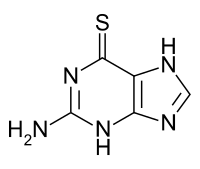
Tioguanine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Lanvis, Tabloid, others |
| AHFS/Drugs.com | International Drug Names |
| MedlinePlus | a682099 |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 30% (range 14% to 46%) |
| Metabolism | Intracellular |
| Elimination half-life | 80 minutes (range 25–240 minutes) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Tioguanine, also known as thioguanine or 6-thioguanine (6-TG) or tabloid is a medication used to treat
Common side effects include
Tioguanine was developed between 1949 and 1951.[5][6] It is on the World Health Organization's List of Essential Medicines.[7]
Medical uses
- Acute leukemias in both adults and children
- Chronic myelogenous leukemia
- Inflammatory bowel disease, especially ulcerative colitis
- Psoriasis[8]
- Colorectal cancer in mice resistant to immunotherapy[9]
Side effects
- Leukopenia and neutropenia
- Thrombocytopenia
- Anemia
- Anorexia
- Nausea and vomiting
- Hepatotoxicity: this manifests as:
Hepatic veno-occlusive disease
The major concern that has inhibited the use of thioguanine has been veno-occlusive disease (VOD) and its histological precursor nodular regenerative hyperplasia (NRH). The incidence of NRH with thioguanine was reported as between 33 and 76%.[10] The risk of ensuing VOD is serious and frequently irreversible so this side effect has been a major concern. However, recent evidence using an animal model for thioguanine-induced NRH/VOD has shown that, contrary to previous assumptions, NRH/VOD is dose dependent and the mechanism for this has been demonstrated.[11] This has been confirmed in human trials, where thioguanine has proven to be safe but efficacious for coeliac disease when used at doses below those commonly prescribed.[12] This has led to a revival of interest in thioguanine because of its higher efficacy and faster action compared to other thiopurines and immunosuppressants such as mycophenylate.[13]
Contraindications
- Pregnancy
- Lactation: The safety warning against breastfeeding may have been a conservative assessment, but research evidence suggests that thiopurines do not enter breastmilk.[14]
Interactions
Cancers that do not respond to treatment with mercaptopurine do not respond to thioguanine. On the other hand, some cases of IBD that are resistant to mercaptopurine (or its pro-drug azathioprine) may be responsive to thioguanine.
Pharmacogenetics
The enzyme
Metabolism and pharmacokinetics
A single oral dose of thioguanine has incomplete metabolism, absorption and high interindividual variability. The bioavailability of thioguanine has an average of 30% (range 14-46%). The maximum concentration in plasma after a single oral dose is attained after 8 hours.
Thioguanine, like other thiopurines, is cytotoxic to white cells; as a result it is immunosuppressive at lower doses and anti-leukemic/anti-neoplastic at higher doses. Thioguanine is incorporated into human bone marrow cells, but like other thiopurines, it is not known to cross the blood-brain barrier. Thioguanine cannot be demonstrated in cerebrospinal fluid, similar to the closely related compound 6-mercaptopurine which also cannot penetrate to the brain.
The plasma half-life of thioguanine is short, due to the rapid uptake into liver and blood cells and conversion to 6-TGN. The median plasma half-life of 80-minutes with a range of 25–240 minutes. Thioguanine is excreted primarily through the kidneys in urine, but mainly as a metabolite, 2-amino-6-methylthiopurine. However, the intra-cellular thio-nucleotide metabolites of thioguanine (6-TGN) have longer half-lives and can therefore be measured after thioguanine is eliminated from the plasma.
Thioguanine is catabolized (broken down) via two pathways.[22] One route is through the deamination by the enzyme guanine deaminase to 6-thioxanthine, which has minimal anti-neoplastic activity, then by oxidation by xanthine oxidase of the thioxanthine to thiouric acid. This metabolic pathway is not dependent on the efficacy of xanthine oxidase, so that the inhibitor of xanthine oxidase, the drug allopurinol, does not block the breakdown of thioguanine, in contrast to its inhibition of the breakdown of the related thiopurine 6-mercaptopurine. The second pathway is the methylation of thioguanine to 2-amino-6-methylthiopurine, which is minimally effective as an anti-neoplastic and significantly less toxic than thioguanine. This pathway also is independent of the enzyme activity of xanthine oxidase.
Mechanism of action
6-Thioguanine is a thio analogue of the naturally occurring purine base guanine. 6-thioguanine utilises the enzyme
TGMP is converted by phosphorylation to thioguanosine diphosphate (TGDP) and thioguanosine triphosphate (TGTP). Simultaneously deoxyribosyl analogs are formed, via the enzyme
Chemistry
It is a pale yellow, odorless, crystalline powder.
Names
Tioguanine (
).Thioguanine is administered by mouth (as a tablet – 'Lanvis').
References
- ^ "Product monograph brand safety updates". Health Canada. February 2024. Retrieved 24 March 2024.
- ^ ISBN 978-0-85711-156-2.
- ^ a b c "Tioguanine 40 mg Tablets – Summary of Product Characteristics (SPC) – (eMC)". www.medicines.org.uk. Archived from the original on 21 December 2016. Retrieved 21 December 2016.
- ISBN 978-1-60831-270-2. Archivedfrom the original on 2016-12-21.
- ISBN 978-0-8247-9549-8. Archivedfrom the original on 2016-12-21.
- ISBN 978-0-941901-21-5. Archivedfrom the original on 2016-12-21.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- PMID 11148479.
- PMID 36584674.
- PMID 12891528.
- S2CID 29585979.
- S2CID 24811114.
- ^ Van Asseldonk DP, Oancea I, Jharap B, et al. (March 2012). "Is thioguanine-associated sinusoidal obstruction syndrome avoidable? Lessons learned from 6-thioguanine treatment of inflammatory bowel disease and a mouse model" (PDF). Journal of the Brazilian Medical Association. 58 (Suppl.1): S8–13.
- PMID 16995866.
- ^ PMID 21270794.
- PMID 19952870.
- from the original on 2013-01-12.
- ISBN 978-3-8047-1763-3.
- PMID 17255139.
- ^ "TABLOID- thioguanine tablet". DailyMed. Retrieved 17 March 2015.
- PMID 20154640.
- ^ Oncea I, Duley J (2008). "Chapter 38. Pharmacogenetics of Thiopurines.". In Brunton LL, Lazo JS, Parker K (eds.). Goodman & Gilman's The Pharmacological Basis of Therapeutics (11th ed.). McGraw-Hill's Access Medicine (on-line).
- S2CID 34015182.
- S2CID 23686284.
Further reading
- Dean L (2012). "Thioguanine Therapy and TPMT Genotype". In Pratt VM, McLeod HL, Rubinstein WS, et al. (eds.). Medical Genetics Summaries. PMID 28520351. Bookshelf ID: NBK100663.
