourable for the patient. The choice of treatment in the conditioning therapy can have a profound impact on progression-free survival (PNS), overall survival (OS), relapse incidence (RI) and non-relapse mortality (NRM). The studies mentioned summarize key findings comparing various conditioning regimens.
Studies on conditioning regimens for
Metabolism

The metabolism of thiotepa primarily takes place in the liver, following both phase 1 and phase 2 metabolic pathways. Phase 1 involves reactions which change chemical moieties such as oxidation, reduction, and hydrolysis, while phase 2 includes the addition of endogenous groups to foreign compounds.[21]
Phase 1 metabolism of thiotepa is predominantly mediated by the cytochrome P450 enzyme system, major CYP2B6 and minor CYP3A4. In this phase an oxidation and desulfuration reactions convert thiotepa into its more active metabolite tepa.[10][22] Tepa itself exhibits a longer plasma half-life (3 to 24 hours) than thiotepa (1 to 3 hours) and contributes to the overall pharmacological activity of the drug.[23]
In phase 2 thiotepa is detoxified via the conjugation with
Enzymes responsible for metabolising compounds can show varying efficiency in different individuals or populations, this is called polymorphism. In a study regarding thiotepa metabolism by Ekhart et al., it was found that glutathione S-transferase shows polymorphism. This variation resulted in some patients in slower glutathione conjugation and consequently, to a 45% increase in combined exposure to thiotepa and tepa.[26]
The volume of distribution has been reported to range from 40,8 L/m2 to 75,0 L/m2.[7] This high value is due to the highly lipophilic character of thiotepa and can therefore easily cross cell membranes and distribute into fatty tissues. In addition, thiotepa can easily cross the blood brain barrier and can rapidly penetrate the central nervous system.[27][28] In plasma, 70 to 90% of the compound remains unbound to proteins, while the remaining 10–30% is primarily bound to gamma globulin, with minimal binding to albumin.[15] Gamma globulin primarily functions as antibodies for the immune system,[29] while albumin serves as a transport protein.[30]
All metabolites are excreted in the urine, which is nearly complete in 6 to 8 hours, with tepa and thiotepa-mercapturate each accounting for approximately 11.1% of the excretion. In contrast, the excretion of monochloride tepa and thiotepa is significantly lower, at only 0.5% each.[10][15] The total clearance of thiotepa ranged from 11,4 to 23,2 L/h/m2.[15] The total excretion of thiotepa and its identified metabolites accounts for 54 to 100% of the total alkylating activity, suggesting the existence of other alkylating metabolites. During the conversion of glutathione conjugates into N-acetylcysteine conjugates, intermediates such as glutathione, cysteinyl glycine, and cysteine conjugates are formed. These metabolites are not detected in urine and, if formed, are likely excreted in bile or rapidly converted into thiotepa-mercapturate.[10] Additionally, due to its high lipophilicity, thiotepa is excreted in minor amounts by the skin via sweat.[31][32]
Molecular mechanism of action

Thiotepa, as well as its more reactive metabolite, tepa, work as an alkylating agent via its aziridine ring. Due to the basic nature of aziridine and the physiological pH, aziridine is protonated to form the
Toxicity
Thiotepa is associated with a range of side effects. The severity and type of side effects may vary based on the dosage, duration of treatment, and individual patient factors.
Myelosuppression
Proliferating cells, such as tumour cells, are more sensitive to alkylating agents, rendering these drugs useful for chemotherapy.
Monoalkylation of DNA leads to mispairing of bases and, if not repaired, can reside in the DNA sequence. Mutated DNA that does not undergo cell death can find its way into daughter cells and potentially cause genetic disorders such as cancer.[21] As a result of cell mutation in the bone marrow, chemotherapies with alkylating agents are known to cause acute myeloid leukaemia (AML) and myelodysplastic syndrome (MDS).[46]
Additional toxic effects
Apart from its mutagenic nature, thiotepa can exert skin toxicity, such as redness and
Women and men of childbearing potential have to use effective contraception during treatment. A pregnancy test should be performed before treatment is started. Men should not father a child during and a year after cessation of treatment. There is no data on the administration of thiotepa during pregnancy, But as in-vivo animal experiments showed teratogenic effects the use of thiotepa during pregnancy is contraindicated. It is not known whether thiotepa is excreted in human breast milk, but due to its high lipophilicity, this cannot be ruled out. Due to its pharmacological properties and potential for toxicity in newborns/infants breast feeding is contraindicated during treatment with thiotepa.[50]
Drug interactions
Thiotepa can have various interactions with other medications or therapies that can impact patient safety and treatment efficacy. Aprepitant, a drug that prevents nausea and vomiting that may occur during chemotherapy, inhibits CYP-enzymes, which decrease the metabolism of thiotepa to tepa. Its importance is relatively minor because inhibition itself is small and due to the variability of thiotepa clearance in different individuals.[51] The anti-seizure medication phenytoin induces the CYP3A4 enzyme. This leads to an increased rate of tepa formation from thiotepa, which highly influences its clearance and concentration. Higher local concentrations of the more reactive tepa can potentially induce hepatotoxicity. Additionally, cytotoxic drugs such as thiotepa can reduce the absorption of phenytoin leading to the increased risk of seizures. It is advised to avoid the use of both drugs simultaneously or decrease thiotepa doses.[50][52]
Myelosuppressive/myelotoxic agents such as melphalan, busulfan, treosulfan, and cyclophosphamide, as well as the concurrent use of thiotepa, may increase the risk of hematologic adverse reactions and pulmonary toxicity, as they share similar toxicity profiles. Additionally, the use of live attenuated vaccines (including yellow fever) poses a risk of systemic and potentially fatal infection, with the risk further heightened in patients who are already immunosuppressed due to their underlying disease. In general, thiotepa is a potent inhibitor of CYP2B6, which can lead to increased plasma levels of drugs that are substrates of this enzyme. In addition to this, it may reduce the levels of potentially active metabolites, such as 4-hydroxycyclophosphamide, from cyclophosphamide. Likewise, co-administration with inhibitors of thiotepa’s metabolising enzymes can lead to increased thiotepa plasma concentrations. Finally, prolonged apnea has been reported by the administration of thiotepa and is thought to be a result of the inhibition of pseudocholinesterase by thiotepa. For this reason, inhibitors such as succinylcholine and pancuronium should be prevented during thiotepa administration to prevent respiratory failure.[15]
References
- FDA. Retrieved 22 October 2023.
- ^ "Tepadina (Link Medical Products Pty Ltd T/A Link Pharmaceuticals)". Therapeutic Goods Administration (TGA). 28 September 2022. Archived from the original on 18 March 2023. Retrieved 29 April 2023.
- ^ "Cancer therapies". Health Canada. 8 May 2018. Retrieved 13 April 2024.
- ^ "Thiotepa 100 mg powder for concentrate for solution for infusion". (emc). 27 October 2022. Retrieved 14 August 2024.
- ^ a b "Tepadina- thiotepa injection, powder, for solution". DailyMed. Archived from the original on 12 August 2021. Retrieved 11 August 2021.
- ^ "Highlights of prescribing information" (PDF). www.accessdata.fda.gov.
- ^ a b c d "Tepadina EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 6 March 2021. Retrieved 30 April 2021.
- ^ "International Non-Proprietary Names for Pharmaceutical Preparations. Recommended International Non-Proprietary Names (Rec. I.N.N.): List 4" (PDF). World Health Organization. March 1962. p. 111. Archived from the original (PDF) on 18 May 2016. Retrieved 27 November 2016.
- ^ U.S. Food and Drug Administration(FDA). 25 November 2011. Retrieved 14 March 2025.
- ^ PMID 10913381.
- .
- ^ "EMA Grants Adienne Marketing Rights for Tepadina". Drug Discovery & Development. 19 March 2010. Retrieved 25 November 2011.
- ^ "Shorla Oncology Announces FDA Approval for Tepylute, A Novel Formulation to Treat Breast and Ovarian Cancer" (Press release). Shorla Oncology. 28 June 2024 – via Business Wire.
- ^ ISBN 978-0-08-098212-0.
- ^ a b c d e f g "Tepadina EPAR". European Medicines Agency (EMA). 6 May 2010. Retrieved 14 March 2025.
- PMID 33788375.
- ^ a b "Thiotepa for Injection USP" (PDF). SteriMax Inc. 5 April 2022.
- PMID 33956047.
- PMID 26569093.
- PMID 38755458.
- ^ ISBN 978-0-8493-7302-2.
- ^ PMID 12107550.
- ISBN 978-0-12-369400-3, retrieved 15 March 2025
- ^ PMID 26900903.
- PMID 10493531.
- PMID 19076156.
- PMID 36329150.
- ^ "Thiotepa Monograph" (PDF). BC Cancer Drug Manual. BC Cancer Provincial Pharmacy. 1 December 2024.
- PMC 7150067.
- .
- ^ PMID 39912674.
- ^ PMID 30185131.
- PMID 1799576.
- PMID 1714342.
- PMID 3121169.
- PMID 1374653.
- PMID 3324117.
- ISSN 1354-3776.
- PMID 1904247.
- PMID 6415297.
- PMID 1705489.
- PMID 6812036.
- PMID 3111687.
- PMID 2492594.
- PMID 8104539.
- ISBN 978-1-260-45230-3.
- PMID 15349862.
- ^ "Thiotepa for Injection, USP" (PDF). Sagent Pharmaceuticals, Inc. 20 August 2021.
- PMID 13871143.
- ^ a b "TEPYLUTE (thiotepa) injection, for intravenous use" (PDF). Shorla Oncology Inc. U.S. Food and Drug Administration.
- PMID 15838656.
- ISSN 0022-2623.