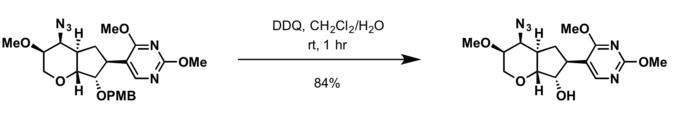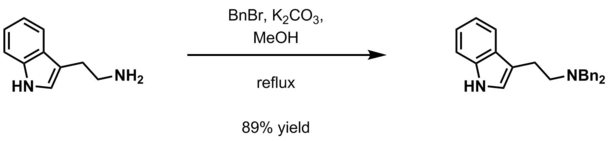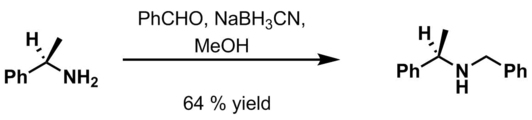Benzyl group
In organic chemistry, benzyl is the substituent or molecular fragment possessing the structure R−CH2−C6H5. Benzyl features a benzene ring (C6H6) attached to a methylene group (−CH2−) group.[1]
Nomenclature
In
Abbreviations
Benzyl is most commonly abbreviated Bn. For example,
Reactivity of benzylic centers
The enhanced reactivity of benzylic positions is attributed to the low
| Bond | Bond | Bond-dissociation energy[2][3] | Comment | |
|---|---|---|---|---|
| (kcal/mol) | (kJ/mol) | |||
| C6H5CH2−H | benzylic C−H bond | 90 | 377 | akin to allylic C−H bonds such bonds show enhanced reactivity |
| H3C−H | methyl C−H bond
|
105 | 439 | one of the strongest aliphatic C−H bonds |
| C2H5−H | ethyl C−H bond | 101 | 423 | slightly weaker than H3C−H |
| C6H5−H | phenyl C−H bond
|
113 | 473 | comparable to vinyl radical, rare |
| CH2=CHCH2−H | allylic C–H bond
|
89 | 372 | similar to benzylic C-H |
| (C6H4)2CH−H | fluorenyl C–H bond
|
80 | more activated vs diphenylmethyle ( pKa = 22.6)
| |
| (C6H5)2CH−H | diphenylmethyl C–H bond | 82 | "doubly benzylic" ( pKa = 32.2)
| |
| (C6H5)3C−H | trityl C–H bond
|
81 | 339 | "triply benzylic" |
The weakness of the C−H bond reflects the stability of the benzylic radical. For related reasons, benzylic substituents exhibit enhanced reactivity, as in
Millions of tonnes of terephthalic acid are produced annually by this method.[4]
Functionalization at the benzylic position
In a few cases, these benzylic transformations occur under conditions suitable for lab synthesis. The Wohl-Ziegler reaction will brominate a benzylic C–H bond: (ArCHR2 → ArCBrR2).[5] Any non-tertiary benzylic alkyl group will be oxidized to a carboxyl group by aqueous potassium permanganate (KMnO4) or concentrated nitric acid (HNO3): (ArCHR2 → ArCOOH).[6] Finally, the complex of chromium trioxide and 3,5-dimethylpyrazole (CrO3−dmpyz) will selectively oxidize a benzylic methylene group to a carbonyl: (ArCH2R → ArC(O)R).[7] 2-iodoxybenzoic acid in DMSO performs similarly.[8]
As a protecting group
Benzyl groups are occasionally employed as protecting groups in organic synthesis. Their installation and especially their removal require relatively harsh conditions, so benzyl is not typically preferred for protection.[9]
Alcohol protection
Benzyl is commonly used in organic synthesis as a robust protecting group for alcohols and carboxylic acids.
- Treatment of alcohol with a strong base such as powdered potassium hydroxide or sodium hydride and benzyl halide (BnCl or BnBr)[9][10]
- Monobenzylation of Ag2O in dimethylformamide (DMF) at ambient to elevated temperatures[11]
- Primary alcohols can be selectively benzylated in presence of phenol functional groups using Cu(acac)2[12]
Deprotection methods
Benzyl ethers can be removed under
- Removed using hydrogenolysis[13]
- Single electron process with Na/NH3 or Li/NH3
- Benzyl protecting groups can be removed using a wide range of oxidizing agents including:
- CrO3/acetic acid at ambient temperature
- Ozone
- N-Bromosuccinimide (NBS)
- N-Iodosuccinimide (NIS)
- Trimethylsilyl iodide (Me3SiI) in dichloromethaneat ambient temperature (selectivity can be achieved under specific conditions)
The p-methoxybenzyl protecting group
p-Methoxybenzyl (PMB) is used as a
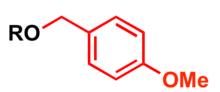
- Strong base such as powdered potassium hydroxide or sodium hydride and p-methoxybenzyl halide (chloride or bromide)[14][15]
- 4-methoxybenzyl-2,2,2-trichloroacetimidate can be used to install the PMB group in presence of:
- Scandium (III) triflate (Sc(OTf)3) in toluene at 0 °C[16]
- Trifluoromethanesulfonic acid (TfOH) in dichloromethane at 0 °C[17]
Deprotection methods
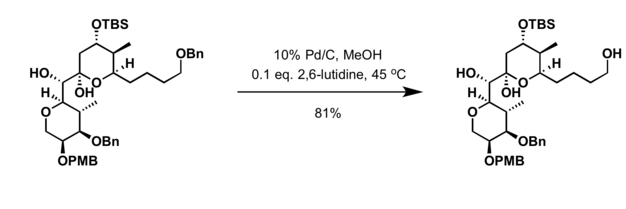
- 2,3-Dichloro-5,6-dicyano-p-benzoquinone (DDQ)[18]
- Conditions for deprotection of benzyl group are applicable for cleavage of the PMB protecting group
Amine protection
The benzyl group is occasionally used as a protecting group for amines in organic synthesis. Other methods exist.[9]
- Aqueous potassium carbonate and benzyl halide (BnCl, BnBr) in methanol[19]
- Benzaldehyde, 6 M HCl and NaBH3CN in methanol[20]
Deprotection methods
- Hydrogenation in the presence of the palladium catalyst[21]
See also
References
- ISBN 9780387448978.
- PMID 28281752.
- .
- ISBN 978-3527306732.
- OCLC 1007924903.)
{{cite book}}: CS1 maint: location missing publisher (link) CS1 maint: multiple names: authors list (link - )
- ISBN 9780470842898
- PMID 11457049.
- ^ S2CID 83393227.
- .
- ISSN 0022-3263.
- .
- PMID 12605509.
- .
- ISSN 0022-3263.
- PMID 17985906.
- ISSN 1521-3765.
- PMID 14601979.
- ISSN 0022-3263.
- .
- PMID 14723540.
- .
External links
 Chemistry portal
Chemistry portal Quotations related to Benzyl group at Wikiquote
Quotations related to Benzyl group at Wikiquote