Gap junction
| Gap junction | |
|---|---|
 Vertebrate gap junction | |
| Identifiers | |
| MeSH | D017629 |
| TH | H1.00.01.1.02024 |
| FMA | 67423 |
| Anatomical terminology | |
This article's tone or style may not reflect the encyclopedic tone used on Wikipedia. (April 2024) |
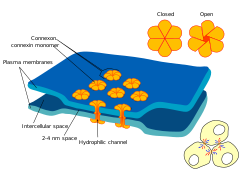
Gap junctions are one of three broad categories of intercellular connections that form between a multitude of animal cell types.[1][2] First photographed around 1952,[3][4] it wasn't until 1969 that gap junctions were referred to as gap junctions.[5] Named after the 2-4 nm gap they bridged between cell membranes, they had been characterised in more detail by 1967.[6]
Within a gap junction reside protein complexes (referred initially to as globules, observed to connect one cell to another) and vesicles within a cell to the outer cell membrane.[7] By 1974 one of the major gap junction proteins was dubbed a connexin,[8][9] and six connexins were observed to form a channel called a connexon due to the connections connexon pairs made between cells. The initial discovery of gap junctions in nerve cells lent credence to their function in transmission of electrical impulses. Experimental confirmation followed, with molecules, ions, and electrical impulses being shown to pass through the connexons; this proved them to be a generalized regulated gate between cells in gap junctions. A type of hemichannel connexon also forms channels to the extracellular regions as well.[10][11][12][13]
More than 26 different connexins are known to comprise gap junctions in various different tissues; there are at least 12 other components
The increasing ability to sequence the DNA of organisms also led to an increase in the complexity of the gap junction family of proteins. The term connexin was used to describe the gap junction proteins connecting two cells with pores. Sequencing of these pore proteins showed them to be structurally similar between vertebrates and invertebrates but different in sequence.[19] As a result, the term innexin was used to differentiate invertebrate connexins from vertebrate ones.[20] While the sequencing of invertebrate species is far less complete than for vertebrates, more than 20 innexins have already been uncovered,[21] along with unnexins in parasites and vinnexins in viruses.
A gap junction is less frequently called a nexus or macula communicans. While an ephapse, like a gap junction, also involves the transmission of electrical signals, the two are distinct from each other. Ephaptic coupling involves electrical signals external to the cells. Ephapses are often studied in the context of electrically induced potentials propagated among groups of nerve cell membranes, even in the absence of gap junction communication, with no discrete subcellular structures known.[22][23] Unlike gap junctions, no specific structure related to an ephapse has yet been described, so the process is often referred to as ephaptic coupling rather than as an ephapse.
Structure
Invertebrate gap junctions comprise proteins from the innexin family. Innexins have no significant sequence homology with connexins.[24] Though differing in sequence to connexins, innexins are similar enough to connexins to state that innexins form gap junctions in vivo in the same way connexins do.[25][26][27]
The more recently characterized
At gap junctions, the intercellular space is 2-4 nm.[33] Hemichannels in the membrane of each cell are aligned with one another.[34]
Gap junction channels formed from two identical hemichannels are called homotypic, while those with differing hemichannels are heterotypic. In turn, hemichannels of uniform protein composition are called
Before innexins and connexins were well characterized, the genes coding for the connexin gap junction channels were classified in one of three groups (A, B and C; for example, GJA1, GJC1), based on gene mapping and
However, connexin genes do not code directly for the expression of gap junction channels; genes can produce only the proteins that make up gap junction channels. An alternative naming system based on the protein's molecular weight is the most widely used (for example, connexin43=GJA1, connexin30.3=GJB4).Levels of organization
In vertebrates, two pairs of six connexin proteins form a connexon. In invertebrates, six innexin proteins form an innexon. Otherwise, the structures are similar. The term hemichannel used in this description refers to half a gap junction channel.
- The connexin genes (DNA) are transcribed to RNA, which is then translated to produce a connexin.
- One connexin protein has four transmembrane domains[33][39]
- Six connexin proteins create one connexon channel (hemichannel). When identical connexin proteins join to form one connexon, it is called a homomeric connexon. When different connexin proteins join to form one connexon, it is called a heteromeric connexon.
- Two connexons, joined across a cell membrane, comprise a Gap Junction channel.
When two identical connexons come together to form a Gap junction channel, it is called a homotypic channel. When one homomeric connexon and one heteromeric connexon come together, it is called a heterotypic gap junction channel. When two heteromeric connexons join, it is also called a heterotypic gap junction channel. - Tens to thousands of gap junction channels cluster in areas to enable connexon pairs to form.
Properties of connexon pairs

A connexon or innexon channel pair:
- Allows for direct electrical communication between cells, although different hemichannel subunits can impart different single channel conductances, from about 30 pS to 500 pS.
- Allows for chemical communication between cells through the transmission of small Ca2+although different hemichannel subunits can impart different selectivities for particular molecules.
),[42] - Generally allows transmembrane movement of molecules smaller than 485 daltons[43] (1,100 daltons through invertebrate gap junctions[44]), although different hemichannel subunits may impart different pore sizes and different charge selectivity. Large biomolecules, including nucleic acids and proteins, are precluded from cytoplasmic transfer between cells through gap junction hemichannel pairs.
- Ensures that molecules and current passing through the gap junction do not leak into the intercellular space.
Properties of connexons as hemichannels
Unpaired connexons or innexons can act as hemichannels in a single membrane, allowing the cell to exchange molecules directly with the exterior of the cell. It has been shown that connexons would be available to do this prior to being incorporated into the gap junction plaques.[40] Some of the properties of these unpaired connexons are listed below:
- Pore or transmembrane channel size is highly variable, in the range of approximately 8-20Å in diameter.[45]
- They connect the cytoplasm of the cell to the cell exterior and are thought to be in a closed state by default in order to prevent leakage from the cell.[46][47]
- Some connexons respond to external factors by opening up. Mechanical shear and various diseases can cause this to happen.[48]
Establishing further connexon properties different to those of connexon pairs proves difficult due to separating their effects experimentally in organisms.[48]
Occurrence and distribution
Gap Junctions have been observed in various animal organs and tissues where cells contact each other. From the 1950s to 1970s they were detected in:
- Human islet of Langerhans,[49] myometrium,[50] and eye lens[51]
- Rat pancreas, liver, adrenal cortex, epididymis, duodenum, muscle,[52] and seminiferous tubules[53]
- Rabbit cornea,[54] ovary,[55] and skin[56]
- Monkey retina[57]
- Chick embryos[58]
- Frog embryos[59]
- Fish blastoderm[60]
- Crayfish nerves[61]
- Lamprey and Tunicate heart[62][63]
- Goldfish and hamster pressure-sensing acoustico-vestibular receptors[64]
- Daphnia hepatic caecum[65]
- Cephalopod digestive epithelium[66]
- Hydra muscle[67]
- Cockroach hemocyte capsules[68]
- Reaggregated cells[69][70]
Since the 1970s, gap junctions have continued to be found in nearly all animal cells that touch each other. By the 1990s new technology such as confocal microscopy allowed more rapid survey of large areas of tissue. Since the 1970s tissues that were traditionally considered to have isolated cells (such as bone) were shown to have cells that were still connected with gap junctions, however tenuously.[71] Gap junctions appear to be in all animal organs and tissues; there have been no exceptions to this other than cells not normally in contact with neighboring cells. Adult skeletal muscle is a possible exception. If present in skeletal muscle, gap junctions may propagate contractions in an arbitrary way through cells making up the muscle. In some cases this may not be the case, as shown in other muscle types that do have gap junctions.[72] An indication of what results from reduction or absence of gap junctions may be found by analyses of cancers[73][74][75] or the aging process.[76]
As we learn more, the term gap junction cannot be defined by a single protein such as a connexin. The term appears to be an accurate description of the regular intercellular gap as it was originally coined for rather than a junction defined by a single family of proteins. One example of this includes the gap junction-like structures found in
Functions
At least five discrete functions have been ascribed to gap junction proteins:
- Electrical and metabolic coupling between cells
- Electrical and metabolic exchange through hemichannels
- Tumor suppressor genes (Cx43, Cx32 and Cx36)
- Adhesive function independent of conductive gap junction channel (neural migration in neocortex)
- Role of carboxyl-terminal in signaling cytoplasmic pathways (Cx43)
In a more general sense, gap junctions may be seen to function at the simplest level as a direct cell to cell pathway for electrical currents, small molecules and ions. The control of this communication allows complex downstream effects on multicellular organisms.
Embryonic, organ and tissue development
In the 1980s, more subtle roles of gap junctions in communication have been investigated. It was discovered that gap junction communication could be disrupted by adding anti-connexin antibodies into embryonic cells.[84][85] Embryos with areas of blocked gap junctions failed to develop normally. The mechanism by which antibodies blocked the gap junctions was unclear; systematic studies were undertaken to elucidate the mechanism.[86][87] Refinement of these studies suggested that gap junctions were key in the development of cell polarity[88] and the left-right symmetry in animals.[89][90] While signaling that determines the position of body organs appears to rely on gap junctions, so does the more fundamental differentiation of cells at later stages of embryonic development.[91][92][93][94][95]
Gap junctions were found to be responsible for the transmission of signals required for drugs to have an effect.[96] Conversely, some drugs were shown to block gap junction channels.[97]
The bystander effect and disease
Cell death
The bystander effect has its connotations of the innocent bystander being killed. When cells are dying or compromised due to disease or injury, messages are transmitted to neighboring cells by gap junctions. This can cause otherwise healthy bystander cells to also die.[98] The bystander effect is, therefore, important to consider in diseased cells, opening an avenue for more funding and a flourish of research.[99][100][101][102][103][104][105][106][107]
The bystander effect was later researched with regard to cells damaged by radiation or mechanical injury and in turn wound healing.[108][109][110][111][112] Disease seems to have an effect on the ability of gap junctions to fulfill their roles in wound healing.[113][114] The oral administration of gap junction blockers to reduce the symptoms of disease in remote parts of the body is slowly becoming a reality.[115]
Tissue restructuring
While there has been a tendency to focus on the bystander effect in disease due to the possibility of therapeutic avenues, there is evidence that there is a more central role in normal development of tissues. Death of some cells and their surrounding matrix may be required for a tissue to reach its final configuration; gap junctions appear essential to this process.[116][117] There are also more complex studies that try to combine our understanding of the simultaneous roles of gap junctions in both wound healing and tissue development.[118][119][120]
Disease
Mutations in connexins have been associated with many diseases in humans, including deafness, heart atrial fibrillation (standstill) and cataracts. The study of these mutations has helped clarify some of the functions of connexins.[121][122]
Hemichannels are thought to play a general role in the progression and severity of many diseases; this is in part due to hemichannels being an open door to the outside of each cell.[48]
Areas of electrical coupling
Gap junctions electrically and chemically couple cells throughout the body of most animals. Electrical coupling can be relatively fast acting and can be used over short distances within an organism. Tissues in this section have well known functions observed to be coordinated by gap junctions, with intercellular signaling happening in time frames of microseconds or less.
Heart

Gap junctions are particularly important in cardiac muscle: the signal to contract is passed efficiently through gap junctions, allowing the heart muscle cells to contract in unison. The importance is emphasized by a secondary ephaptic pathway for the signal to contract also being associated with the gap junction plaques. This redundancy in signal transmission associated with gap junction plaques is the first to be described and involves sodium channels rather than connexins.[16][123]
Eye lens
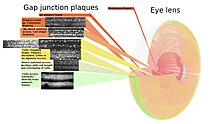
Precise control of light refraction, structural dimensions and transparency are key aspects of the eye lens structure that allow focusing by the eye. Transparency is aided by the absence of nerves and blood vessels from the lens, so gap junctions are left with a larger loading of intercellular communication than in other tissues reflected in large numbers of gap junctions. The crystallinity of the lens also means the cells and gap junctions are well ordered for systematic mapping of where the gap junction plaques are. As no cells are lost from the lens interior during the life of the animal, a complete map of the gap junctions is possible.[124]
The associated figure shows how the size, shape, and frequency of gap junction plaques change with cell growth. With growth, fiber cells are progressively isolated from more direct metabolite exchange with the aqueous humor through the capsule and lens epithelium. The isolation correlates with the classical circular shape of larger plaques shown in the yellow zone being disrupted. Changing the fiber cells' morphology requires the movements of vesicles through the gap junction plaques at higher frequencies in this area.[125]
Neurons
A gap junction located in a neuron is often referred to as an
There has been some observation of coupling in the
Connexin proteins expressed in neuronal gap junctions include m
Retina
Neurons within the retina show extensive coupling, both within populations of one cell type and between different cell types.[128]
Uterus
The
Hemichannel function
Hemichannels contribute to a cellular network of gap junctions and allow the release of
On a larger scale, the one-to-many communication of cells is typically carried out by the vascular and nervous systems. This makes detecting the contribution of hemichannels to extracellular communication more difficult in whole organisms. With the eye lens, the vascular and nervous systems are absent, making reliance on hemichannels greater and their detection easier. At the interface of the lens with the aqueous humor (where the lens exchanges metabolites), both gap junction plaques and more diffused connexon distribution can be seen in the accompanying micrographs.
Discovery
Form to function
Well before the demonstration of the gap in gap junctions, they were seen at the junction of neighboring nerve cells. The close proximity of the neighboring cell membranes at the gap junction led researchers to speculate that they had a role in intercellular communication, in particular the transmission of electrical signals.[61][132][133] Gap junctions were also proven to be electrically rectifying and referred to as an electrical synapse.[134][135] Later, it was found that chemicals could also be transported between cells through gap junctions.[136]
Implicit or explicit in most of the early studies is that the area of the gap junction was different in structure to the surrounding membranes in a way that made it look different. The gap junction had been shown to create a micro-environment between the two cells in the extracellular space or gap. This portion of extracellular space was somewhat isolated from the surrounding space and also bridged by what we now call connexon pairs, which form even more tightly sealed bridges that cross the gap junction gap between two cells. When viewed in the plane of the membrane by freeze-fracture techniques, higher-resolution distribution of connexons within the gap junction plaque is possible.[137]
Connexin free islands are observed in some junctions. The observation was largely without explanation until vesicles were shown by Peracchia using transmission electron microscopy (TEM) thin sections to be systematically associated with gap junction plaques.[138] Peracchia's study was probably also the first study to describe paired connexon structures, which he called a globule. Studies showing vesicles associated with gap junctions and proposing the vesicle contents may move across the junction plaques between two cells were rare, as most studies focused on connexons rather than vesicles. A later study using a combination of microscopy techniques confirmed the early evidence of a probable function for gap junctions in intercellular vesicle transfer. Areas of vesicle transfer were associated with connexin free islands within gap junction plaques.[125] Connexin 43 has been shown to be necessary for the transfer of whole mitochondrias to neighboring cells, though whether the mitochondria is transferred directly through the membrane or within a vesicle has not been determined [139]
Electrical and chemical nerve synapses
Because of the widespread occurrence of gap junctions in cell types other than nerve cells, the term gap junction became more generally used than terms such as electrical synapse or nexus. Another dimension in the relationship between nerve cells and gap junctions was revealed by studying chemical synapse formation and gap junction presence. By tracing nerve development in leeches with gap junction expression suppressed it was shown that the bidirectional gap junction (electrical nerve synapse) needs to form between two cells before they can grow to form a unidirectional chemical nerve synapse.[140] The chemical nerve synapse is the synapse most often truncated to the more ambiguous term nerve synapse.
Composition
Connexins
The purification
The ultrastructure and biochemistry of isolated gap junctions already referenced had indicated the connexins preferentially group in gap junction plaques or domains and connexins were the best characterized constituent. It has been noted that the organisation of proteins into arrays with a gap junction plaque may be significant.[59][150] It is likely this early work was already reflecting the presence of more than just connexins in gap junctions. Combining the emerging fields of freeze-fracture to see inside membranes and immunocytochemistry to label cell components (Freeze-fracture replica immunolabelling or FRIL and thin section immunolabelling) showed gap junction plaques in vivo contained the connexin protein.[151][124] Later studies using immunofluorescence microscopy of larger areas of tissue clarified diversity in earlier results. Gap junction plaques were confirmed to have variable composition being home to connexon and non-connexin proteins as well making the modern usage of the terms "gap junction" and "gap junction plaque" non-interchangeable.[18] To summarize, in early literature the term "gap junction" referred to the regular gap between membranes in vertebrates and non-vertebrates apparently bridged by "globules". The junction correlated with the cell's ability to directly couple with its neighbors through pores in their membranes. Then for a while gap junctions were only referring to a structure that contains connexins and nothing more was thought to be involved. Later, the gap junction "plaque" was also found to contain other molecules that helped define it and make it function.
The "plaque" or "formation plaque"
Early descriptions of gap junctions, connexons or innexons did not refer to them as such; many other terms were used. It is likely that synaptic disks[152] were an accurate reference to gap junction plaques. While the detailed structure and function of the connexon was described in a limited way at the time the gross disk structure was relatively large and easily seen by various TEM techniques. Disks allowed researchers using TEM to easily locate the connexons contained within the disk like patches in vivo and in vitro. The disk or plaque appeared to have structural properties different from those imparted by the connexons/innexons alone.[67] It was thought that if the area of membrane in the plaque transmitted signals, the area of membrane would have to be sealed in some way to prevent leakage.[153] Later studies showed gap junction plaques are home to non-connexin proteins, making the modern usage of the terms "gap junction" and "gap junction plaque" non-interchangeable as the area of the gap junction plaque may contain proteins other than connexins.[18][125] Just as connexins do not always occupy the entire area of the plaque, the other components described in the literature may be only long-term or short-term residents.[154][35][155]
Studies allowing views inside the plane of the membrane of gap junctions during formation indicated that a "formation plaque" formed between two cells prior to the connexins moving in. They were particle free areas—when observed by TEM FF, indicated very small or no transmembrane proteins were likely present. Little is known about what structures make up the formation plaque or how the formation plaque's structure changes when connexins and other components move in and out. One of the earlier studies of the formation of small gap junctions describes rows of particles and particle free halos.[156] With larger gap junctions they were described as formation plaques with connexins moving into them. The particulate gap junctions were thought to form 4–6 hours after the formation plaques appeared.[157] How the connexins may be transported to the plaques using tubulin is becoming clearer.[88][158]
The formation of plaque and the non-connexin part of the classical gap junction plaque have been difficult for early researchers to analyse. It appears in TEM FF and thin section to be a lipid membrane domain that can somehow form a comparatively rigid barrier to other lipids and proteins. There has been indirect evidence for certain lipids being preferentially involved with the formation plaque, however this cannot be considered definitive.[159][160] It is difficult to envisage breaking up the membrane to analyse membrane plaques without affecting their composition. By study of connexins still in membranes lipids associated with the connexins have been studied.[161] It was found that specific connexins tended to associate preferentially with specific phospholipids. As formation plaques precede connexins these results still give no certainty as to what is unique about the composition of plaques themselves. Other findings show connexins associate with protein scaffolds used in another junction, the zonula occludens ZO-1.[162] While this helps us understand how connexins may be moved into a gap junction formation plaque, the composition of the plaque itself is still somewhat sketchy. Some headway on the in vivo composition of the gap junction plaque is being made using TEM FRIL.[154][162]
See also
- Gap junction modulation
- Gap junction protein
- Innexin
- Vinnexin
- Intercalated disc
- Ion channel
- Junctional complex
- Tight junction
References
- PMID 11146276.
- S2CID 22486987.
- ^ Robertson, J. D. (1952). Ultrastructure of an invertebrate synapse (Thesis). Cambridge, MA: Massachusetts Institute of Technology.
- PMID 13037850.
- PMID 5765759.
- PMID 6036535.
- ^ PMID 4120611.
- PMID 4363961.
- PMID 2826492.
- S2CID 4216387.
- PMID 15109565.
- PMID 11368307.
- PMID 18853183.
- PMID 17507078.
- ^ PMID 19704902.
- ^ PMID 34533834.
- PMID 15802270.
- ^ PMID 2691517.
- S2CID 205003383.
- PMID 9769729.
- PMID 36740225.
- PMID 32069981.
- PMID 36619115.
- PMID 9851916.
- PMID 10079517.
- PMID 8707836.
- PMID 24575048.
- PMID 16565476.
- S2CID 20001454.
- PMID 24548565.
- PMID 21532340.
- PMID 30813901.
- ^ S2CID 4431769.
- PMID 9514740.
- ^ PMID 17507078.
- S2CID 44622463.
- PMID 1320430.
- PMID 8227133.
- PMID 27905396.
- ^ PMID 12149451.
- PMID 19116647.
- ISBN 978-0-8153-3218-3.[page needed]
- S2CID 19289550.
- S2CID 22820528.
- PMID 34129834.
- S2CID 18103080.
- PMID 30310236.
- ^ PMID 33800706.
- PMID 1102552.[permanent dead link]
- S2CID 31610495.
- PMID 511455.
- PMID 4337577.
- S2CID 19611753.
- PMID 976372.
- PMID 4417791.
- PMID 1109802.
- PMID 4198274.
- S2CID 38441429.
- ^ a b J. Cell Biol. 1974 Jul;62(1) 32-47.Assembly of gap junctions during amphibian neurulation. Decker RS, Friend DS.
- PMID 5545331.
- ^ S2CID 39294652.
- S2CID 21426059.
- S2CID 21604678.
- S2CID 30090247.
- PMID 5563454.
- PMID 7414602.
- ^ PMID 4109925.
- PMID 1179417.
- PMID 4373716.
- PMID 96453.
- S2CID 33191311.
- PMID 15679888.
- S2CID 2831462.
- PMID 270694.
- PMID 11145599.
- PMID 761600.
- PMID 15921654.
- PMID 16359939.
- S2CID 244116450.
- PMID 16005277.
- PMID 36619868.
- ^ Bergquist, P.R.; Green, C.R. (1977). "An ultrastructural study of settlement and metamorphosis in sponge larvae". Cah. Biol. Mar. 18: 289–302.
- ^ Green, C.R.; Bergquist, P.R. (1979). "Cell membrane specializations in the Porifera". Coll Int Cent Natn Res Scient. 291: 153–158.
- S2CID 2620476.
- )
- S2CID 20408672.
- S2CID 23234120.
- ^ PMID 22022608.
- PMID 9806775.
- PMID 10518488.
- PMID 10079512.
- S2CID 41420671.
- S2CID 9894657.
- PMID 12826585.
- S2CID 5057466.
- PMID 10523421.
- PMID 11535816.
- PMID 8186287.
- S2CID 41136083.
- PMID 7548276.
- PMID 7479939.
- PMID 8929915.
- PMID 8700844.
- PMID 8622159.
- PMID 8665496.
- PMID 8986442.
- PMID 12014914.
- PMID 12194273.
- PMID 12194291.
- S2CID 44821116.
- S2CID 25113646.
- PMID 15927148.
- PMID 17717278.
- PMID 9550065.
- PMID 36835288.
- PMID 12878681.
- S2CID 24363587.
- PMID 16628254.
- PMID 19966054.
- PMID 19528242.
- PMID 28457858.
- PMID 10099690.
- PMID 36359872.
- ^ .
- ^ PMID 30813901.
- PMID 15217338.
- PMID 18236012.
- PMID 19491906.
- ^ Garfield, RE; Sims, SM; Kannan, MS; Daniel, EE (November 1978). "Possible role of gap junctions in activation of myometrium during parturition". Am. J. Physiol. 235 (5): C168–79. doi:10.1152/ajpcell.1978.235.5.C168. PMID 727239. S2CID [https://www.semanticscholar.org/paper/Possible-role-of-gap-junctions-in-activation-of-Garfield-Sims/ce78aa441efb2f15ff4749d082197ed4db1b2584 31610495
- ^ Boros-Rausch, A., Shynlova, O., & Lye, S. J. (2021). "A Broad-Spectrum Chemokine Inhibitor Blocks Inflammation-Induced Myometrial Myocyte-Macrophage Crosstalk and Myometrial Contraction". Cells. 11 (1): 128. doi: 10.3390/cells11010128 PMID 35011690
- PMID 16761954.
- OCLC 261587041.[page needed]
- PMID 7033238.
- S2CID 4216387.
- PMID 13642302.
- S2CID 24701801.
- PMID 4935338.
- PMID 4120610.
- PMID 22504485.
- PMID 21068332.
- PMID 4339819.
- PMID 4363961.
- PMID 2875078.
- PMID 5531667.
- PMID 4935338.
- S2CID 39342230.
- PMID 2987225.
- PMID 2826492.
- PMID 3891760.
- PMID 4504340.
- PMID 3818793.
- PMID 14069795.
- PMID 14206423.
- ^ PMID 21858170.
- PMID 33321985.
- PMID 4135001.
- PMID 1083855.
- PMID 12149451.
- PMID 1698798.
- PMID 22049024.
- PMID 19686581.
- ^ PMID 18815262.
Further reading
- Harris, Andrew; Locke, Darren, eds. (2009). Connexins. New York: Springer. ISBN 978-1-934115-46-6.
External links
- Gap+Junctions at the U.S. National Library of Medicine Medical Subject Headings (MeSH)


