Entinostat
| |
| Names | |
|---|---|
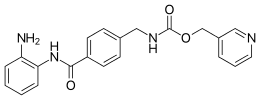
| Preferred IUPAC name
(Pyridin-3-yl)methyl ({4-[(2-aminophenyl)carbamoyl]phenyl}methyl)carbamate | |
| Other names
SNDX-275; MS-275
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.158.999 |
IUPHAR/BPS |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C21H20N4O3 | |
| Molar mass | 376.4085 g/mol |
| Pharmacology | |
| L01XH05 (WHO) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Entinostat, also known as SNDX-275 and MS-275, is a benzamide histone deacetylase inhibitor undergoing clinical trials for treatment of various cancers.[1][2][3][4]
Entinostat inhibits class I HDAC1 and HDAC3 with IC50 of 0.51 μM and 1.7 μM, respectively.[5]
Syndax pharmaceuticals currently holds the rights to entinostat and recently received $26.6 million in funds to advance treatments of resistant cancers using epigenetic tools.[6]
It has also been investigated as a potential male contraceptive drug.[7]
References
- .
- S2CID 219169967.
- S2CID 235378858.
- PMID 36969235.
- ^ US 2009/0263353, Maier T, Beckers T, Hummel RP, Feth M, Muller M, Bar T, Volz J, "Novel Sulphonylpyrroles as Inhibitors of Hdac S Novel Sulphonylpyrroles", issued 31 July 2012, assigned to 4SC AG
- ^ "Company Prepares for Pivotal Phase 3 Study of Entinostat, Most Advanced HDAC Inhibitor in Development for ER+ Metastatic Breast Cancer" (PDF). Syndax Pharmaceuticals. 27 August 2013. Archived from the original (PDF) on 17 June 2016.
- PMID 38377195.
