Fertilizer

A fertilizer (
Historically fertilization came from natural or organic sources: compost, animal manure, human manure, harvested minerals, crop rotations and byproducts of human-nature industries (i.e. fish processing waste, or bloodmeal from animal slaughter). However, starting in the 19th century, after innovations in plant nutrition, an agricultural industry developed around synthetically created fertilizers. This transition was important in transforming the global food system, allowing for larger-scale industrial agriculture with large crop yields.
The use of artificial and industrially-applied fertilizers has caused environmental consequences such as
History




Management of
The
The 1910s and 1920s witnessed the rise of the
The development of synthetic nitrogen fertilizer has significantly supported global population growth. It has been estimated that almost half the people on the Earth are currently fed as a result of synthetic nitrogen fertilizer use.[16] The use of phosphate fertilizers has also increased from 9 million tonnes per year in 1960 to 40 million tonnes per year in 2000.
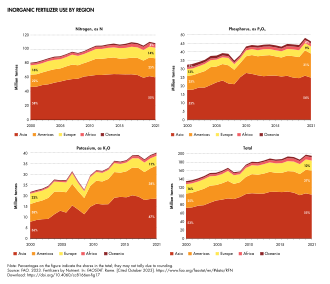
Agricultural use of inorganic fertilizers in 2021 was 195 million tonnes of nutrients, of which 56% was nitrogen.[17] Asia represented 53% of world total agricultural use of inorganic fertilizers in 2021, followed by the Americas (29%), Europe (12%), Africa (4%) and Oceania (2%). This ranking of the regions is the same for all nutrients. The main users of inorganic fertilizers are, in descending order, China, India, Brazil and the United States of America (see Table 15), with China the largest user of each nutrient.[17]
A maize crop yielding 6–9 tonnes of grain per hectare (2.5 acres) requires 31–50 kilograms (68–110 lb) of phosphate fertilizer to be applied; soybean crops require about half, 20–25 kg per hectare.[18] Yara International is the world's largest producer of nitrogen-based fertilizers.[19]
Mechanism

Fertilizers enhance the growth of plants. This goal is met in two ways, the traditional one being additives that provide nutrients. The second mode by which some fertilizers act is to enhance the effectiveness of the soil by modifying its water retention and aeration. This article, like many on fertilizers, emphasises the nutritional aspect. Fertilizers typically provide, in varying proportions:[21]
- three main macronutrients (NPK):
- Nitrogen (N): leaf growth
- Phosphorus (P): development of roots, flowers, seeds, fruit;
- Potassium (K): strong stem growth, movement of water in plants, promotion of flowering and fruiting;
- three secondary macronutrients: calcium (Ca), magnesium (Mg), and sulfur (S);
- micronutrients: (V).
The nutrients required for healthy plant life are classified according to the elements, but the elements are not used as fertilizers. Instead
Microbiological considerations
Two sets of
- Urease
The first is the hydrolysis (reaction with water) of urea (CO(NH2)2). Many soil bacteria possess the enzyme urease, which catalyzes the conversion of urea to ammonium ion (NH+4) and bicarbonate ion (HCO−3).
- Ammonia oxidation
Classification
Fertilizers are classified in several ways. They are classified according to whether they provide a single nutrient (e.g., K, P, or N), in which case they are classified as "straight fertilizers". "Multinutrient fertilizers" (or "complex fertilizers") provide two or more nutrients, for example N and P. Fertilizers are also sometimes classified as inorganic (the topic of most of this article) versus organic. Inorganic fertilizers exclude carbon-containing materials except ureas. Organic fertilizers are usually (recycled) plant- or animal-derived matter. Inorganic are sometimes called synthetic fertilizers since various chemical treatments are required for their manufacture.[23]
Single nutrient ("straight") fertilizers
The main nitrogen-based straight fertilizer is ammonia (NH3) ammonium (NH4+) or its solutions, including:
- Ammonium nitrate (NH4NO3) is also widely used.
- Urea (CO(NH2)2), another popular source of nitrogen, having the advantage that it is solid and non-explosive, unlike ammonia and ammonium nitrate.
- Calcium ammonium nitrate (Ca(NO3)2 · NH4 · 10 H2O), reportedly holding a small share of the nitrogen fertilizer market (4% in 2007).[24]
The main straight phosphate fertilizers are the superphosphates:
- "Single superphosphate" (SSP) consisting of 14–18% P2O5, again in the form of Ca(H2PO4)2, but also phosphogypsum (CaSO4 · 2 H2O).
- Triple superphosphate(TSP) typically consists of 44–48% of P2O5 and no gypsum.
A mixture of single superphosphate and triple superphosphate is called double superphosphate. More than 90% of a typical superphosphate fertilizer is water-soluble.
The main potassium-based straight fertilizer is
Multinutrient fertilizers
These fertilizers are common. They consist of two or more nutrient components.
- Binary (NP, NK, PK) fertilizers
Major two-component fertilizers provide both nitrogen and phosphorus to the plants. These are called NP fertilizers. The main NP fertilizers are
- NPK fertilizers
NPK fertilizers are three-component fertilizers providing nitrogen, phosphorus, and potassium. There exist two types of NPK fertilizers: compound and blends. Compound NPK fertilizers contain chemically bound ingredients, while blended NPK fertilizers are physical mixtures of single nutrient components.
Micronutrients
Production
The production of synthetic, or inorganic, fertilizers requires prepared chemicals, whereas organic fertilizers are derived from the organic processes of plants and animals in biological processes using biochemicals.
Nitrogen fertilizers

Nitrogen fertilizers are made from
Deposits of
Phosphate fertilizers

Phosphate fertilizers are obtained by extraction from
Potassium fertilizers
Potash is a mixture of potassium minerals used to make potassium (chemical symbol: K) fertilizers. Potash is soluble in water, so the main effort in producing this nutrient from the ore involves some purification steps; e.g., to remove sodium chloride (NaCl) (common salt). Sometimes potash is referred to as K2O, as a matter of convenience to those describing the potassium content. In fact, potash fertilizers are usually potassium chloride, potassium sulfate, potassium carbonate, or potassium nitrate.[36]
NPK fertilizers
There are four major routes for manufacturing NPK fertilizers (named for their main ingredients: nitrogen (N), phosphorus (P), and potassium (K)):
- steam granulation,
- chemical granulation,
- compaction,
- bulk blending.
The first three processes are used to produce compound NPKs. During steam granulation raw materials are mixed and further granulated using steam as binding agent. Chemical granulation process is based on chemical reactions between liquid raw materials (such as phosphoric acid, sulfuric acid, ammonia) and solid raw materials (such as potassium chloride, recycle material). Compaction implements high pressure to agglomerate dry powder materials. Lastly, bulk blends are produced by mixing straight fertilizers.
Organic fertilizers


"
Fertilizers of an organic origin (the first definition) include
In terms of volume, peat is the most widely used packaged organic soil amendment. It is an immature form of coal and improves the soil by aeration and absorbing water but confers no nutritional value to the plants. It is therefore not a fertilizer as defined in the beginning of the article, but rather an amendment. Coir, (derived from coconut husks), bark, and sawdust when added to soil all act similarly (but not identically) to peat and are also considered organic soil amendments – or texturizers – because of their limited nutritive inputs. Some organic additives can have a reverse effect on nutrients – fresh sawdust can consume soil nutrients as it breaks down, and may lower soil pH – but these same organic texturizers (as well as compost, etc.) may increase the availability of nutrients through improved cation exchange, or through increased growth of microorganisms that in turn increase availability of certain plant nutrients. Organic fertilizers such as composts and manures may be distributed locally without going into industry production, making actual consumption more difficult to quantify.
Statistics

| Country | Total N use (Mt pa) |
N use for feed and pasture (Mt pa) |
|---|---|---|
| China | 18.7 | 3.0 |
| India | 11.9 | n/a[40] |
| U.S. | 9.1 | 4.7 |
| France | 2.5 | 1.3 |
| Germany | 2.0 | 1.2 |
| Brazil | 1.7 | 0.7 |
| Canada | 1.6 | 0.9 |
| Turkey | 1.5 | 0.3 |
| UK | 1.3 | 0.9 |
| Mexico | 1.3 | 0.3 |
| Spain | 1.2 | 0.5 |
| Argentina | 0.4 | 0.1 |
China has become the largest producer and consumer of nitrogen fertilizers[41] while Africa has little reliance on nitrogen fertilizers.[42] Agricultural and chemical minerals are very important in industrial use of fertilizers, which is valued at approximately $200 billion.[43] Nitrogen has a significant impact in the global mineral use, followed by potash and phosphate. The production of nitrogen has drastically increased since the 1960s. Phosphate and potash have increased in price since the 1960s, which is larger than the consumer price index.[43] Potash is produced in Canada, Russia and Belarus, together making up over half of the world production.[43] Potash production in Canada rose in 2017 and 2018 by 18.6%.[44] Conservative estimates report 30 to 50% of crop yields are attributed to natural or synthetic commercial fertilizers.[36][45] Fertilizer consumption has surpassed the amount of farmland in the United States.[43]
Data on the fertilizer consumption per hectare
Application


Fertilizers are commonly used for growing all crops, with application rates depending on the soil fertility, usually as measured by a soil test and according to the particular crop. Legumes, for example, fix nitrogen from the atmosphere and generally do not require nitrogen fertilizer.
Liquid vs solid
Fertilizers are applied to crops both as solids and as liquid. About 90% of fertilizers are applied as solids. The most widely used solid inorganic fertilizers are urea, diammonium phosphate and potassium chloride.[49] Solid fertilizer is typically granulated or powdered. Often solids are available as prills, a solid globule. Liquid fertilizers comprise anhydrous ammonia, aqueous solutions of ammonia, aqueous solutions of ammonium nitrate or urea. These concentrated products may be diluted with water to form a concentrated liquid fertilizer (e.g., UAN). Advantages of liquid fertilizer are its more rapid effect and easier coverage.[21] The addition of fertilizer to irrigation water is called "fertigation".[36]
Urea
Urea is highly soluble in water and is therefore also very suitable for use in fertilizer solutions (in combination with ammonium nitrate: UAN), e.g., in 'foliar feed' fertilizers. For fertilizer use, granules are preferred over prills because of their narrower particle size distribution, which is an advantage for mechanical application.
Urea is usually spread at rates of between 40 and 300 kg/ha (35 to 270 lbs/acre) but rates vary. Smaller applications incur lower losses due to leaching. During summer, urea is often spread just before or during rain to minimize losses from volatilization (a process wherein nitrogen is lost to the atmosphere as ammonia gas).
Because of the high nitrogen concentration in urea, it is very important to achieve an even spread. Drilling must not occur on contact with or close to seed, due to the risk of germination damage. Urea dissolves in water for application as a spray or through irrigation systems.
In grain and cotton crops, urea is often applied at the time of the last cultivation before planting. In high rainfall areas and on sandy soils (where nitrogen can be lost through leaching) and where good in-season rainfall is expected, urea can be side- or top-dressed during the growing season. Top-dressing is also popular on pasture and forage crops. In cultivating sugarcane, urea is side-dressed after planting, and applied to each ratoon crop.
Because it absorbs moisture from the atmosphere, urea is often stored in closed containers.
Overdose or placing urea near seed is harmful.[50]
Slow- and controlled-release fertilizers
Foliar application
Foliar fertilizers are applied directly to leaves. This method is almost invariably used to apply water-soluble straight nitrogen fertilizers and used especially for high-value crops such as fruits. Urea is the most common foliar fertilizer.[21]

Chemicals that affect nitrogen uptake

Various chemicals are used to enhance the efficiency of nitrogen-based fertilizers. In this way farmers can limit the
Overfertilization
Careful use of fertilization technologies is important because excess nutrients can be detrimental.[54] Fertilizer burn can occur when too much fertilizer is applied, resulting in damage or even death of the plant. Fertilizers vary in their tendency to burn roughly in accordance with their salt index.[55][56]
Environmental effects
Synthetic fertilizer used in agriculture has wide-reaching environmental consequences
According to the
In order to mitigate environmental and food security concerns, the international community has included food systems in Sustainable Development Goal 2 which focuses on creating a climate-friendly and sustainable food production system.[58] Most policy and regulatory approaches to address these issues focus on pivoting agricultural practices towards sustainable or regenerative agricultural practices: these use less synthetic fertilizers, better soil management (for example no-till agriculture) and more organic fertilizers.

For each ton of phosphoric acid produced by the processing of phosphate rock, five tons of waste are generated. This waste takes the form of impure, useless, radioactive solid called phosphogypsum. Estimates range from 100,000,000 and 280,000,000 tons of phosphogypsum waste produced annually worldwide.[59]
Water

Phosphorus and nitrogen fertilizers can affect soil, surface water, and groundwater due to the dispersion of minerals
The nitrogen-rich compounds found in fertilizer runoff are the primary cause of serious oxygen depletion in many parts of oceans, especially in coastal zones, lakes and rivers. The resulting lack of dissolved oxygen greatly reduces the ability of these areas to sustain oceanic fauna.[66] The number of oceanic dead zones near inhabited coastlines is increasing.[67]
As of 2006, the application of nitrogen fertilizer is being increasingly controlled in northwestern Europe[68] and the United States.[69][70] In cases where eutrophication can be reversed, it may nevertheless take decades[71] and significant soil management[72] before the accumulated nitrates in groundwater can be broken down by natural processes.
Nitrate pollution
Only a fraction of the nitrogen-based fertilizers is converted to plant matter. The remainder accumulates in the soil or is lost as run-off.
Nitrate levels above 10 mg/L (10 ppm) in groundwater can cause 'blue baby syndrome' (acquired methemoglobinemia).[78] The nutrients, especially nitrates, in fertilizers can cause problems for natural habitats and for human health if they are washed off soil into watercourses or leached through soil into groundwater.[79] Run-off can lead to fertilizing blooms of algae that use up all the oxygen and leave huge "dead zones" behind where other fish and aquatic life can not live.[80]
Soil
Acidification
Soil acidification refers to the process by which the pH level of soil becomes more acidic over time. Soil pH is a measure of the soil's acidity or alkalinity and is determined on a scale from 0 to 14, with 7 being neutral. A pH value below 7 indicates acidic soil, while a pH value above 7 indicates alkaline or basic soil.
Soil acidification is a significant concern in agriculture and horticulture. It refers to the process of the soil becoming more acidic over time.
Nitrogen-containing fertilizers can cause soil acidification when added.[81][82] This may lead to decrease in nutrient availability which may be offset by liming. These fertilizers release ammonium or nitrate ions, which can acidify the soil as they undergo chemical reactions.
When these nitrogen-containing fertilizers are added to the soil, they increase the concentration of hydrogen ions (H+) in the soil solution, which lowers the pH of the soil.
Accumulation of toxic elements
Cadmium
The concentration of
Fluoride
Phosphate rocks contain high levels of fluoride. Consequently, the widespread use of phosphate fertilizers has increased soil fluoride concentrations.[89] It has been found that food contamination from fertilizer is of little concern as plants accumulate little fluoride from the soil; of greater concern is the possibility of fluoride toxicity to livestock that ingest contaminated soils.[93][94] Also of possible concern are the effects of fluoride on soil microorganisms.[93][94][95]
Radioactive elements
The radioactive content of the fertilizers varies considerably and depends both on their concentrations in the parent mineral and on the fertilizer production process.[89][96] Uranium-238 concentrations can range from 7 to 100 pCi/g (picocuries per gram) in phosphate rock[97] and from 1 to 67 pCi/g in phosphate fertilizers.[98][99][100] Where high annual rates of phosphorus fertilizer are used, this can result in uranium-238 concentrations in soils and drainage waters that are several times greater than are normally present.[99][101] However, the impact of these increases on the risk to human health from radinuclide contamination of foods is very small (less than 0.05 mSv/y).[99][102][103]
Other metals
Steel industry wastes, recycled into fertilizers for their high levels of zinc (essential to plant growth), wastes can include the following toxic metals: lead[104] arsenic, cadmium,[104] chromium, and nickel. The most common toxic elements in this type of fertilizer are mercury, lead, and arsenic.[105][106][107] These potentially harmful impurities can be removed; however, this significantly increases cost. Highly pure fertilizers are widely available and perhaps best known as the highly water-soluble fertilizers containing blue dyes used around households, such as Miracle-Gro. These highly water-soluble fertilizers are used in the plant nursery business and are available in larger packages at significantly less cost than retail quantities. Some inexpensive retail granular garden fertilizers are made with high purity ingredients.
Trace mineral depletion
Attention has been addressed to the decreasing concentrations of elements such as iron, zinc, copper and magnesium in many foods over the last 50–60 years.[108][109] Intensive farming practices, including the use of synthetic fertilizers are frequently suggested as reasons for these declines and organic farming is often suggested as a solution.[109] Although improved crop yields resulting from NPK fertilizers are known to dilute the concentrations of other nutrients in plants,[108][110] much of the measured decline can be attributed to the use of progressively higher-yielding crop varieties that produce foods with lower mineral concentrations than their less-productive ancestors.[108][111][112] It is, therefore, unlikely that organic farming or reduced use of fertilizers will solve the problem; foods with high nutrient density are posited to be achieved using older, lower-yielding varieties or the development of new high-yield, nutrient-dense varieties.[108][113]
Fertilizers are, in fact, more likely to solve trace mineral deficiency problems than cause them: In Western Australia deficiencies of zinc, copper, manganese, iron and molybdenum were identified as limiting the growth of broad-acre crops and pastures in the 1940s and 1950s.[114] Soils in Western Australia are very old, highly weathered and deficient in many of the major nutrients and trace elements.[114] Since this time these trace elements are routinely added to fertilizers used in agriculture in this state.[114] Many other soils around the world are deficient in zinc, leading to deficiency in both plants and humans, and zinc fertilizers are widely used to solve this problem.[115]
Changes in soil biology
High levels of fertilizer may cause the breakdown of the symbiotic relationships between plant roots and mycorrhizal fungi.[116]
Energy consumption and sustainability
In the US in 2004, 317 billion cubic feet of natural gas were consumed in the industrial production of ammonia, less than 1.5% of total U.S. annual consumption of natural gas.[117] A 2002 report suggested that the production of ammonia consumes about 5% of global natural gas consumption, which is somewhat under 2% of world energy production.[118]
Ammonia is produced from natural gas and air.[119] The cost of natural gas makes up about 90% of the cost of producing ammonia.[120] The increase in price of natural gases over the past decade, along with other factors such as increasing demand, have contributed to an increase in fertilizer price.[121]
Contribution to climate change
The amount of
Atmosphere

Through the increasing use of nitrogen fertilizer, which was used at a rate of about 110 million tons (of N) per year in 2012,[126][127] adding to the already existing amount of reactive nitrogen, nitrous oxide (N2O) has become the third most important greenhouse gas after carbon dioxide and methane. It has a global warming potential 296 times larger than an equal mass of carbon dioxide and it also contributes to stratospheric ozone depletion.[128] By changing processes and procedures, it is possible to mitigate some, but not all, of these effects on anthropogenic climate change.[129]
Methane emissions from crop fields (notably rice paddy fields) are increased by the application of ammonium-based fertilizers. These emissions contribute to global climate change as methane is a potent greenhouse gas.[130][131]
Policy
Regulation
In Europe, problems with high nitrate concentrations in runoff are being addressed by the European Union's Nitrates Directive.[132] Within Britain, farmers are encouraged to manage their land more sustainably in 'catchment-sensitive farming'.[133] In the US, high concentrations of nitrate and phosphorus in runoff and drainage water are classified as nonpoint source pollutants due to their diffuse origin; this pollution is regulated at the state level.[134] Oregon and Washington, both in the United States, have fertilizer registration programs with on-line databases listing chemical analyses of fertilizers.[135][136]
In China, regulations have been implemented to control the use of N fertilizers in farming. In 2008, Chinese governments began to partially withdraw fertilizer subsidies, including subsidies to fertilizer transportation and to electricity and natural gas use in the industry. In consequence, the price of fertilizer has gone up and large-scale farms have begun to use less fertilizer. If large-scale farms keep reducing their use of fertilizer subsidies, they have no choice but to optimize the fertilizer they have which would therefore gain an increase in both grain yield and profit.[137]
In March 2022, the United States Department of Agriculture announced a new $250M grant to promote American fertilizer production. Part of the Commodity Credit Corporation, the grant program will support fertilizer production that is independent of dominant fertilizer suppliers, made in America, and utilizing innovative production techniques to jumpstart future competition.[138]
Two types of agricultural management practices include organic agriculture and conventional agriculture. The former encourages soil fertility using local resources to maximize efficiency. Organic agriculture avoids synthetic agrochemicals. Conventional agriculture uses all the components that organic agriculture does not use.[139]
See also
- Agroecology
- Circulus (theory)
- Fertigation
- Food and Agriculture Organization
- History of organic farming
- Milorganite
- Leaf Color Chart
- Nutrient Recovery and Reuse
- Phosphogypsum
- Peak phosphorus
- Soil defertilisation
- Seaweed fertilizer
References
- ^ ISBN 978-3527306732.
- ^ "Fritz Haber". Science History Institute. 1 June 2016. Retrieved 16 December 2022.
- ^ a b Mbow et al. 2019.
- ^ "Total fertilizer production by nutrient". Our World in Data. Retrieved 7 March 2020.
- ^ "World population with and without synthetic nitrogen fertilizers". Our World in Data. Retrieved 5 March 2020.
- ISBN 978-1-119-70634-2.
- ISSN 2073-445X.
- ISBN 978-3-5253-1705-1.
- JSTOR 10.3098/ah.2014.88.1.68.
- ^ This article incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). "Lawes, Sir John Bennet". Encyclopædia Britannica (11th ed.). Cambridge University Press.
- ISBN 978-0-486-64235-2.
- ISBN 978-0-19-516582-1.
- ISBN 978-0-19-858159-8.
- ^ Philpott, Tom. "A Brief History of Our Deadly Addiction to Nitrogen Fertilizer". Mother Jones. Retrieved 24 March 2021.
- .
- S2CID 94880859. Archived from the originalon 23 July 2010. Retrieved 22 October 2010.
- ^ ISBN 978-92-5-138262-2. Retrieved 13 December 2023.
- S2CID 53490640.
- ^ "Mergers in the fertiliser industry". The Economist. 18 February 2010. Retrieved 21 February 2010.
- )
- ^ ISBN 978-3527306732.
- S2CID 6245255.
- ISBN 978-1-4398-1610-3.
- ^ ISBN 978-0-262-69313-4.
- ^ "Summary of State Fertilizer Laws" (PDF). EPA. Archived (PDF) from the original on 9 October 2022. Retrieved 14 March 2013.
- ^ "Label Requirements of specialty and other bagged fertilizers". Michigan Department of Agriculture and Rural Development. Retrieved 14 March 2013.
- ^ "National Code of Practice for Fertilizer Description & Labelling" (PDF). Australian Government Department of Agriculture, Fisheries and Forestry. Archived from the original (PDF) on 28 February 2015. Retrieved 14 March 2013.
- ^ "AESL Plant Analysis Handbook – Nutrient Content of Plant". Aesl.ces.uga.edu. Retrieved 11 September 2015.
- ISBN 978-1-878148-05-6.
- ^ "Boron Deficiency". Archived from the original on 6 March 2019. Retrieved 4 March 2019.
- ^ "Supplemental technical report for sodium nitrate (crops)". ams.usda.gov. Archived from the original on 14 July 2014. Retrieved 6 July 2014.
- ^ "Caliche Ore". sqm.com. Archived from the original on 14 July 2014. Retrieved 6 July 2014.
- S2CID 1450932.
- ISBN 978-0-08-037941-8.
- ^ EFMA (2000). "Best available techniques for pollution prevention and control in the European fertilizer industry. Booklet No. 7 of 8: Production of NPK fertilizers by the nitrophosphate route" (PDF). fertilizerseurope.com. European Fertilizer Manufacturers' Association. Archived from the original (PDF) on 29 July 2014. Retrieved 28 June 2014.
- ^
- S2CID 20113235– via Springer Link.
- S2CID 242794287.
- ^ Livestock's Long Shadow: Environmental Issues and Options, Table 3.3. Retrieved 29 June 2009. United Nations Food and Agriculture Organization.
- ^ "Production & Inputs | Government of India, Department of Fertilizers, Ministry of Chemicals and Fertilizers".
- ISBN 978-1-119-94253-5.
- ISBN 978-0-262-01856-2.
- ^ ISBN 978-1-107-07491-0.
- ^ "Industry Stats – Fertilizer Canada". Fertilizer Canada. Archived from the original on 4 April 2018. Retrieved 28 March 2018.
- .
- ^ "Fertilizer consumption (Kilograms per hectare of arable land) | Data".
- ^ "Eurostat - Data Explorer". Archived from the original on 6 October 2014. Retrieved 19 October 2011.
- ^ Arable land
- ^ "About Fertilizers Home Page". fertilizer.org. International Fertilizer Association. Retrieved 19 December 2017.[permanent dead link]
- ^ Mikkelsen, R.L. (2007). "Biuret in Urea Fertilizers" (PDF). Better Crops. 91 (3): 6–7. Archived from the original (PDF) on 22 December 2015. Retrieved 2 May 2015.
- ISBN 978-3527306732.
- LCCN 2001025292. Retrieved 9 December 2011.
- PMID 26902689.
- ^ "Nitrogen Fertilization: General Information". Hubcap.clemson.edu. Archived from the original on 29 June 2012. Retrieved 17 June 2012.
- ISBN 978-0-292-72849-3.
- ^ "Understanding Salt index of fertilizers" (PDF). Archived from the original (PDF) on 28 May 2013. Retrieved 22 July 2012.
- PMID 31211785.
- ^ United Nations (2017) Resolution adopted by the General Assembly on 6 July 2017, Work of the Statistical Commission pertaining to the 2030 Agenda for Sustainable Development (A/RES/71/313)
- S2CID 24111765.
- ^ S2CID 227522409.
- ^ "Environmental impact of nitrogen and phosphorus fertilisers in high rainfall areas". Agriculture and Food | Department of Primary Industries and Regional Development. Retrieved 9 April 2018.
- ^ "The Sources and Solutions: Agriculture". US Environmental Protection Agency. Archived from the original on 5 April 2023. Retrieved 4 May 2023.
- ^
- ^ "UPDATE (9:30 a.m.): Do-not-drink water advisory lifted for City of Toledo | Toledo Free Press". Archived from the original on 5 August 2014. Retrieved 5 August 2014.
- PMID 23676698.
- ^ "Rapid Growth Found in Oxygen-Starved Ocean 'Dead Zones'", NY Times, 14 August 2008
- ^ John Heilprin, Associated Press. "Discovery Channel :: News – Animals :: U.N.: Ocean 'Dead Zones' Growing". Dsc.discovery.com. Archived from the original on 18 June 2010. Retrieved 25 August 2010.
- hdl:1854/LU-3072131.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ "A Farmer's Guide To Agriculture and Water Quality Issues: 3. Environmental Requirements & Incentive Programs For Nutrient Management". cals.ncsu.edu. Archived from the original on 23 September 2015. Retrieved 3 July 2014.
- ^ State-EPA Nutrient Innovations Task Group (2009). "An Urgent Call to Action – Report of the State-EPA Nutrient Innovations Task Group" (PDF). epa.gov. Archived (PDF) from the original on 9 October 2022. Retrieved 3 July 2014.
- ^ "Study shows eutrophic lakes may not recover for a millennium". news.wisc.edu. Retrieved 3 November 2022.
- ISBN 978-0-12-804792-7, retrieved 3 November 2022
- ISBN 978-94-007-7813-9.
- ^ C. J. Rosen; B. P. Horgan (9 January 2009). "Preventing Pollution Problems from Lawn and Garden Fertilizers". Extension.umn.edu. Archived from the original on 10 March 2014. Retrieved 25 August 2010.
- .
- ^ "NOFA Interstate Council: The Natural Farmer. Ecologically Sound Nitrogen Management. Mark Schonbeck". Nofa.org. 25 February 2004. Archived from the original on 24 March 2004. Retrieved 25 August 2010.
- PMID 18444903.
- PMID 10903623.
- ^ Nitrogen and Water
- ^ Biello, David (14 March 2008). "Fertilizer Runoff Overwhelms Streams and Rivers--Creating Vast "Dead Zones"". Scientific American.
- PMID 19423798.
- .
- .
- (PDF) from the original on 9 October 2022. Retrieved 27 June 2014.
- ^ Zapata, F.; Roy, R.N. (2004). "Use of Phosphate Rocks for Sustainable Agriculture: Secondary nutrients, micronutrients, liming effect and hazardous elements associated with phosphate rock use". fao.org. FAO. Retrieved 27 June 2014.
- .
- .
- PMID 9496656.
- ^ ISBN 9780123942784.
- ^ Oosterhuis, F.H.; Brouwer, F.M.; Wijnants, H.J. (2000). "A possible EU wide charge on cadmium in phosphate fertilisers: Economic and environmental implications" (PDF). dare.ubvu.vu.nl. Archived (PDF) from the original on 9 October 2022. Retrieved 27 June 2014.
- ^ "Putting all the cards on the table" (PDF). Fertilizers International. fertilizerseurope.com. 2014. Archived from the original (PDF) on 8 August 2014.
- ^ Wates, J. (2014). "Revision of the EU fertilizer regulation and cadmium content of fertilisers". iatp.org. Retrieved 27 June 2014.
- ^ PMID 18020303.
- ^ .
- S2CID 1225884.
- ^ Mortvedt, JJ; Beaton, JD. "Heavy Metal and Radionuclide Contaminants in Phosphate Fertilizers". Archived from the original on 26 July 2014. Retrieved 16 July 2014.
- ^ "TENORM: Fertilizer and Fertilizer Production Wastes". US EPA. 2016. Retrieved 30 August 2017.
- ^ Khater, A. E. M. (2008). "Uranium and heavy metals in phosphate fertilizers" (PDF). radioecology.info. Archived from the original (PDF) on 24 July 2014. Retrieved 17 July 2014.
- ^ a b c NCRP (1987). Radiation Exposure of the U.S. Population from Consumer Products and Miscellaneous Sources. National Council on Radiation Protection and Measurements. pp. 29–32. Retrieved 17 July 2014.[permanent dead link]
- PMID 8056596.
- .
- ^ Hanlon, E. A. (2012). "Naturally Occurring Radionuclides in Agricultural Products". edis.ifas.ufl.edu. University of Florida. Archived from the original on 25 July 2014. Retrieved 17 July 2014.
- )
- ^ a b Wilson, Duff (3 July 1997). "Business | Fear in the Fields – How Hazardous Wastes Become Fertilizer – Spreading Heavy Metals on Farmland Is Perfectly Legal, But Little Research Has Been Done To Find Out Whether It's Safe | Seattle Times Newspaper". Community.seattletimes.nwsource.com. Retrieved 25 August 2010.
- ^ "Waste Lands: The Threat of Toxic Fertilizer". Pirg.org. 3 July 1997. Archived from the original on 26 November 2010. Retrieved 25 August 2010.
- ^ mindfully.org. "Waste Lands: The Threat of Toxic Fertilizer Released by PIRG Toxic Wastes Found in Fertilizers Cat Lazaroff / ENS 7may01". Mindfully.org. Archived from the original on 11 January 2002. Retrieved 25 August 2010.
- ^ Zapata, F; Roy, RN (2004). Use of phosphate rocks for sustainable agriculture (PDF). Rome: FAO. p. 82. Retrieved 16 July 2014.[permanent dead link]
- ^ S2CID 13595345.
- ^ S2CID 372456.
- )
- PMID 19013359.
- .
- .
- ^ ISBN 978-0-7307-0057-9.
- ^ "Zinc in Soils and Crop Nutrition". Scribd.com. 25 August 2010. Retrieved 17 June 2012.
- ISBN 978-0-88192-611-8.
- ^ Aleksander Abram; D. Lynn Forster (2005). "A Primer on Ammonia, Nitrogen Fertilizers, and Natural Gas Markets". Department of Agricultural, Environmental, and Development Economics, Ohio State University: 38.
{{cite journal}}: Cite journal requires|journal=(help) - ^ IFA – Statistics – Fertilizer Indicators – Details – Raw material reserves, (2002–10) Archived 24 April 2008 at the Wayback Machine
- ISBN 978-3-527-30673-2.
- ^ Sawyer JE (2001). "Natural gas prices affect nitrogen fertilizer costs". IC-486. 1: 8.
- ^ "Table 8—Fertilizer price indexes, 1960–2007". Archived from the original on 6 March 2010.
- ^ "Carbon emissions from fertilizers could be reduced by as much as 80% by 2050". Science Daily. University of Cambridge. Retrieved 17 February 2023.
- ^ "How Fertilizer Is Making Climate Change Worse". BloombergQuint. 10 September 2020. Retrieved 25 March 2021.
- ^ "Nitrogen fertiliser use could 'threaten global climate goals'". Carbon Brief. 7 October 2020. Retrieved 25 March 2021.
- ^ FAO (2012). Current world fertilizer trends and outlook to 2016 (PDF). Rome: Food and Agriculture Organization of the United Nations. p. 13. Archived from the original (PDF) on 18 May 2017. Retrieved 3 July 2014.
- PMID 18202647.
- ^ "Human alteration of the nitrogen cycle, threats, benefits and opportunities" Archived 14 January 2009 at the Wayback Machine UNESCO – SCOPE Policy briefs, April 2007
- S2CID 905322. Archived from the original(PDF) on 24 September 2015. Retrieved 3 July 2014.
- S2CID 4351801.)
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: numeric names: authors list (link - S2CID 31666406.
- ^ European Union (15 January 2024). "Nitrates Directive".
- ^ Defra. "Catchment-Sensitive Farming". Archived from the original on 30 June 2011.
- ^ "Polluted Runoff: Nonpoint Source Pollution". EPA. Retrieved 23 July 2014.
- ^ "Washington State Dept. of Agriculture Fertilizer Product Database". Agr.wa.gov. 23 May 2012. Archived from the original on 30 October 2013. Retrieved 17 June 2012.
- ^ "Metals Content for Fertilizer and Soil Amendment Products". regulatory-info-sc.com. Retrieved 21 July 2022.
- doi:10.1016/j.gloenvcha.2016.08.005.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ Andrew, Hannah (5 July 2022). "Addressing Consolidation in Agriculture: USDA's Response to President Biden's Directive to Promote Competition in the American Economy" (PDF). Center for Agriculture and Food Systems, Vermont Law and Graduate School. p. 7. Archived (PDF) from the original on 20 July 2022. Retrieved 7 November 2022.
- S2CID 83736589– via Taylor & Francis Online.
Cited sources
- Mbow, C.; Rosenzweig, C.; Barioni, L. G.; Benton, T.; et al. (2019). "Chapter 5: Food Security" (PDF). Climate Change and Land: an IPCC special report on climate change, desertification, land degradation, sustainable land management, food security, and greenhouse gas fluxes in terrestrial ecosystems. p. 454.
 This article incorporates text from a free content work. Licensed under CC BY-SA IGO 3.0 (license statement/permission). Text taken from World Food and Agriculture – Statistical Yearbook 2023, FAO, FAO.
This article incorporates text from a free content work. Licensed under CC BY-SA IGO 3.0 (license statement/permission). Text taken from World Food and Agriculture – Statistical Yearbook 2023, FAO, FAO.
External links
- Nitrogen for Feeding Our Food, Its Earthly Origin, Haber Process
- International Fertilizer Industry Association (IFA)
- Agriculture Guide, Complete Guide to Fertilizers and Fertilization (archived 6 October 2011)
- Nitrogen-Phosphorus-Potassium Values of Organic Fertilizers. Archived 26 February 2021 at the Wayback Machine.

