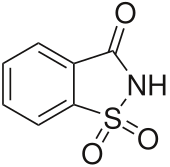
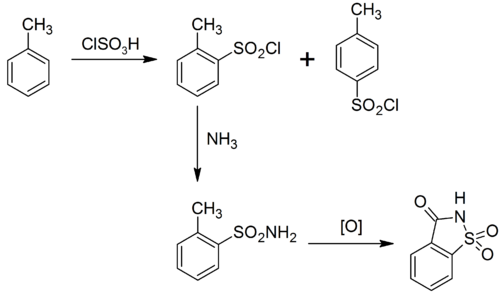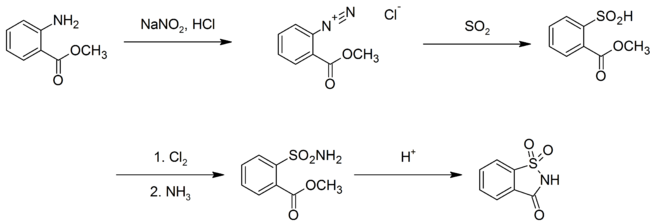Saccharin
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
1H-1λ6,2-Benzothiazole-1,1,3(2H)-trione | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.001.202 |
| E number | E954 (glazing agents, ...) |
IUPHAR/BPS |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C7H5NO3S | |
| Molar mass | 183.18 g·mol−1 |
| Appearance | White crystalline solid |
| Density | 0.828 g/cm3 |
| Melting point | 228.8 to 229.7 °C (443.8 to 445.5 °F; 501.9 to 502.8 K) |
| 1 g per 290 mL | |
| Acidity (pKa) | 1.6[4] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Saccharin, also called saccharine or benzosulfimide, or used in saccharin sodium or saccharin calcium forms, is a non-nutritive
Etymology
Saccharin derives its name from the word "saccharine", meaning "sugary". The word saccharine is used figuratively, often in a derogative sense, to describe something "unpleasantly over-polite" or "overly sweet". (table sugar).
Properties

Saccharin is heat-stable.
In its acid form, saccharin is not water-soluble. The form used as an artificial sweetener is usually its
Safety and health effects
In the 1970s, studies performed on laboratory rats found an association between consumption of high doses of saccharin and the development of
Saccharin has no food energy and no nutritional value.[15] It is safe to consume for individuals with diabetes or prediabetes.[16][17]
People with sulfonamide allergies can experience allergic reactions to saccharin, although it has been suggested that this may be due to a general predisposition to allergic reactions rather than a specific cross-reaction between antimicrobial sulfonamides and non-antimicrobial ones (like saccharin).[18][19] Saccharin in toothpaste can cause burning sensations, swelling, and rashes of the mouth and lips in sensitive individuals.[20]
History


Saccharin was produced first in 1879, by
Although saccharin was commercialized not long after its discovery, until sugar shortages during World War I, its use had not become widespread. Its popularity further increased during the 1960s and 1970s among dieters, since saccharin is a calorie-free sweetener. In the United States, saccharin is often found in restaurants in pink packets; the most popular brand is "Sweet'n Low".
Because of the difficulty of importing sugar from the West Indies, the British Saccharin Company was founded in 1917 to produce saccharin at its Paragon Works near Accrington, Lancashire. Production was licensed and controlled by the Board of Trade in London. Production continued on the site until 1926.
Government regulation
The examples and perspective in this section deal primarily with the United States and do not represent a worldwide view of the subject. (October 2015) |
Starting in 1907, the
In 1911, Food Inspection Decision 135 stated that foods containing saccharin were
More controversy was stirred in 1969 with the discovery of files from the FDA's investigations of 1948 and 1949. These investigations, which had originally argued against saccharin use, were shown to prove little about saccharin being harmful to human health.[citation needed] In 1977, the FDA made an attempt to completely ban the substance,[11][30] following studies showing that the substance caused bladder cancer in rats. The attempted ban was unsuccessful due to public opposition that was encouraged by industry advertisements,[30] and instead the following label was mandated: "Use of this product may be hazardous to your health. This product contains saccharin which has been determined to cause cancer in laboratory animals". That requirement was dropped in 2000 following new research that concluded humans reacted differently than rats and were not at risk of cancer at typical intake levels.[30] (See also: § Warning label addition and removal below.) The sweetener has continued to be widely used in the United States and is now the third-most popular artificial sweetener behind sucralose and aspartame.
In the European Union, saccharin is also known by the E number (additive code) E954.
The current status of saccharin is that it is allowed in most countries, and countries such as Canada have lifted their previous ban of it as a food additive.[31] The claims that it is associated with bladder cancer were shown to be unfounded in experiments on primates.[32] (It is, however, prohibited to mail saccharin tablets or packets to France.[33])
Saccharin was formerly on California's list of chemicals known to the state to cause cancer for the purposes of
Warning label addition and removal
In 1958, the United States Congress amended the
However, in 2000, the warning labels were removed because scientists learned that rodents, unlike humans, have a unique combination of high pH, high calcium phosphate, and high protein levels in their urine.[36][37] One or more of the proteins that are more prevalent in male rats combine with calcium phosphate and saccharin to produce microcrystals that damage the lining of the bladder. Over time, the rat's bladder responds to this damage by overproducing cells to repair the damage, which leads to tumor formation. Since this does not occur in humans, there is no elevated risk of bladder cancer.[38]
The delisting of saccharin led to legislation repealing the warning label requirement for products containing saccharin.
The
Chemistry
Preparation
Saccharin can be produced in various ways.
In 1950, an improved synthesis was developed at the Maumee Chemical Company of Toledo, Ohio. In this synthesis, the methyl anthranilate successively reacts with nitrous acid (from sodium nitrite and hydrochloric acid), sulfur dioxide, chlorine, and then ammonia to yield saccharin:[43]
Properties and reactions
The free acid of saccharin has a low
See also
- Aspartame
- Neotame
- Saccharose
- Sodium cyclamate
- Steviol glycoside
- Sugar substitute
- Sucralose
- Xylitol
- Phthalimide
References
- ^ a b c d e "Saccharin". PubChem, US National Library of Medicine. 13 June 2023. Retrieved 15 June 2023.
- ^ "Saccharin (CAS: 81-07-2)". Merck Millipore. 2023. Retrieved August 22, 2022.
- ISBN 81-7450-716-7.
- ^ .
- ^ a b "Saccharin". Drugs.com. 16 August 2022. Retrieved 15 June 2023.
- ^ "Saccharine". Reference.com. Archived from the original on 2007-03-03.
- ^ "Saccharine". etymonline.com. Archived from the original on 2006-03-23.
- ^ "Sweetener Comparisons". Food Ingredient Series. NCSU. 2006. Archived from the original on 2019-01-20.
- PMID 24741154.
- ^ from the original on 2013-05-13.
- ^ S2CID 40526005.
- ^ a b c "Artificial Sweeteners and Cancer". National Cancer Institute. 2005-08-18. Archived from the original on 2015-12-08.
- PMID 15367404.
- ^ "Saccharin: FDA Agencies". University of Minnesota, Environmental Health Sciences. Archived from the original on 2016-02-27.
- ^ "Common Terms: S–Z". American Diabetes Association. Archived from the original on 2015-11-28.
- ^ "Low-Calorie Sweeteners: What's News, What's New". American Diabetes Association. Archived from the original on 2016-03-04.
- ^ "Are Artificial Sweeteners Safe for People With Diabetes?". Cleveland Clinic. 2015-06-29. Archived from the original on 2016-10-02.
- PMID 14573734.
- PMID 31514363.
- ^ "Toothpaste Allergy: Yes, You Can be Allergic to Toothpaste". Archived from the original on 2020-03-29. Retrieved 2020-03-29.
- ^ (As discussed below, the relative contributions of Fahlberg and Remsen to the discovery were later contested, with no final resolution in sight; the 1879 paper announcing the discovery lists both names as authors, with Fahlberg's name first.)
- ^ Fahlberg's account of how he discovered the sweetness of saccharin appears in: Anon. (July 17, 1886). "The inventor of saccharine". Scientific American. new series. 60 (3): 36. Archived from the original on 2017-03-14.
- ISBN 978-0-313-33758-1.
- ^ Remsen I, Fahlberg C (February 1880). "On the Oxidation of Substitution Products of Aromatic Hydrocarbons. IV. – On the oxidation of orthotoluenesulphamide". American Chemical Journal. 1 (6): 426–439. Archived from the original on 2021-02-24. Retrieved 2022-02-24. From pages 430–431:
- "It possesses a 'very marked sweet taste, being much sweeter than cane-sugar'. The taste is perfectly pure. The minutest quantity of the substance, a bit of its powder scarcely visible, if placed upon the tip of the tongue, causes a sensation of pleasant sweetness throughout the entire cavity of the mouth. As stated above, the substance is soluble to only a slight extent in cold water, but if a few drops of the cold aqueous solution be placed in an ordinary goblet full of water, the latter then tastes like the sweetest syrup. Its presence can hence easily be detected in the dilutest solutions by the taste."
- ^ US 319082, Fahlberg, C. & List, A., "Manufacture of saccharine compounds"
- OL 6411359M.
- ^ "Sugar: A Cautionary Tale". FDA.gov. Archived from the original on 2010-04-28. Retrieved 2010-06-20.
- ^ Dunn CW (1913). Federal, State, and Territorial Reference Manual of Pure Food and Drug Law: Dunn's Pure Food and Drug Legal Manual. p. 1327. Archived from the original on 2017-03-13.
- ^ "Monthly Bulletin". California State Board of Health. 1921. p. 21. Archived from the original on 2017-03-14.
- ^ a b c d Conis E (December 27, 2010). "Saccharin's mostly sweet following". Los Angeles Times. Archived from the original on 2011-02-17. Retrieved 2011-01-14.
- ^ "Saccharin cleared for use in foods in Canada". AgCanada.com. Health Canada. Archived from the original on 2021-05-15. Retrieved 2022-02-24.
- PMID 9428778.
- ^ "USPS Mailing Conditions". Archived from the original on 2013-04-04. Retrieved 2013-04-07.
- ^ Sun M (28 December 2015). "Saccharin Delisted Effective April 6, 2001 as Known to the State to Cause Cancer". CA.gov. Archived from the original on 10 March 2011.
- ^ "Saccharin warning". Associated Press via Telegraph-Herald. 1973-05-22. Archived from the original on 2021-12-23. Retrieved 2011-06-09.
- PMID 8910956.
- PMID 12505296.
- ^ "Report on Carcinogens, Thirteenth Edition, Appendix B" (PDF). National Toxicology Program. pp. 2–4. Archived from the original (PDF) on 2016-05-20.
Linked from "Saccharin index page". National Toxicology Program. November 18, 2014. Archived from the original on 2016-03-07. Retrieved 2016-02-29. - ^ "Artificial Sweeteners and Cancer". National Cancer Institute. August 5, 2009. Archived from the original on 2015-12-08. Retrieved 2016-02-29.
- ^ "EPA Removes Saccharin from Hazardous Substances Listing". December 14, 2010. Archived from the original on 2010-12-24. Retrieved 2011-01-14.
- .
- ^ Bungard G (1967). "Die Süßstoffe" [Sweeteners]. Der Deutscher Apotheker. 19: 150.
- ^ ISBN 978-3527306732.
- PMID 23134535.
- .
- from the original on 2017-10-03.
External links
 Media related to Saccharin at Wikimedia Commons
Media related to Saccharin at Wikimedia Commons